Abstract
Background
Non-alcoholic fatty liver disease is classified into simple steatosis (SS) and non-alcoholic steatohepatitis (NASH) according to histological findings from liver biopsies. Phosphatidylcholine (PC), the main component of phospholipids in serum lipoproteins, is easily oxidized to phosphatidylcholine hydroperoxide (PC-OOH). Although a lipid composition in the low-density lipoproteins (LDL) from patients with NASH could be abnormal, it remains unclear. Here, to better understand the characteristics of lipids in the LDL from NASH and SS, we compared the composition of PC and PC-OOH species in LDL particles (LDL-PC, LDL-PCOOH) from these patients, then clarified the association between these lipids and NASH severity.
Methods
The serum samples from patients with NASH (female, n = 9) and SS (female, n = 4; male, n = 2) were used for isolation of LDL. Total lipids were extracted from isolated LDL, and the species of PC and PC-OOH were measured using liquid chromatography-mass spectrometry/mass spectrometry.
Results
The sum of LDL-PC and the sum of LDL-PCOOH were significantly higher in NASH than in SS. Several LDL-PC (PC 32:0, 32:1, 32:2, 34:3, 36:2, sum of PC with saturated fatty acyl chains and sum of LDL-PC with polyunsaturated fatty acyl chains) and several LDL-PCOOH (34:2, 36:2, 36:3 and total) were increased significantly with increasing fibrosis score. In particular, a series of LDL-PCOOH were more reflective of the severity of fibrosis score.
Conclusions
LDL-PC and LDL-PCOOH species were strongly correlated with the fibrosis score in NASH, which suggests that abnormal LDL is involved in the development of liver fibrosis.
Introduction
Non-alcoholic fatty liver disease (NAFLD) refers to hepatic steatosis not caused by alcohol, viruses, or drugs. NAFLD is classified into simple steatosis (SS) and non-alcoholic steatohepatitis (NASH) according to histological findings from liver biopsies. Patients with NASH are more likely to progress to cirrhosis and hepatocellular carcinoma than patients with SS. A retrospective study of patients with NAFLD reported that liver fibrosis was independently associated with increased liver-related mortality. 1 However, noninvasive risk assessment markers for NAFLD are still lacking.
In this study, we focus on phosphatidylcholine (PC) in the serum low-density lipoproteins (LDL) of patients with NASH. Because PC is located on the surface of lipoproteins, PC is susceptible to oxidation, which results in phosphatidylcholine hydroperoxide (PC-OOH). Oxidized LDL likely plays a role in the development of NASH. 2 Taken together, these reports suggest an abnormal lipid composition in the LDL of patients with NASH. However, the profile of PC and PC-OOH in LDL particles from patients with NASH remains unclear.
Therefore, to better understand the characteristics of PC and its hydroperoxides in the LDL from patients with SS and NASH, we perform a comprehensive analysis of PC and PC-OOH in LDL samples with liquid chromatography-mass spectrometry/mass spectrometry (LC-MS/MS). We also clarify the correlation between these lipid indicators and the histological degree of severity in NASH.
Materials and methods
Serum samples from six and nine patients with SS and NASH, respectively, were provided by Okayama University Hospital (Okayama, Japan). The serum samples were randomly selected from samples used in a previous study. 3 It should be noted that the research objectives and methods differ between the previous and current studies. As previously reported, NASH was diagnosed according to the NAFLD activity score, (≥5 points) and the degree of liver fibrosis was assessed using Brunt Classification (Supplemental Table 1). This study was conducted with ethical approval from Okayama University (approval number: 1604-011), Okayama City Hospital (approval number: 30-2), and Hokkaido University (approval number: 18-69-6). Informed consent was obtained from all subjects involved in the study.
For separation of LDL, the serum was separated by combination of ultracentrifugation and gel-filtration chromatography using two Superose 6 columns with minor modification of a previous report. 4 Protein concentrations in the LDL fraction were measured using the Lowry method. 5 According to a previous report, lipids were extracted from the LDL fraction as samples and measured by Orbitrap mass spectrometer (LTQ Orbitrap XL, Thermo Fisher Scientific, Waltham, MA, USA) (Supplemental Table 2). 4 PC and PC-OOH were targeted as representative markers of phospholipids and oxidative stress, respectively (Supplemental Table 3).
All analyses were performed using GraphPad Prism 7 software (GraphPad Software, San Diego, CA, USA). The Mann-Whitney U-test and receiver operating characteristic curve-area under the curve (ROC-AUC) was used for comparison of all PC and PC-OOH species in LDL particles (LDL-PC, LDL-PCOOH). Spearman’s rank correlation coefficient was calculated to clarify the relationship between LDL-PC and LDL-PCOOH species that showed significantly high values in NASH and historical findings. P < .05 was considered statistically significant.
Results
Six PC species were significantly higher in NASH than in SS (Supplemental Figure 1(a) and (b)). The total values of PC species in LDL (sum of LDL-PC) were significantly higher in NASH than in SS (P < .05) (Supplemental Figure 1(c)). The sum of LDL-PC with saturated fatty acyl chains (sum of LDL-PC with SFA), the sum of LDL-PC with monounsaturated fatty acyl chains (sum of LDL-PC with MUFA), and the sum of LDL-PC with polyunsaturated fatty acyl chains (sum of LDL-PC with PUFA) were all significantly higher in NASH than in SS (Supplemental Figure 2). All PC-OOH species detected in LDL were significantly higher in NASH than in SS (Supplemental Figure 3(a) and (b)). The total values of PC-OOH species (sum of LDL-PCOOH) were significantly higher in NASH than in SS (P < .01) (Supplemental Figure 3(c)). ROC-AUC (NASH vs SS) of some LDL-PC and LDL-PCOOH showed preferable values, 0.81 or over (Supplemental Table 4).
Association Between Histological Findings and Phosphatidylcholine (PC) and Phosphatidylcholine Hydroperoxide (PC-OOH) Species in LDL Particles.
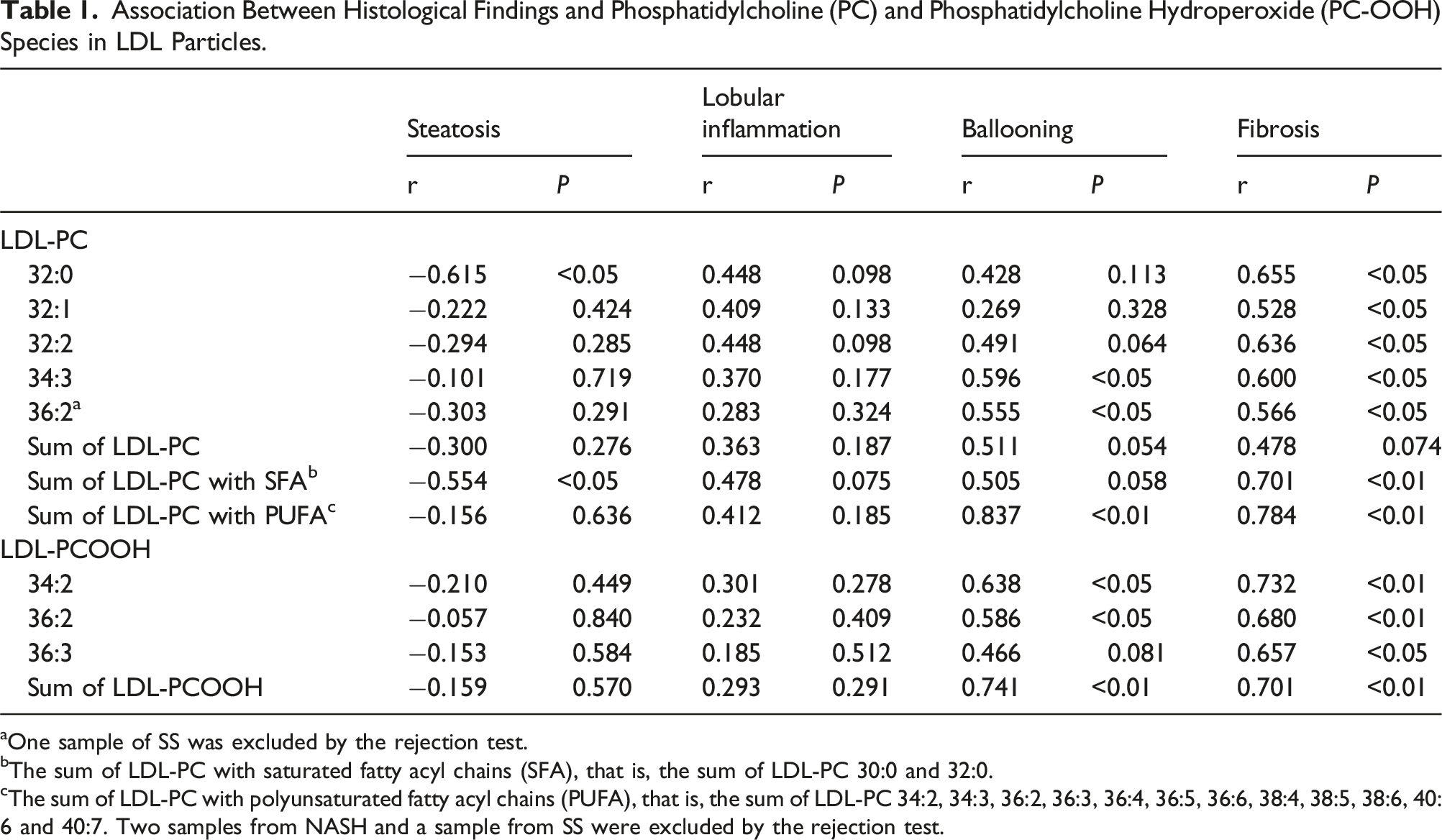
aOne sample of SS was excluded by the rejection test.
bThe sum of LDL-PC with saturated fatty acyl chains (SFA), that is, the sum of LDL-PC 30:0 and 32:0.
cThe sum of LDL-PC with polyunsaturated fatty acyl chains (PUFA), that is, the sum of LDL-PC 34:2, 34:3, 36:2, 36:3, 36:4, 36:5, 36:6, 38:4, 38:5, 38:6, 40:6 and 40:7. Two samples from NASH and a sample from SS were excluded by the rejection test.
Discussion
LC-MS/MS revealed an abnormal lipid composition of LDL in patients with NASH, characterized by an enrichment of PC and PC-OOH compared to patients with SS. Regarding with PC, a possible reason could be the suppression of PC species degradation from PC to lysophosphatidylcholine in the livers of patients NASH. In a previous report using NASH model mice, the protein expression of calcium-independent phospholipase A2β was decreased in the liver, 6 which may suggest increased LDL-PC. Regarding with PC-OOH, we speculate that the abundance of sum of PC species containing PUFA in the LDL of patients with NASH contributed to the increase in PC-OOH because PUFA were relatively easily oxidized.
This study revealed positive correlations between the severity of fibrosis in patients with NASH and several LDL-PC and LDL-PCOOH species. In particular, LDL-PCOOH is more reflective of its severity. Considering that oxidized LDL induced liver fibrosis, 2 PC-OOH in LDL might be one of the factors causing liver fibrosis.
One of the limitations of this study was a small sample size; this is because LDL separation, lipid extraction and LC-MS/MS analysis are complicated and time-consuming for large sample sizes. However, it is clear that analysing isolated lipoproteins using LC-MS/MS will provide beneficial information in the pathological analysis of NASH.
Conclusions
LDL-PC and LDL-PCOOH species were strongly correlated with the fibrosis score in NASH, which suggests that abnormal LDL is involved in the development of liver fibrosis.
Supplemental Material
Supplemental Material - Increased phosphatidylcholine and its hydroperoxides in serum low-density lipoproteins from patients with non-alcoholic steatohepatitis
Supplemental Material for Increased phosphatidylcholine and its hydroperoxides in serum low-density lipoproteins from patients with non-alcoholic steatohepatitis by Nanao Murakami, Toshihiro Sakurai, Arisa Yamahata, Akiko Sakurai, Kazuhiro Nouso, Yuki Fujii, Hitoshi Chiba and Shu-Ping Hui in Annals of Clinical Biochemistry.
Footnotes
Acknowledgements
The authors thank the Central Research Laboratory, Faculty of Health Sciences, Hokkaido University for kindly providing the workspace and laboratory apparatus. We thank members of the Laboratory for Advanced Lipid Analysis for kind discussion and technical advice. We also thank Fujifilm Co. Ltd. for funding.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Japan Society for the Promotion of Science, KAKENHI (grant numbers: 19K20174 and 22K11797) and Fujifilm Co. Ltd.
Ethical approval
This study was conducted with ethical approval from Okayama University (approval number: 1604-011), Okayama City Hospital (approval number: 30-2), and Hokkaido University (approval number: 18-69-6). Informed consent was obtained from all subjects involved in the study.
Guarantor
SPH.
Contributorship
NM, TS, AS, HC and SPH conceived the study. KN and YF were involved in protocol development, gaining ethical approval, patient recruitment and data analysis. NM, TS and AY were involved in sample preparation, data acquisition and analysis on LC-MS/MS. NM, TS, AS, HC and SPH wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
