Abstract
Background
Medication-related interferences with measurements of catecholamines and their metabolites represent important causes of false-positive results during diagnosis of phaeochromocytomas and paragangliomas (PPGLs). Such interferences are less troublesome with measurements by liquid chromatography with tandem mass-spectrometry (LC-MS/MS) than by other methods, but can still present problems for some drugs. Levodopa, the precursor for dopamine used in the treatment of Parkinson’s disease, represents one potentially interfering medication.
Methods
Plasma and urine samples, obtained from 20 Parkinsonian patients receiving levodopa, were analysed for concentrations of catecholamines and their O-methylated metabolites by LC-MS/MS. Results were compared with those from a group of 120 age-matched subjects and 18 patients with PPGLs.
Results
Plasma and urinary free and deconjugated (free + conjugated) methoxytyramine, as well as urinary dopamine, showed 22- to 148-fold higher (
Conclusions
These data establish that although levodopa treatment confounds identification of PPGLs that produce dopamine, the therapy is not a problem for use of LC-MS/MS measurements of plasma and urinary normetanephrine and metanephrine to diagnose more commonly encountered PPGLs that produce noradrenaline or adrenaline.
Keywords
Introduction
Medication-associated interferences with measurements of plasma and urinary catecholamines and metabolites as a cause of false-positive test results during diagnosis of phaeochromocytomas and paragangliomas (PPGLs) are wide-ranging, but in general present in two main forms: (1) pharmaco-physiological effects and (2) direct interference with the analytical method. One of the most common pharmaco-physiological causes of false-positive test results during diagnosis of PPGLs involves the effect of tricyclic antidepressants and related drugs to block sympathoneuronal reuptake of noradrenaline and thereby increase plasma and urinary concentrations of the amine and its metabolites.1–4 Direct analytical interferences with high-performance liquid chromatography electrochemical detection (HPLC-ECD)-based measurements of plasma or urinary catecholamines and their O-methylated metabolites can result from numerous medications, including labetolol,5,6 buspirone, 7 acetaminophen,8,9 mesalamine 10 and its prodrug, sulphasalazine. 11
While pharmaco-physiological causes of false-positive results during diagnosis of PPGLs are method independent, those involving analytical interferences are usually method and analyte specific. Analytical interference is less of a problem for liquid chromatography with tandem mass spectrometry (LC-MS/MS)-based methods than less analytically specific methods, such as HPLC-ECD. 12 Nevertheless, certain medications remain a problem for LC-MS/MS. Levodopa in particular, as a prodrug for dopamine used for treatment of Parkinson’s disease and sometimes for restless legs syndrome, can be metabolized by pathways that lead to numerous products with significant potential for both analytical interference and pharmaco-physiological increases in concentrations of catecholamines and their metabolites.13–19
Davidson
The study by Davison
Although PPGLs in patients with Parkinson’s disease have only rarely been described, 24 numerous reports in Parkinsonian patients of false-positive test results, pseudophaeochromocytoma or hypertensive crises,13–16,18,19,25–27 indicate that suspicion of catecholamine-producing tumours in such patients is not infrequent. This underscores the importance of a reliable diagnostic test for this particular clinical presentation. The aim of the present study was therefore to examine the influences of levodopa therapy on LC-MS/MS measurements of plasma free and urinary free and deconjugated (conjugated + free) normetanephrine, metanephrine and methoxytyramine. Since advanced age has substantial impact on plasma concentrations of normetanephrine,28,29 the study included an age-matched reference group for comparison and a third group of age-matched patients with PPGLs.
Subjects and methods
Subjects
Characteristics of patient populations.
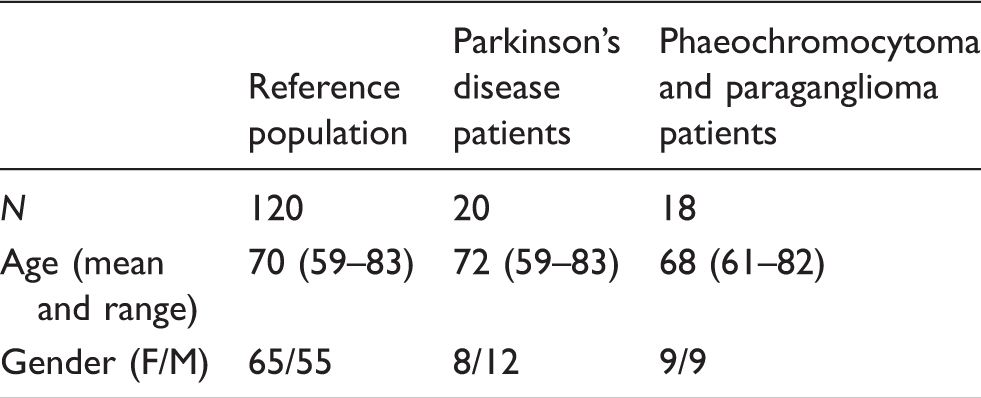
Patients with Parkinson’s disease received levodopa in doses of 100–200 mg three times daily. Levodopa was delivered to all patients in combination with a DOPA decarboxylase inhibitor (carbidopa or benserazide). Twelve of these patients also received dopamine agonist therapy with ropinirole (
The 120 subjects in the age-matched reference group were selected from larger populations of 165 normotensive and hypertensive volunteers and 533 patients tested for PPGLs and in whom tumours were not detected. Selection of subjects into the reference group was based primarily on advanced age with secondary consideration of gender. Similarly, the 18 age-matched patients with PPGLs were selected on the basis of advanced age from a larger group of 74 patients with tumours. PPGLs were confirmed in all patients either on the basis of histopathological examination of resected tumours or in two cases with inoperable metastatic disease, by prior history and findings of metastatic disease by imaging studies.
Patients with Parkinson’s disease were all enrolled and evaluated under a clinical protocol at the University Hospital of Dresden, whereas subjects in the other groups were enrolled under a multicentre protocol (the prospective monoamine-producing tumour study https://pmt-study.pressor.org) involving four institutions: (1) University Hospital of Dresden, Dresden, Germany; (2) Institute of Cardiology, Warsaw, Poland; (3) University Hospital Würzburg, Würzburg, Germany and (4) Medizinische Klinik und Poliklinik IV, Klinikum der Universität München, Munich, Germany. All subjects provided written informed consent under protocols approved by the local Ethics Committees at each Centre.
Collections of blood and urine samples
Samples of heparinized blood (8 mL) were collected according to instructions requiring the draw after at least 20 min of supine rest. Samples were placed on ice or stored refrigerated until centrifuged (4℃) to separate plasma, which was stored at −80℃ until assayed. For the patients with Parkinson’s disease, blood was drawn on an average 3 h (range 0.5–8 h) after the last dose of levodopa. Random urine samples (5 mL) were collected within an hour before or after blood collections and stored at 4℃ until aliquoting and storage at −80℃ within the day of collection. All other urine samples were collected as 24-hour specimens as described previously. 30 Samples of plasma and urine from the three participating centres outside of Dresden were all delivered frozen on dry ice to the Dresden analytical laboratory.
Laboratory analyses
Measurements of plasma concentrations of free normetanephrine, metanephrine and methoxytyramine were by LC-MS/MS as detailed elsewhere. 23 Intra-assay coefficients of variation ranged from 2.9% for high concentrations to 13.5% for low concentrations, while inter-assay coefficients of variation ranged from 6.7% to 11.4%.
Measurements of urinary free normetanephrine, metanephrine and methoxytyramine utilized a similar LC-MS/MS method with modifications that allowed simultaneous measurements of urinary noradrenaline, adrenaline and dopamine.
30
The same assay in a different chromatographic run also enabled measurements of urinary deconjugated normetanephrine, metanephrine and methoxytyramine following an acid hydrolysis procedure as detailed previously.
30
Intra-assay coefficients of variation ranged from 2.7% to 10.3%, while inter-assay coefficients of variation ranged from 6.3% to 13.6%. Measurements of urinary analytes were expressed as ratios of analytes to creatinine (
Additional measurements of plasma concentrations of levodopa, dopamine and 3,4-dihydroxyphenylacetic acid (DOPAC), the deaminated metabolite of dopamine, were carried out in all specimens from patients with Parkinson’s disease and in a subset of specimens from 20 subjects in the reference group. These measurements were by HPLC-ECD with an alumina extraction procedure for specimen purification and enrichment prior to injection onto the HPLC column. 31 Intra-assay and inter-assay coefficients of variation for this assay ranged from 2.5% to 11.0%.
Statistics
Statistical analyses utilized the JMP statistics software package (SAS Institute Inc, Cary, NC). Because plasma and urinary concentrations of the measured analytes show non-normal distributions, results for these analytes are presented as medians and ranges with upper cut-offs for the reference population established using the 97.5 percentiles of distributions. 32 Wilcoxon tests were used for two-group comparisons, whereas the Steel–Dwass method was used for non-parametric comparisons among all pairs for three groups. Significance of relationships was determined using Spearman’s test.
Results
Plasma levodopa, dopamine and DOPAC
Plasma concentrations (medians and ranges) of levodopa, dopamine and DOPAC in Parkinson’s disease patients compared to reference subjects.
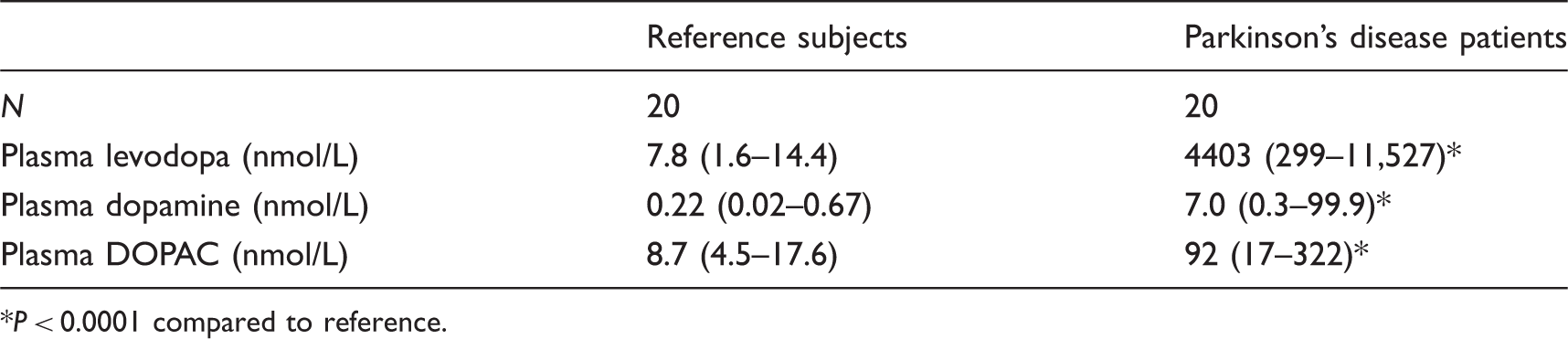
Among patients with Parkinson’s disease, plasma concentrations of levodopa, dopamine and DOPAC varied widely, but showed no relationship to doses of levodopa with and without consideration of time from the last dose before blood samples were drawn. While plasma concentrations of levodopa were positively correlated with those of DOPAC (
Plasma free normetanephrine, metanephrine and methoxytyramine
Plasma concentrations of free normetanephrine in patients with Parkinson’s disease treated with levodopa did not differ from concentrations in the age-matched reference group, but were 6-fold higher ( Dot and box-plots of plasma concentrations of free normetanephrine (a), metanephrine (b) and methoxytyramine (c) in the reference group (REF), in patients with Parkinson’s disease receiving levodopa (PARK) and in patients with PPGLs (PPGL). The dashed horizontal lines with values immediately above represent the 97.5 percentiles for distributions in the reference population. 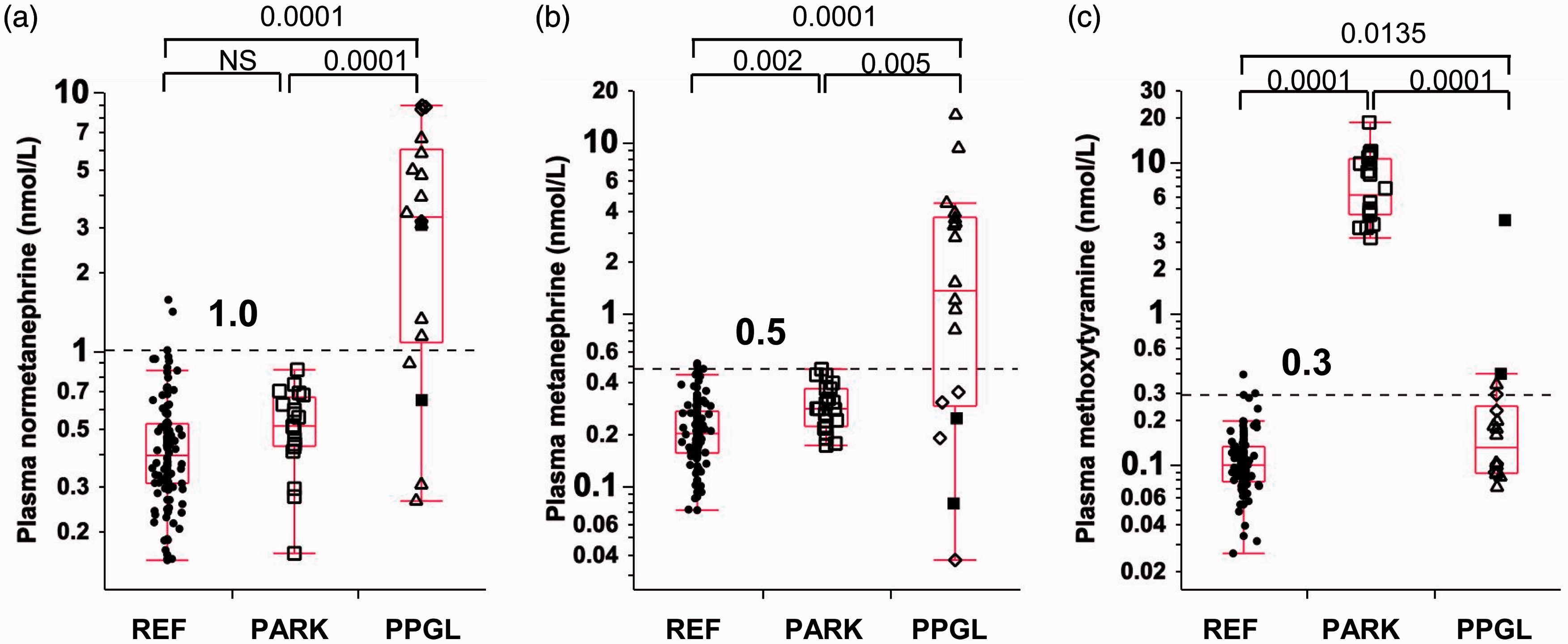
None of the Parkinsonian patients treated with levodopa had plasma concentrations of normetanephrine above the upper cut-offs of the reference group (Figure 1(a)). Furthermore, only one patient had plasma concentrations of metanephrine above the upper cut-offs (Figure 1(b)). In contrast, plasma concentrations of methoxytyramine for all patients treated with levodopa were increased by more than 10-fold above the upper cut-offs of the reference group (Figure 1(c)).
Urinary catecholamines and O-methylated metabolites
Similar to the findings for plasma normetanephrine (Figure 1(a)), urinary concentrations of noradrenaline and free and deconjugated normetanephrine, expressed as ratios to urinary creatinine concentrations, showed no differences between patients with Parkinson’s disease treated with levodopa and the reference population (Figure 2(a), (d) and (g)). None of the levodopa-treated Parkinsonian patients showed urinary outputs of noradrenaline or normetanephrine above the 97.5 percentiles of the reference group and only one patient showed outputs of urinary deconjugated normetanephrine above the cut-offs.
Dot and box-plots of creatinine-normalised urinary outputs of noradrenaline (a), adrenaline (b), dopamine (c), free normetanephrine (d), free metanephrine (e), free methoxytyramine (f), deconjugated normetanephrine (g), deconjugated metanephrine (h) and deconjugated methoxytyramine (i) in the reference group (REF), in patients with Parkinson’s disease receiving levodopa (PARK) and in patients with PPGLs (PPGL). The dashed horizontal lines with values immediately above represent the 97.5 percentiles for distributions in the reference population. 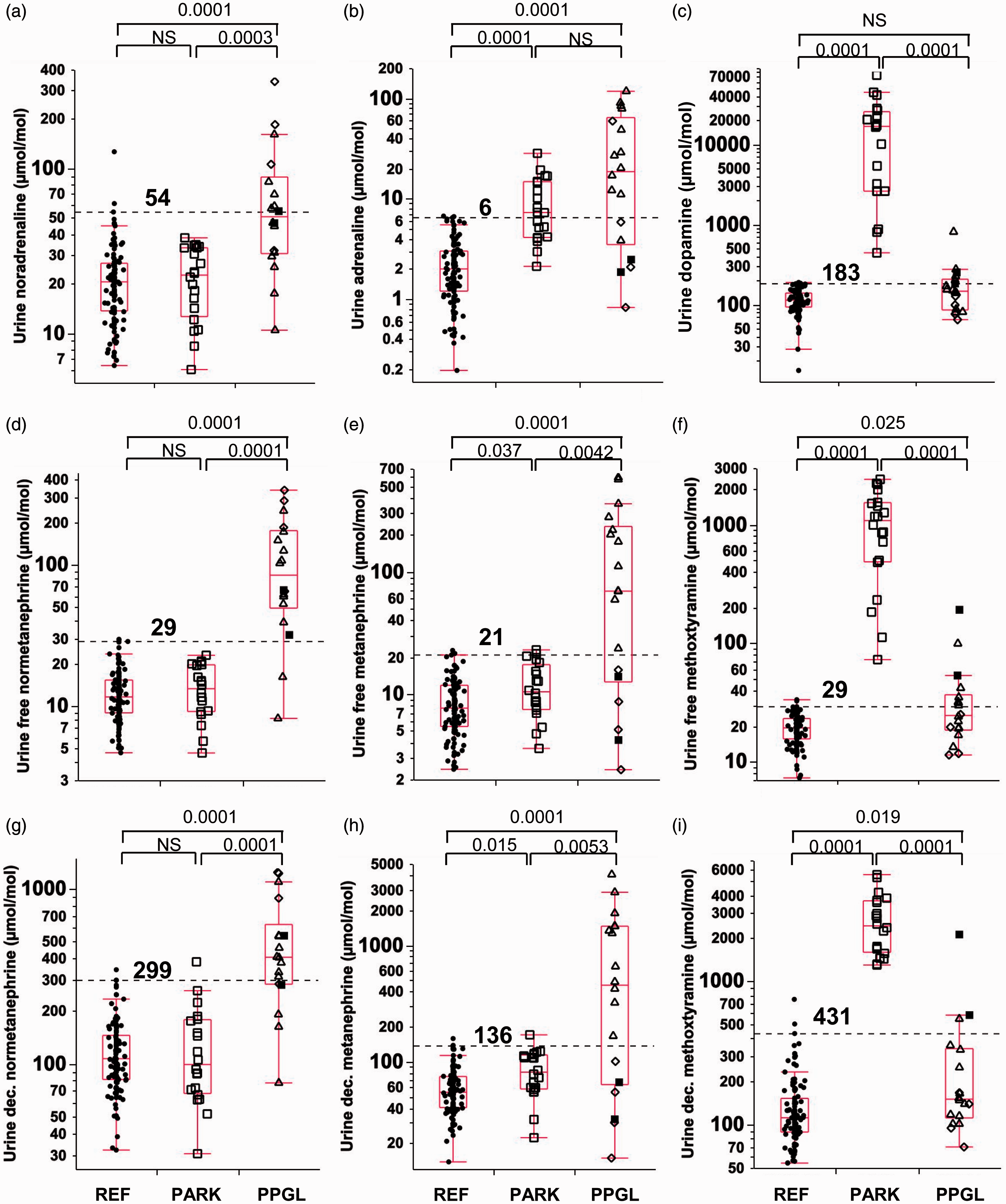
In contrast to noradrenaline and its O-methylated metabolites, but similar to the findings for plasma metanephrine (Figure 1(b)), creatinine-normalized urinary outputs of adrenaline and free and deconjugated metanephrine were consistently higher (
Also similar to findings for plasma methoxytyramine (Figure 1(c)), urinary dopamine and free and deconjugated methoxytyramine were all substantially higher (
Patients with PPGLs
All except four patients with PPGLs had plasma concentrations of normetanephrine above the 97.5 percentiles of the reference group, but three of these four had elevated plasma concentrations of metanephrine while one had solitary elevations of plasma methoxytyramine (Figure 1(a), (b) and (c)). Similarly, all except a few patients with PPGLs showed increases in urinary free and deconjugated normetanephrine above the upper cut-offs of the reference group (Figure 2(d) and (e)), but among those, all had increases of either urinary metanephrine or methoxytyramine (Figure 2(e), (f), (h) and (i)).
In contrast to normetanephrine, urinary noradrenaline in patients with PPGLs showed concentrations under the upper cut-offs of the reference group in nearly one-half of all patients (Figure 2(a)). Thus, urinary noradrenaline in patients with PPGLs was only increased (
Discussion
This study establishes that while levodopa therapy confounds identification of PPGLs that produce dopamine, the drug is not a problem for the diagnosis of more commonly encountered noradrenaline or adrenaline-producing PPGLs. Our findings that plasma concentrations and urinary outputs of normetanephrine and metanephrine are largely unaffected by levodopa therapy contrasts with findings by Davidson
The above conclusion, however, was based on measurements by HPLC-ECD, whereas the present findings of minimal influence of levodopa on plasma free normetanephrine and metanephrine as well as the urinary free and deconjugated metabolites was based on measurements by LC-MS/MS. Mass spectrometric methods, in which detection involves multiple reaction monitoring of precursor and product ions, offer improved specificity and relative freedom from analytical interferences compared to other methods, such as HPLC-ECD.
Apart from metabolism to dopamine, and potentially to downstream catecholamines and their metabolites, levodopa may also be metabolized by other pathways to numerous products, such as dihydroxyphenylpyruvic acid, 3-O-methyldopa and vanillactic acid.33,34 Such products have potential for analytical interference, a separate source of false-positive results from the well-established pharmaco-physiological effects of levodopa to substantially increase plasma and urinary excretion of dopamine and dopamine metabolites, such as DOPAC, homovanillic acid and methoxytyramine.33,35 The present findings of substantially increased plasma and urinary excretion of methoxytyramine after levodopa is in agreement with the latter observations, establishing that measurements of this metabolite in both plasma and urine cannot be used to identify dopamine-producing PPGLs in patients taking levodopa.
Interestingly, while urinary noradrenaline and plasma and urinary normetanephrine were unaffected by levodopa therapy, adrenaline in urine and its O-methylated metabolites in both plasma and urine were consistently higher in Parkinsonian patients treated with levodopa than in age-matched controls. Nevertheless, while the difference for urinary adrenaline was clear, the differences for metanephrine measured in both plasma and urine were relatively minor. More importantly, as shown by the comparisons with the reference group and patients with PPGLs, the small magnitude of the increases in plasma and urinary metanephrine in the patients with Parkinson’s disease is unlikely to significantly impact the utility of these measurements for diagnosis of PPGLs.
The reason for the 3.6-fold higher urinary outputs of adrenaline and smaller increases of metanephrine in the Parkinsonian compared to the reference group is unclear and not entirely consistent with earlier studies,17,36–38 some of which have indicated either no differences or even lowered concentrations of urinary adrenaline in Parkinsonian patients.37,38 Nevertheless, there have been two reports of increased urinary adrenaline excretion in Parkinsonian patients, but in both cases this was only observed while patients were not receiving levodopa.17,36 Possibly, therefore, the presently observed increases in urinary excretion of adrenaline and metanephrine in the Parkinsonian group are unrelated to any direct influence of levodopa.
While the minimal influence of levodopa on plasma and urinary normetanephrine and metanephrine is not in agreement with the study of Davidson
A limitation of this study was that Parkinsonian patients, besides receiving levodopa combined with a dopa-decarboxylase inhibitor, were all receiving highly variable combinations of other medications. These medications included MAO-B or COMT inhibitors and antidepressants, all with significant potential for influences on the metabolism or disposition of catecholamines. Dopamine agonist therapy can also influence sympathoadrenal activity in divergent directions, with increases in adrenaline release. 39 While the highly variable combinations of medications confounds interpretation of some of the findings, such as the increased urinary excretion of adrenaline and lesser increases in metanephrine, this is not a problem for the primary conclusions of the study concerning the minimal impact of levodopa therapy on plasma and urinary normetanephrine and metanephrine. In fact, the highly variable combinations of medications reflects the clinical scenario that can be expected with testing in Parkinsonian patients, providing additional confidence that other medications commonly used in these patients are not overtly troublesome for diagnosis of PPGLs.
In summary, with LC-MS/MS-based measurements of plasma free or urinary free or deconjugated normetanephrine and metanephrine, excluding or confirming PPGLs in patients on levodopa therapy should no longer be considered a significant diagnostic challenge.
Footnotes
Acknowledgements
Thanks are extended to Carmen Berndt and Nan Qin for technical assistance and Drs Roland Därr, Dirk Weismann and Urs Lichtenauer for assistance in patient recruitment.
Declaration of conflicting interests
The authors declare no conflict of interest.
Funding
This work was supported by the Deutsche Forschungsgesellschaft (INST515/6-1; EI855/1-1) and the Institute of Cardiology, Warsaw, Poland (Grant no: 2.21/VII/12).
Ethical approval
Patients were enrolled into the study under approvals from Institutional Review Boards of the Technical University of Dresden, Dresden, Germany (EK189062010 & EK 37010211), the Institute of Cardiology, Warsaw, Poland (1233-08112010), the Medical University Hospital of Würzburg, Würzburg, Germany (104/11) and the University Hospital of Munich, Munich, Germany (173-11).
Guarantor
GE.
Contributorship
GE, GS and HR conceived the study and together with SG, PL, FB, MF, AP and AJ were involved in development of protocols and gaining ethical approvals. MP and DP contributed to analyses, while SB, SG, AP, MS and FB contributed to patient recruitment and compilation of data, which was managed by AS, SG, GE, MP and DP. GE wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version.
