Abstract
Objective
To determine the necessity for acid-washed containers for 24-h urine copper analysis.
Methods
Copper solutions, with concentrations relevant to clinical decision limits, were prepared by spiking both assay diluent and unknown urine samples with the copper calibrator. Samples were split between plain and acid-washed 24-h urine containers, and copper analysis was performed using inductively coupled mass spectrometry (ICP-MS).
Results
Measurement of copper in both spiked diluent and spiked urine samples showed minimal concentration bias between acid-washed and plain 24-h urine containers.
Conclusions
Acid-washed containers are not required for the measurement of copper in 24-h urine samples.
Introduction
In the diagnosis of Wilson’s Disease, 24-h urine copper measurement is one of the primary biochemical investigations. 1 At Manchester University Hospitals NHS Foundation Trust (MFT), we currently specify that 24-h urine copper samples should be collected into plastic bottles that have been washed with 30% nitric acid before rinsing with 18 MOhm Ultrapure water to preclude copper contamination from the container. A recent review at MFT showed that, over a six-month period, 40 requests were received for urine copper. Eleven of these samples (27.5%) were rejected, having not been collected in the correct 24-h urine container. Of the rejected samples we received, four were collected into plain (not acid-washed) containers, of which two were random urine samples (24-h urine sample required). Five of the rejected samples were collected into 24-h urine bottles containing 2 molar hydrochloric acid preservative (2 M HCl), and the specimen container type received was not documented for two samples. Timed urines are difficult to collect especially in children, and rejection of specimens causes diagnostic delay as well as difficulty for patients in providing a new collection. Patients are also at risk of harm should they come into contact with any residual acid not properly washed from the container. The necessity of acid-washing plastic bottles is unproven, and there is some evidence that it may be unnecessary.1,2
Aim
We set out to seek evidence of significant environmental contamination in urine copper measurements, from plastic 24-h urine bottles, by testing unwashed urine containers.
Method
Four solutions were prepared by spiking the diluent used in our inductively-coupled plasma mass spectrometry (ICP-MS) method (0.14% nitric acid) with ICP-MS copper calibrator (1000 mg/L Certipur, Merk [VWR]) to give a range of copper concentrations. Copper concentrations were selected that were relevant to clinical decision limits, each of these 50 mL solutions was divided between an acid-washed and a plain 24-h urine bottle (5 L). Each sample was mixed well in the 24-h urine container to ensure contact with all surfaces. Samples were stored at +2–8°C for three days, after which they were aliquoted into 20 mL tubes, for ease of storage, and analysed for urine copper by ICP-MS as per our standard operating procedure.
To account for any sample matrix effects, four additional samples were prepared by spiking aliquots of a 24 h urine sample of unknown copper concentration with the same range of concentrations of the copper ICP calibrator described above. Each of these four samples was split between an acid-washed and a plain 24-h urine bottle (2.5 L) and stored at +2–8°C for three days prior to aliquoting into 20 mL secondary tubes. Six further urine solutions were then prepared in the same way, using a different batch of containers, to cover the reportable range of the assay and to provide sufficient sample numbers for a statistically valid Student’s t-test. Measurement of urine copper was performed in duplicate using ICP-MS on an Agilent 7800 series instrument (Agilent Technologies UK Limited Registered Office) with Mass Hunter software for data analysis. Verification of the urine copper analysis method demonstrated <1.5% intermediate imprecision for IQC samples (1.85 micromol/L and 2.50 μmol/L) and a lower reporting limit of 0.15 μmol/L.
Copper ICP calibrator spike concentrations (clinical cut-off concentrations are approximate as patient urine volumes will vary):
Zero –to detect any copper contamination from the sample container or present in the reagents 0.24 μmol/L – Concentration close to the lower reporting limit of 0.15 μmol/L 6.30 μmol/L – Clinical cut-off (>6 μmol/24 h supports a diagnosis of Wilson’s disease) 23.62 μmol/L – Clinical cut-off (>25 μmol/24 h postpenicillamine supports a diagnosis of Wilson’s disease).
Results
Analysis of blank diluent (zero spike) from both plain and acid-washed containers yielded results above the limit of detection of the assay (0.0015 μmol/L) but below the limit of quantification (0.15 μmol/L), indicating minimal contamination from either container or from the reagents. The 0.24 μmol/L sample gave a combined %CV of 7.96 for all four measurements including both container types. This is within the expected variation for the assay at this concentration. Combined %CVs were <2% for samples with concentrations relevant to clinical decision limits. Bias between the two different container types is also minimal at these concentrations (Table 1).
Summary of results for copper analysis of spiked diluent samples.
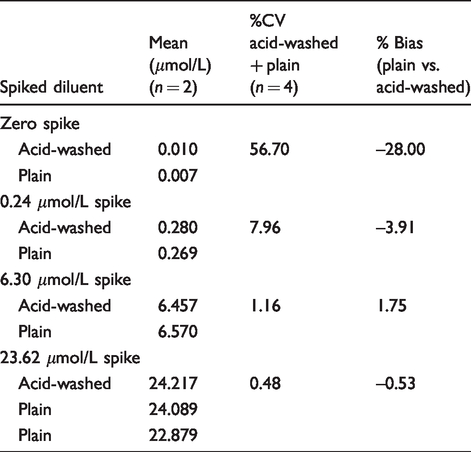
Analysis of 10 spiked urine samples (concentration range 0.23–72.3 μmol/L) from both plain and acid-washed containers showed no statistical difference between container type (P-value = 0.8108, mean difference 0.0078 μmol/L). Combined %CVs were <2% for all spiked urine samples and, as with spiked diluent samples, bias between the two different container types was found to be minimal. Calculated recoveries were acceptable (90–105%) for all clinically relevant copper concentrations and were found to be comparable between different sample types (diluent/urine) and containers (acid-washed/plain).
Conclusion
The copper concentration bias between plain and acid-washed plastic urine containers was found to be minimal for both spiked diluent and spiked urine samples, across a range of clinically relevant concentrations. The difference in measured copper concentration from spiked urine samples stored in plain and acid-washed containers was also statistically insignificant across the analytical concentration range of the assay. These data, combined with excellent recoveries, demonstrate that plain 24-h plastic urine containers are suitable for urine copper analysis by ICP-MS and that prewashing with nitric acid is unnecessary. We recommend the use of plain 24-h urine containers for urinary copper estimation, minimizing repeat collections and eliminating any potential risks associated with residual nitric acid.
Footnotes
Acknowledgements
The authors would like to acknowledge Andrew Wood for analysing additional samples prepared by AFR.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect research, authorship, and/or publication of this article
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
AFR.
Contributorship
AFR, LT and CMC researched the literature and conceived the study. AFR and PL prepared samples and performed analysis. AFR wrote the first draft of the article. All authors reviewed, edited and approved the final version of the article.
