Abstract
Plasma metanephrines have become the biochemical test of choice for suspected phaeochromocytomas and paragangliomas in many institutions. We encountered two separate cases of significantly elevated plasma metanephrines in patients taking midodrine, a sympathomimetic drug used in the treatment of severe postural hypotension, in the absence of a diagnosis of phaeochromocytomas and paragangliomas. Upon stopping midodrine treatment, plasma metanephrine concentrations returned to normal in both patients. To explore the hypothesis that midodrine or its metabolite desglymidodrine might interfere with the metanephrines assay, we tested the interaction of midodrine with metanephrine assays from two different centres. High-performance liquid chromatography tandem mass spectrometry on plasma samples and on methanolic extract of midodrine demonstrated co-elution of the metabolite desglymidodrine with metanephrine. We conclude that patients taking midodrine may have falsely elevated plasma metanephrine as a result of analytical interference, and clinicians need to be aware of this problem.
Keywords
Introduction
Phaeochromocytomas and paragangliomas (PPGL) are rare adrenal and extra-adrenal tumours of chromaffin cells. The secretion of catecholamines from these cells leads to the characteristic but by no means universal features of sweating, hypertension, headaches and palpitations, with the potential for serious cardiovascular complications if left untreated. 1 Although serious, these tumours are also relatively rare, with an annual incidence of 1.55 to eight cases per million. 2 This combination presents a problem for the laboratory assessment for PPGL: test procedures must be sensitive enough not to miss a diagnosis with potentially fatal complications, but specific enough to exclude what is, in very many cases, unlikely to be the cause of a patient’s symptoms.
The initial diagnosis of PPGL is made through biochemical testing. Classically, plasma or urinary catecholamines or urinary vanillylmandelic acid (VMA) have been used to establish a diagnosis. However, VMA measurements are of poor accuracy, and catecholamines may be secreted intermittently. Most recently, plasma or urinary metanephrines have become the biochemical test of choice, as catecholamines are metabolized to metanephrines within tumours. 3 It has been suggested that free plasma metanephrines may serve as the single best test for both the confirmation and exclusion of PPGL, with 99% sensitivity and 89% specificity demonstrated in one trial. 4 This compared favourably to urinary fractionated metanephrines, but neither of which could give the combination of high sensitivity and specificity in one test. However, the specificity of plasma metanephrines as a stand-alone investigation has been disputed, with further research showing that the false-positive rate is too high to confidently confirm PPGL, especially in cases where the pretest probability of phaeochromocytoma is low.2,5,6 Others have supported the use of 24 h urinary metanephrines in these circumstances, but it is generally agreed that assessment of metanephrines, either plasma or urinary, is now the prime diagnostic test.
It is essential to avoid false-positive tests in the biochemical assessment for PPGLs. 1 Some cases are due to concurrent use of medications which raise endogenous catecholamine secretion, including monoamine oxidase inhibitors, halothane, serotonin–norepinephrine/noradrenaline reuptake inhibitors, and cocaine derivatives, e.g. lidocaine. In other cases, pathophysiological conditions such as heart failure and cardiogenic shock can increase circulating catecholamine concentrations. Finally, there is the possibility of analytical interference with the diagnostic assay. Such analytical interference has previously been documented for a number of drugs on measurement of metanephrines by liquid chromatography electrochemical detection, including labetalol, sotalol, buspirone, methyldopa and paracetamol.
We report two patients with clinically inexplicable elevations in plasma metanephrine only when taking the medication midodrine. In addition, we explore the evidence for analytical interference in plasma metanephrines performed on hydrophobic interaction liquid chromatography (HILIC)-based high-performance liquid chromatography tandem mass spectrometry (HPLC-MS/MS) caused by this medication.
Patients
Patient 1
A 41-year-old female was referred to the Oxford Endocrinology Clinic with suspected PPGL. She presented with a six-year history of discrete episodes of dizziness, nausea, dyspnoea and headaches, sometimes accompanied by syncope. Examination revealed sinus tachycardia and hypotension during these episodes, with no other abnormalities. There was no relevant past medical or family history of note. Her medications consisted of bisoprolol, doxazosin, slow sodium and midodrine 7.5 mg 3 hourly, which was started due to a presumed diagnosis of Postural Orthostatic Tachycardia Syndrome. Written consent was obtained prior to participation in the study.
Plasma metanephrines were significantly elevated, with a metanephrine concentration of >25,000 pmol/L (normal < 510 pmol/L) and normetanephrine concentration of 1758 pmol/L (normal < 1180 pmol/L). The plasma metanephrine concentrations remained elevated on repeat testing, whereas 24 h urinary metanephrines, parathyroid hormone, thyroid function tests and a full pituitary profile were normal, as was a CT scan of her adrenals. Upon withholding midodrine, plasma metanephrines returned to normal within one week (Table 1).
Effects of midodrine on plasma metanephrines.

Patient 2
A 35-year-old male presented to Auckland City Hospital Cardiology Department with postural hypotension and unexplained dilated cardiomyopathy. His echocardiogram demonstrated severe global left ventricular dysfunction with a left ventricular ejection fraction of 20–25%. His background history includes a 10-year history of type 2 diabetes and chronic kidney disease. Written consent was obtained.
Midodrine was started as treatment for postural hypotension. Plasma metanephrine was requested to exclude PPGL as an atypical cause for postural hypotension and unexplained cardiomyopathy. A gross elevation in a plasma metanephrine of 72,000 pmol/L (normal < 500 pmol/L) with an abnormal qualifier to quantifier ion ratio was noted. Given suspicions of interference caused by the medication, a repeat plasma metanephrine was requested following the termination of midodrine, which was within the reference interval (Table 1).
Method
Midodrine interference was evaluated using two HILIC-based HPLC-MS/MS assays from different centres: Auckland City Hospital and the Royal Victoria Infirmary, Newcastle (see Table 2 for full details of both assays).
Measurement of free plasma metanephrine by HPLC-MS/MS.
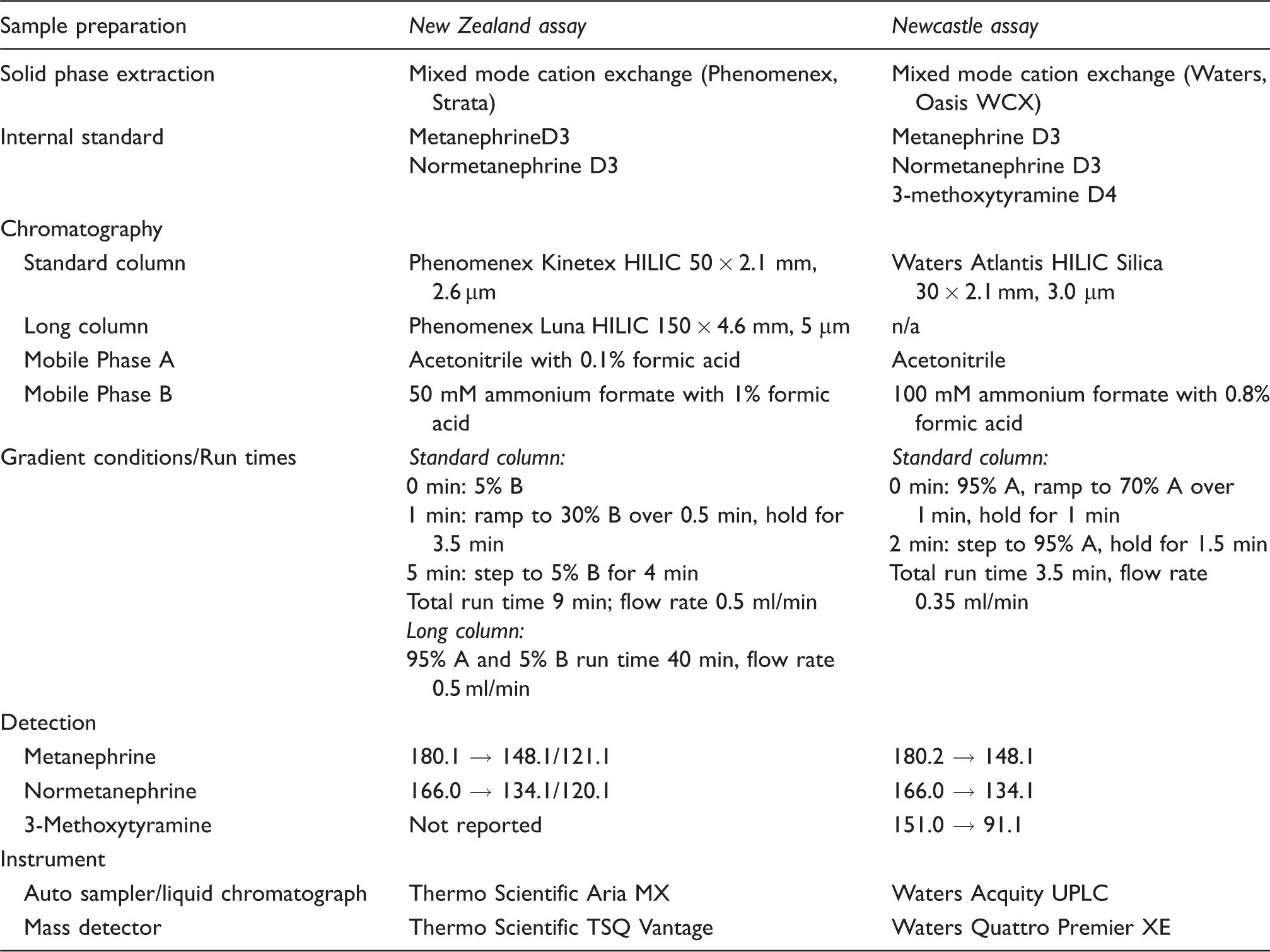
HPLC-MS/MS: high-performance liquid chromatography tandem mass spectrometry.
To confirm analytical interference was caused by midodrine and/or its metabolites midodrine (Douglas Pharmaceuticals, New Zealand) was dissolved in methanol and analysed by the New Zealand assay. Similar analysis was carried out following
Interference studies were carried out by the addition of midodrine and the active metabolite desglymidodrine into residual pooled plasma. Midodrine (Sigma Aldrich) and desglymidodrine (LGC) stock solutions were prepared in methanol. These stock solutions were then spiked into a pooled patient plasma with normal plasma metanephrines to achieve a range of midodrine/desglymidodrine concentrations from 0 to 25 nmol/L. Plasma normetanephrine, metanephrine and 3-methoxytyramine were measured at each spiked midodrine/desglymidodrine concentration (five replicate analyses) using the Newcastle assay. The extent of interference was determined by comparing measurements taken before and after spiking.
Results
Both institutions demonstrated interference by midodrine in their HPLC-MS/MS plasma metanephrines assays. When a methanolic extract of midodrine was injected, a peak was detected at the metanephrine transitions at a later retention time (Figure 1(a)). Following
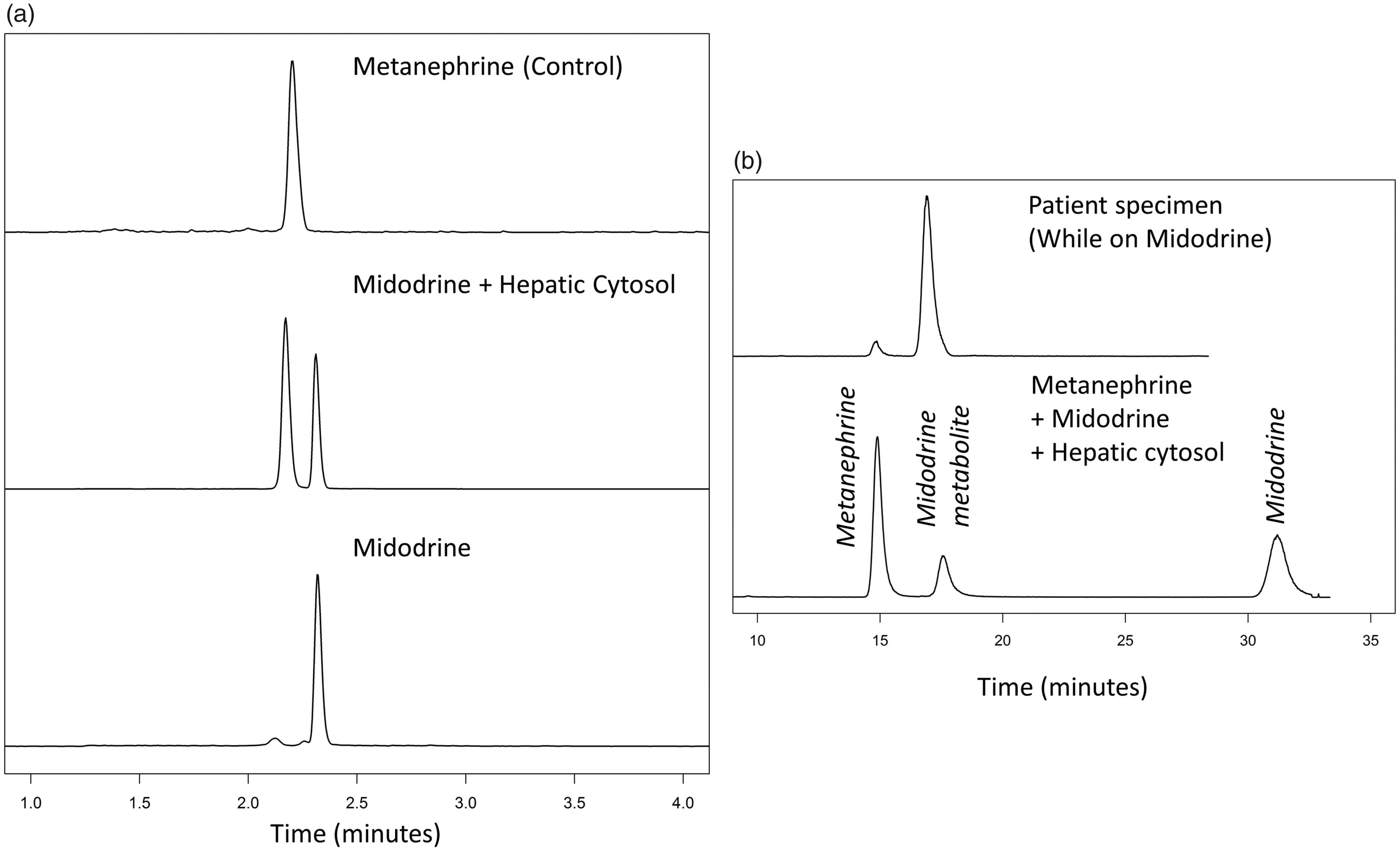
(a) Chromatogram of metanephrine and methanolic extract of midodrine (180.1→148.1) before and after incubation with human hepatic cytosol on the standard column (New Zealand assay). (b) Successful chromatographic separation of metanephrine (180.1→148.1) from midodrine and its metabolite on the longer HILIC column (New Zealand assay). (c) Interference studies of plasma metanephrines by midodrine (top) and desglymidodrine (bottom) as measured by HILIC-based HPLC-MS/MS (Newcastle assay).
Having confirmed analytical interference related to midodrine, an interference study was carried out by spiking known concentrations of midodrine and desglymidodrine to pooled plasma (Figure 1(c)). Unlike the New Zealand assay, in the Newcastle assay both midodrine and desglymidodrine co-eluted with metanephrine. Cross reactivity was estimated at 8% of the spiked midodrine concentration. There was no apparent cross reactivity with normetanephrine or 3-methoxytyramine. Similar experiments carried out by spiking desglymidodrine demonstrated a dose-dependent increase in both metanephrine and methoxytyramine. The cross reactivity was estimated at 5% for metanephrine and 1% for 3-methoxytyramine.
Discussion
In our separate institutions, we have encountered two cases of grossly elevated plasma metanephrines in patients taking midodrine without apparent clinical cause. In both cases, plasma metanephrine as measured by HILIC-based HPLC-MS/MS was significantly raised beyond higher thresholds proposed to distinguish false positives from true cases. 1 On stopping midodrine, plasma metanephrine was within reference range for both patients, suggesting midodrine as a potential cause of their false-positive results. The hypothesis that midodrine or its metabolite desglymidodrine might interfere with the plasma metanephrines was independently confirmed by two laboratories.
Orthostatic hypotension has been estimated to be a clinical feature in 10–60% of patients with PPGL 1 and may be the only clinical feature in some patients.7,8 Midodrine is an alpha-adrenoceptor agonist that can be prescribed for persistent or severe orthostatic hypotension. It is a pro-drug that is rapidly converted to its pharmacologically active form, desglymidodrine, by the removal of glycine. This active metabolite has a much longer half-life (T1/2 = 2–3 h) than midodrine. 9 The results of our spiking experiments indicate that midodrine and desglymidodrine may cause a false elevation in plasma metanephrines in this analytical system. At typical concentrations of midodrine and desglymidodrine observed, 10 we estimate the cross reactivity to plasma metanephrine at 39,000 and 25,000 pmol/L, respectively, on the Newcastle assay. The findings are consistent with the grossly overestimated plasma metanephrine results we encountered in the two patients.
HPLC-MS/MS is a technique falsely presumed by many clinicians to have with infallible analytical specificity. Although mass spectrometry overcomes some of the limitations of immunoassays, it remains a method subjected to possible sporadic analytical interference if quality is not carefully managed. As demonstrated in our study, desglymidodrine fragments to form interfering ions causing analytical false elevation in metanephrine. As both midodrine and its metabolite could be detected at the same transitions for metanephrine, we speculate in-source transformation of midodrine to its metabolite desglymidodrine. When either of these interfering compounds co-elute with metanephrine, as in the case of both patients, a false elevation in metanephrine was observed.
Similar analytical interferences on HILIC-based HPLC-MS/MS for normetanephrine/noradrenaline and metanephrine have previously been reported for 4-hydroxy-3-methoxymetamphetamine a metabolite of 3,4-methylenedioxymethamphetamine (‘Ecstasy’), isoproterenol (a beta-adrenoceptor agonist), isoetharine and methylenedioxyamphetamine.11,12 Laboratories increasingly rely on tandem mass spectrometry for the detection of plasma metanephrines in favour of methods based on electrochemical detection. Increased selectivity using mass spectrometry allows for less stringent chromatographic resolution, thereby enabling the development of higher throughput assays. However, the benefits of measurements of plasma metanephrines by HPLC-MS/MS are not without trade-off (see Eisenhofer et al. 13 for further discussion). HILIC, used by both laboratories to separates metanephrines, was selected to maximize analytical sensitivity.
Despite similarities between methods across the two sites, small chromatographic differences are apparent. As such, the parent drug midodrine, although detected at metanephrine transitions, could be fully separated from the metanephrine peak on the New Zealand assay. In contrast, both desglymidodrine and midodrine caused significant interference on the Newcastle assay. The monitoring of 180.1→121.1 as a qualifying ion appears to be sufficient to detect interference from this drug. Although the pharmacokinetics of the two compounds differ, minor changes in peak shapes/retention times or the presence of two peaks would also raise the suspicion of midodrine-related interference. If interference is suspected, alteration of chromatographic conditions, such as the use of a longer column (Figure 1(b)), could be used to determine the concentration of plasma metanephrine. We would emphasize the importance of considering the impact of column length and type on the ability to separate plasma metanephrines from potentially interfering compounds and of including qualifier ions to improve the detection of interference from co-eluting compounds.
This previously undocumented interference between midodrine and the plasma metanephrine assay is significant for a number of reasons. First, the elevation in plasma metanephrine concentrations was much greater than often seen, and thus cut-offs which have been drawn up to distinguish false-positive from true-positive results would not exclude this particular cause of falsely elevated metanephrines. Second, this interaction is likely to be encountered in clinical practice. Midodrine is recommended for the treatment of severe and intractable postural hypotension, which is a known potential symptom of PPGL, and therefore patients undergoing investigation for PPGL based on such symptoms may be taking the confounding drug as part of their presentation. With plasma metanephrines being used in many institutions as the essential diagnostic test for a PPGL, it is vital for clinicians to be aware of drug interference in their measurement and assessment, not least in order to avoid the need for further investigations with their emotional, financial and procedural costs to healthcare systems. 6
Conclusion
Patients taking midodrine may have falsely elevated plasma metanephrine as a result of analytical interference when measured by HILIC-based HPLC-MS/MS. This potential for interference should be considered in cases where patients taking midodrine require biochemical investigation for potential PPGL.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Written consent was obtained from both patients.
Guarantor
Ashley Grossman.
Contributorship
All authors confirmed they have contributed to the intellectual content of this paper and have met the following three requirements: (a) significant contributions to the conception and design, acquisition of data, or analysis and interpretation of data; (b) drafting or revising the article for intellectual content; and (c) final approval of the published article.
