Abstract
Background
The Endocrine Society Clinical Practice Guideline on Phaeochomocytoma and Paraganglioma recommends phlebotomy for plasma-free metanephrines with patients fasted and supine using appropriately defined reference intervals. Studies have shown higher diagnostic sensitivities using these criteria. Further, with seated-sampling protocols, for result interpretation, reference intervals that do not compromise diagnostic sensitivity should be employed.
Objective
To determine the impact on diagnostic performance and financial cost of using supine reference intervals for result interpretation with our current plasma-free metanephrines fasted/seated-sampling protocol.
Methods
We conducted a retrospective cohort study of patients who underwent screening for PPGL using plasma-free metanephrines from 2009 to 2014 at Galway University Hospitals. Plasma-free metanephrines were measured using liquid chromatography-tandem mass spectrometry. Supine thresholds for plasma normetanephrine and metanephrine set at 610 pmol/L and 310 pmol/L, respectively, were used.
Results
A total of 183 patients were evaluated. Mean age of participants was 53.4 (±16.3) years. Five of 183 (2.7%) patients had histologically confirmed PPGL (males,
Conclusion
Our cost analysis, together with the evidence that fasted/supine-sampling for plasma-free metanephrines, offers more reliable exclusion of PPGL mandates changing our current practice. This study highlights the important advantages of standardized diagnostic protocols for plasma-free metanephrines to ensure the highest diagnostic accuracy for investigation of PPGL.
Introduction
Biochemical evidence of excessive catecholamine production is critical to diagnose phaeochromocytoma and paraganglioma (PPGL). Current evidence suggests that measurements of free plasma metanephrines (PMets) or urinary fractionated metanephrines (normetanephrine (NMN) and metanephrine (MN) separately) are the most sensitive tests for diagnosis and are the most suitable for reliable exclusion of PPGL. 1 The Endocrine Society Clinical Practice Guideline on PPGL recommends phlebotomy for fractionated PMets with patients in the supine position using appropriately defined reference intervals. 1 The emphasis on the supine-sampling protocol is based on observations regarding the strong influence of sympathetic activation and upright posture to stimulate the release of norepinephrine and its metabolism to NMN, and the likely absence of a response to these stimuli in patients with PPGLs. 1 Furthermore, reference intervals for PMets established using a seated-sampling protocol are higher than those defined using supine-sampling protocols. 1
Lenders et al. 2 estimated that the use of upper reference limits (URLs) for PMets determined from a seated-sampling protocol would result in a drop in diagnostic sensitivity and a three-fold increase in false-negative results. Nevertheless, a recent study by Chortis et al. 3 found that there was no difference in the rate of false-negative results between supine and seated sampling, provided the URLs employed for PMets was derived from supine and fasting patients. Not surprisingly, this strategy impacted negatively on diagnostic specificity resulting in increased numbers of false-positive results. These authors demonstrated that implementing this screening strategy would confer an added health-care cost as a consequence of the reduced diagnostic specificity.
Our objective was to determine the impact on diagnostic performance and financial cost of using supine reference intervals for result interpretation with our current fasted/seated-sampling protocol for PMets.
Methods
A retrospective cohort study of all patients who underwent screening for PPGL using PMets from 2009 to 2014 at Galway University Hospitals was conducted. Clinical details were obtained through chart review and interrogation of the radiology and laboratory information systems. Venous blood samples (10 mL) for PMets are collected with patients fasting and canulated for 30 min of continuous seated-rest, into ethylenediaminetetraacetic acid specimen tubes using seated-sampling reference intervals (URLs for NMN = 1180 pmol/L and MN = 510 pmol/L). 4 Specimen tubes are placed without delay on ice and transported within 20 min of blood draw to the laboratory for immediate processing (specimen centrifugation, separation and freezing of plasma at −20℃) prior to analysis. Fractionated PMets (NMN and MN) were measured using liquid chromatography-tandem mass spectrometry 4 at the Freeman Hospital, Newcastle, United Kingdom. Supine URLs for plasma NMN and MN set at 610 pmol/L and 310 pmol/L, respectively1,5 were retrospectively applied to the PMets results.
Cost analysis
The financial costs of both preanalytical protocols (supine/seated) for PMets analysis were computed by addition of each component cost: consumables (specimen tubes: €0.09 (×2), cannula: €0.18, Syringe: €0.31 and adhesive bandage: €0.10). We estimated that one hour per patient was required to perform and complete each protocol and calculated the non-consumable costs accordingly: clerical staff: €15, hospital chair: €14, hospital bed/trolley: €14, medical and nursing supervision: €47, phlebotomy: €22, sample transport to UK: €20.18 and PMets analysis; €28.32.
Statistical analysis
Statistical analysis was performed using R® V3.2.0 (R Foundation for Statistical Computing accessible at www.r-project.org). Summary statistics included means and standard deviations for continuous variables and frequencies and percentages for categorical variables. The diagnostic sensitivity and specificity was then calculated for plasma NMN and MN using both the current laboratory and the supine decision thresholds for PMets.
Results
Phaeochromocytoma patient characteristics and plasma metanephrine biochemistry.
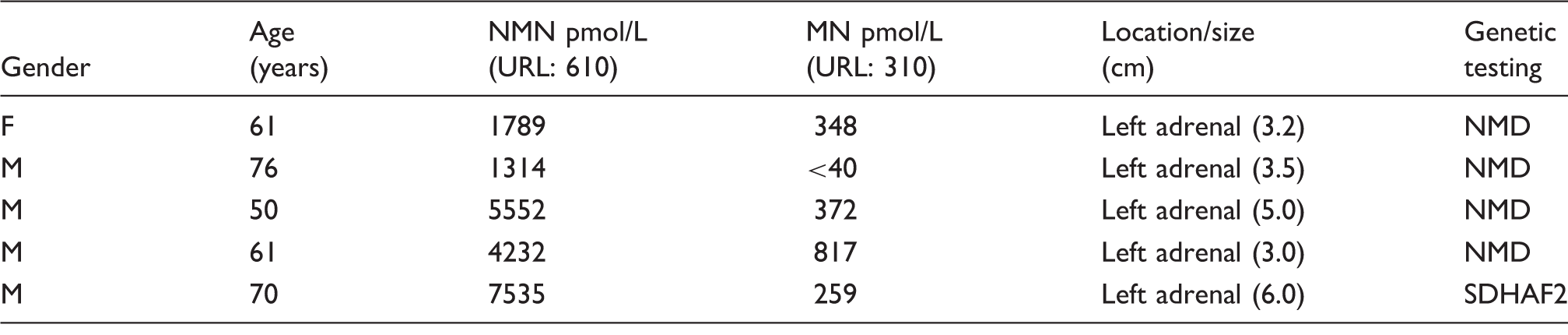
URL: upper reference limit; NMN: normetanephrine; MN: metanephrine; NMD: no mutation detected; F: female; M: male
Using the current laboratory preanalytical protocol,
4
diagnostic sensitivity and specificity was 100% and 98.9%, respectively, with false-positive screens in two patients (1.1%). PPGL was excluded in these two patients following repeat biochemical screening under fasting/supine-sampling conditions using appropriate reference intervals. Retrospective application of supine URLs to PMets results gave a diagnostic sensitivity and specificity of 100% and 74.6%, respectively, with false-positive screens in a total of 45 patients (24.6%). PPGL was excluded in these patients based on repeat negative biochemical screening (
Cost analysis determined that the total cost of the current fasted/seated-sampling PMets protocol is €147.27 and equivalent to the cost of the fasted/supine-sampling protocol. The estimated added health-care cost consequent to the 22-fold increase in false positives observed when supine PMets URLs were applied to our cohort was €6332.61.
Discussion
Recent evidence suggesting that the use of URLs for PMets established from seated sampling can result in a 15% drop in diagnostic sensitivity
5
was the impetus for this study. In our study cohort, diagnostic sensitivity (100%) was equivalent irrespective of the sampling conditions (seated or supine) of the established PMets reference intervals. However, the marked reduction in diagnostic specificity observed would have resulted in a ∼22-fold increase in false-positive screens affecting 43 patients (23.5%) of the study cohort. Such a false-positive rate is unacceptable for a disease with a prevalence of 2–8/million/year. The non-inferior diagnostic sensitivity associated with PMets sampling in the seated position as compared with supine sampling is not in keeping with recent literature.
5
This may be due to the relatively small numbers of patients with PPGL (
In the study by Chortis et al., 3 seated screening was economically judicious. 3 Cost analysis carried out at our institution determined cost equivalence for both pre-analytical sampling strategies.
We acknowledge the limitations of the current study are its retrospective design with only seated samples for PMets being taken. A prospective study is required to directly compare the diagnostic performance of PMets sampled from fasted patients seated with those supine, and using the respective appropriately defined reference intervals.
Recently, useful insight into the patients’ perspective on the issue of supine versus seated sampling for PMets has been eloquently expressed by Sussman et al. 8 Firstly, the authors inform that the highest priority for patients is diagnostic test accuracy. Secondly, that the economic arguments put forward to eliminate supine sampling as first-line investigation for patients with suspected PPGL, 3 underestimate the real costs both financial and emotional. The costs of a false-positive PMets result are those incurred for extra hospital visits, or emotional in terms of anxiety and stress to patients/family, with no account taken of patient preference. Finally, Sussman et al. 8 address the medical and scientific communities and advocate for the universal adoption of standardized testing protocols to ensure the highest diagnostic accuracy.
Conclusion
Our cost analysis, together with the evidence that fasted/supine sampling for PMets, offers more reliable exclusion of PPGL1,5,7 make it medically and economically irresponsible in our institution not to implement the fasted/supine-sampling protocol. Moreover, this study highlights the important advantages of standardized evidence based diagnostic protocols for PMets that ensure the highest diagnostic accuracy for investigation of PPGL.
Footnotes
Acknowledgements
We wish to express our gratitude to the study participants, scientific, medical, and nursing staff at the Centre for Diabetes, Endocrinology and Metabolism (CDEM), and the Department of Clinical Biochemistry at Galway University Hospitals and Saolta Hospital Group, and to Dr Chris Boot and the scientific staff of the Department of Blood Sciences, Freeman Hospital, United Kingdom.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The Research Ethics Committee at Galway University Hospitals granted ethical approval for this study. Ref. no: C.A. 854.
Guarantor
POS.
Contributorship
POS: concept, design and acquisition of data. DW: statistical analysis. RC: made significant contributions to the acquisition of data and wrote the first draft of this manuscript. All authors contributed to the intellectual content and interpretation of data, drafting and revising the article.
