Abstract
Objectives
The interpretation of delta check rules in a panel of tests should be different to that at the single analyte level, as the number of hypothesis tests conducted (i.e. the number of delta check rules) is greater and needs to be taken into account.
Methods
De-identified paediatric laboratory results were extracted, and the first two serial results for each patient were used for analysis. Analytes were grouped into four common laboratory test panels consisting of renal, liver, bone and full blood count panels. The sensitivities and specificities of delta check limits as discrete panel tests were assessed by random permutation of the original data-set to simulate a wrong blood in tube situation.
Results
Generally, as the number of analytes included in a panel increases, the delta check rules deteriorate considerably due to the increased number of false positives, i.e. increased number hypothesis tests performed. To reduce high false-positive rates, patient results may be rejected from autovalidation only if the number of analytes failing the delta check limits exceeds a certain threshold of the total number of analytes in the panel (N). Our study found that the use of the (
Conclusions
The simple
Keywords
Introduction
Reducing patient harm through minimizing the risk of laboratory error is a major safety principle of laboratory practice.1,2 Delta checks are an error-screening tool that calculates the difference between the current and the preceding results and compares this difference against a predefined limit. If this difference is within a predefined delta check limit, the result can be released to the clinical team. If the difference is greater than the predefined delta check limit, this raises the possibility of an error in the pre-analytical stage, including misidentified/swapped patient samples (‘wrong blood in tube’), drip arm contamination, delayed separation, haemolysis or wrong tube type. Less commonly, failed delta check may be due to analytical failure or transcription/transmission error. The result is then held back for further review and investigations before being released to the clinical team.
There is wide variation in the implementation of delta checks, 3 in part due to the lack of evidence base in guidance documents. 4 An approach for selecting optimized delta check limits that seeks to maximize accuracy of detection in a paediatric population has been described recently. 5 At the same time, the impact of the time interval within which the delta check rule is applied on the performance of delta check has been also investigated. 6
Delta check rules are usually set on individual analytes of interest. However, in practice, multiple tests are often grouped together as a panel for clinical assessment. In such instances, multiple delta check rules can be presented to laboratory practitioners as a panel; hence, interpretation of delta check rules in a test panel should be different from that of a single analyte, since the number of hypothesis tests (i.e. the number of delta check rules) applied is much higher and needs to be taken into account. In this work, we empirically examined the performance of delta check rules when applied to a panel of common laboratory tests to provide some guidance on their interpretation.
Methods
Data source
De-identified inpatient laboratory data from a laboratory network servicing a paediatric tertiary-level referral hospital, the Women’s and Children’s Hospital in Adelaide, Australia, was extracted.7,8 Patients with serial/longitudinal results were identified, and patients with single instance were discarded. For patients with two or more serial results, only the first two results were kept, to avoid oversampling of the same subject. After this step, all remaining patients would have exactly two results. The biochemistry and haematology platforms used were the Roche Cobas c6000 platform (Roche Diagnostics, Mannheim, Germany) and Beckman Coulter DxH-800 platform (Beckman Coulter, Brea, CA, USA), respectively. This study was undertaken as a part of an effort to optimize the delta check rules and was exempted from ethics approval.
Panel tests
Laboratory tests were grouped into four commonly encountered panels, corresponding to the renal panel (sodium, potassium, chloride, bicarbonate, anion gap, creatinine and urea), bone panel (total calcium, magnesium, phosphate and albumin), liver panel (protein, albumin, globulin, alanine aminotransferase, gamma-glutamyl transferase and alkaline phosphatase) and full blood count (haemoglobin, packed cell volume, red blood cell count, mean corpuscular volume, mean corpuscular haemoglobin, mean corpuscular haemoglobin concentration, platelet count, mean platelet volume, red cell distribution width and white blood cell count) for further analysis. For each of the panels, patients with missing values for one or more of the analytes in the panel were discarded. The demographic information of the patients is summarized in Supplementary Table 1.
Delta checks
Delta checks can be expressed as relative and absolute differences between two results. The relative difference for patients is calculated by: [(preceding measurement − current measurement)/preceding measurement]. The absolute difference for patients is calculated by: [preceding measurement − current measurement].
Analysis and simulations
Specificity and sensitivity are common performance measures in clinical settings. Specificity is described by the following formula: [true negative/(true negative + false positive)]. Sensitivity (true positive rate) is defined as: [true positive/(true positive + false negative)].
Selection of delta check limits
With the assumption that all results in our data-set are analytically correct, the delta check limit for various specificities (percentage of patient results that fall below the delta check limit) can be determined. By ranging the delta check limit from 1% to 2000% for both relative and absolute differences (expressed as a percentage of the mean of the measurements) for each biochemical and haematological analyte in the four panels, the required delta check for specificities of 70%, 80% and 90% is determined. Our delta check limits are set at these high specificities to reduce the number of unnecessary investigations. 9
Generation of randomly permutated data
Given the assumption of our data-set being analytically correct, to determine the sensitivity of the delta checks limits, a permutated/mismatched data-set was generated. For each of the four panels, the second set of measurements from each patient is randomly re-ordered/shuffled, so that it is now matched to the first set of measurements of a different patient.
Determination of sensitivity of each analyte
Using the delta check limit for specificity at 70%, 80% and 90% for relative and absolute differences, the sensitivity of each analyte is obtained by determining the fraction of patients that exceed the delta check limit in randomly permutated data-set. To obtain stable estimates for sensitivity, 20 rounds of permutation/mismatched data generation were performed, and the average sensitivity was reported.
Determination of performance for each panel of analyte
In a delta check, patients whose difference between paired sequential laboratory results is greater than the predefined limit will be flagged for further review/investigation. Here, true negatives (specificity) correspond to patients whose difference between their paired sequential laboratory results in the original matched data-set is below the predefined delta check limit, and false positives whose difference is greater than the predefined delta check limit. On the other hand, true positives (sensitivity) correspond to patients in the permutated/mismatched data-set whose difference in paired sequential laboratory results is above the predefined delta check and false negatives are those patients whose paired difference was below predefined delta check limit.
While delta check limits are set for individual analytes, a patient is usually having a panel of assays performed. For each panel of analytes, the specificity and sensitivity were determined at different thresholds, i.e. the number of delta check limits that a patient’s sample must violate in order for the patient to be flagged out as a mismatch. For example, a threshold of one in the renal panel means a patient will be flagged as a mismatch if any one of the seven analytes in the renal panel exceeds the delta check limits.
For each patient in the original matched data-set, the number of delta check limits exceeded by that patient is obtained. Next, the threshold is ranged from one to the total number of analytes in the panel, to determine the false-positive rates (FPRs) at each of the thresholds, remembering this data-set is assumed to be analytically correct. The specificity is then obtained by taking 1–FPR.
Similarly, for each patient in the randomly permutated data-set, the fraction of patients with the number of analytes exceeding the delta check limits is obtained at each threshold. This yields the sensitivity of the tests at the different thresholds.
Results
The number of patients available for statistical analysis for each of the common laboratory panels was as follows: renal panel – 10117 patients; bone panel – 1634 patients; liver panel – 6860 patients and full blood count – 19,766 patients.
Sensitivity at different delta checks limits
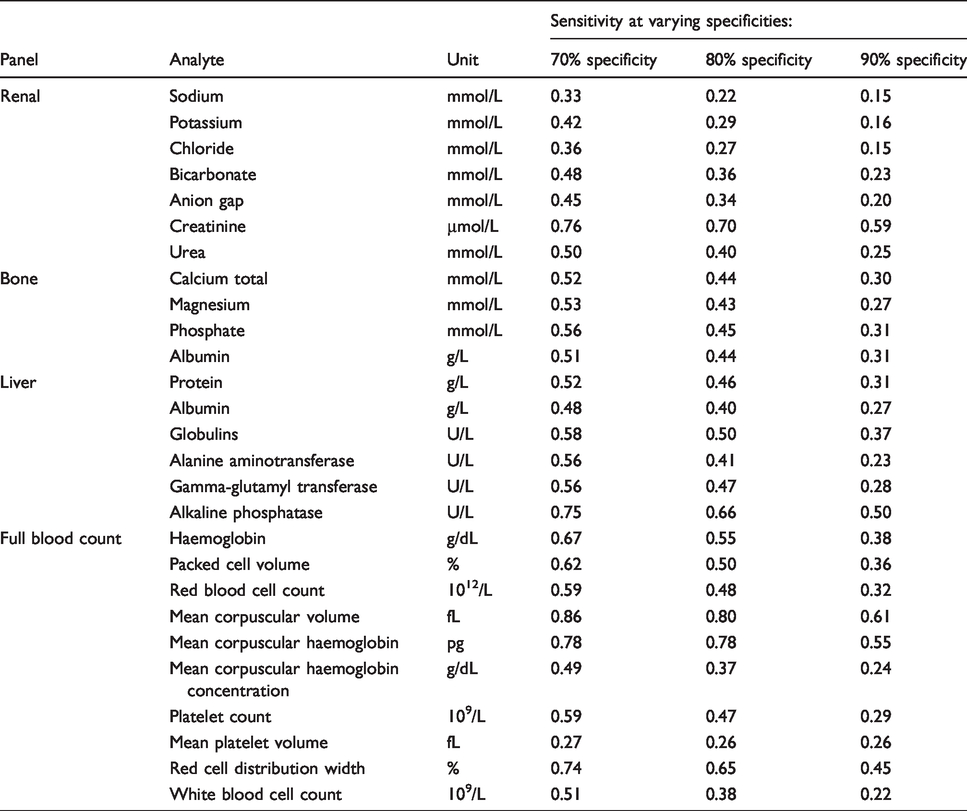
The relative and absolute delta checks limits for 70%, 80% and 90% specificity were determined individually for each of the analytes in the four panels (Supplementary Tables 2 and 3). Using these delta check limits, the sensitivities at each of the specificities were calculated and summarized in Tables 1 and 2. As expected, when specificity increases, the sensitivity decreases, as the delta check limits are being set at a more stringent (i.e. wider) values. The sensitivities obtained using either the absolute or relative difference delta check limits are similar.
Sensitivity obtained for absolute difference delta checks at different levels of specificity (70%, 80% and 90%) for the analytes in the common laboratory test panels.
Sensitivity obtained for relative difference delta checks at different levels of specificity (70%, 80% and 90%) for the analytes in the common laboratory test panels.
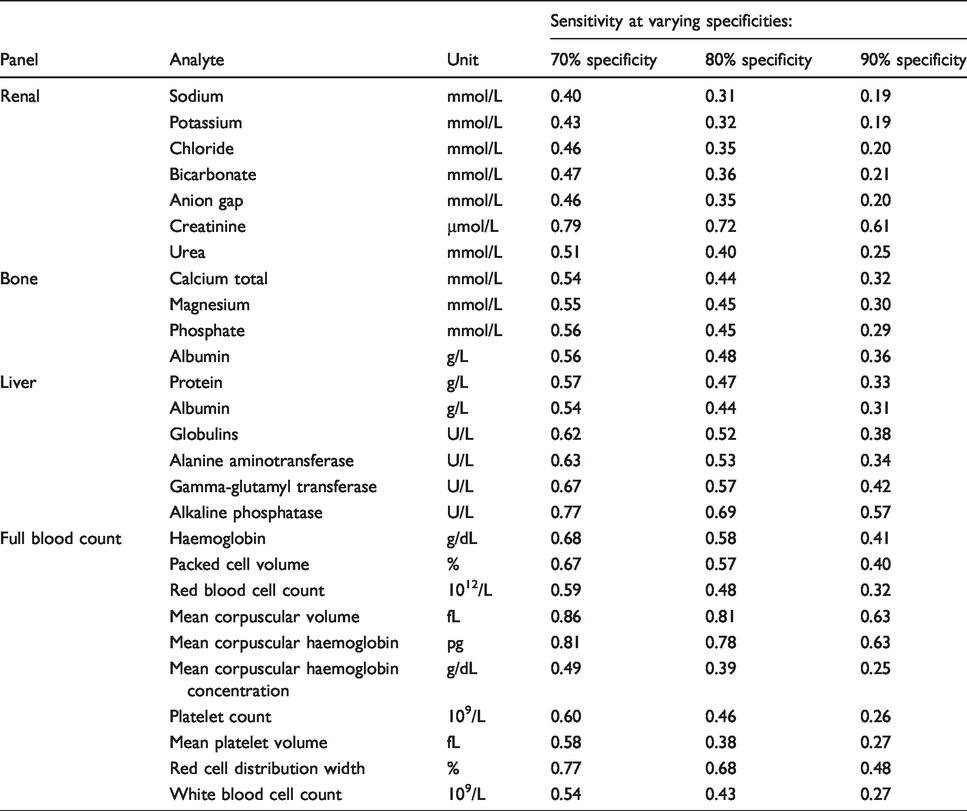
The sensitivities obtained for the individual analytes in the same panel widely differed. For example, in the renal panel using the absolute delta check for creatinine of 13 μmol/L at 90% specificity was found to have a sensitivity of 59%. This is in contrast to the remaining analytes in the renal panel, which only showed sensitivities ranging from 15% to 25%. Similarly, in the liver panel using absolute delta check limit of 89 U/L at 90% specificity for alkaline phosphatase had a sensitivity of 50% in comparison to other analytes in the panel, with sensitivities ranging from 23% to 37%.
Specificity and sensitivity at different thresholds of delta check limit violations
For a patient taking a panel test,
Specificity and sensitivity obtained for different threshold violations of absolute difference delta checks limits. Three different sets of delta check limits for specificities of 70%, 80% and 90% specificity at the single analyte level were used.

Specificity and sensitivity obtained for different threshold violations of relative difference delta checks limits. Three different sets of delta check limits yielding specificities of 70%, 80% and 90% specificity at the single analyte level were used.

From Tables 3 and 4, with increasing specificity determined at the individual analyte level (70%, 80% and 90%), the specificity increases with a concomitant decrease in sensitivity when applied as a panel, likely due to the larger number of hypothesis tests performed. With increasing
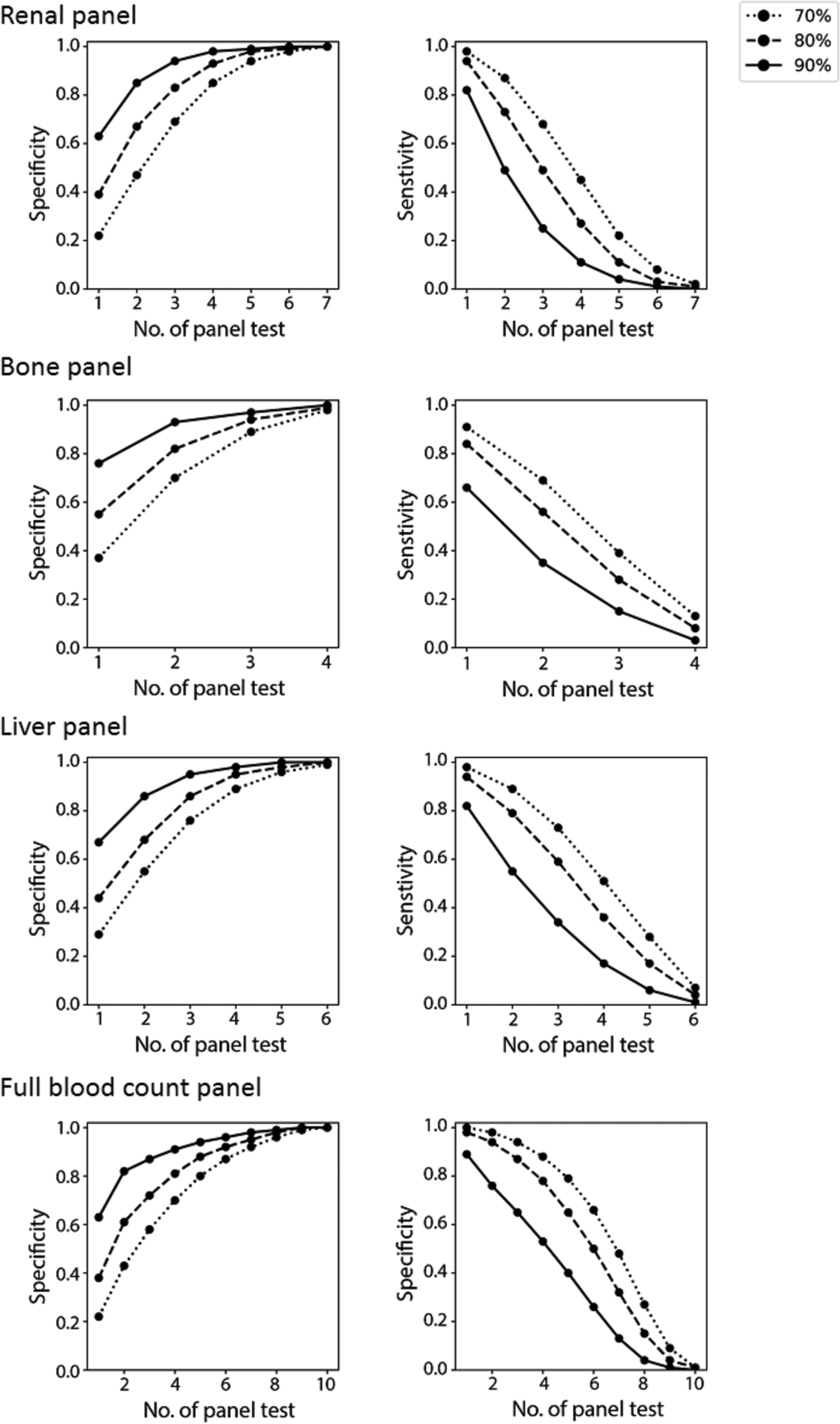
Sensitivity and specificity when absolute difference delta check rules are applied and interpreted collectively in a test panel at increasing threshold (i.e. number of tests failing delta check in a panel) for individual analytes with specificities set at 70%, 80%, 90% (individual lines).
Using the delta check limits at the individual analyte level associated with 90% specificity for the absolute difference, we observed that using a threshold of
The sensitivities obtained using the
Discussion
There is a general requirement for laboratories to quickly return the results to the clinical team for timely decision-making and intervention. This is demonstrated by the turnaround time targets that are set as part of the overall quality indicator of a laboratory.1,2 However, the need for speed should be carefully weighed against the over-riding consideration for quality and patient safety. There is little benefit in delivering an erroneous result quickly to clinicians, which may cause significant harm. Delta checking is a practice that aims to automate the clinical verification of results generated by the laboratory to achieve faster turnaround time and standardize the postanalytical (verification) process for better consistency. Algorithms and implementation of delta checking are gaining greater attention with the increasing use of autoverification in laboratory information systems.
However, there remain gaps in the evidence base to guide this practice. 4 In this work, we reported the performance of delta check rules when applied on multiple analytes in a test panel. In general, the greater the number of tests that are present in a panel, the higher the probability that any of the analytes may be flagged (low specificity), which may trigger a resource-intensive investigative exercise.5,9 It is desirable to have as lowest a false-positive rate as possible to avoid unnecessary rework. Moreover, in a high-volume laboratory setting, even a low false-positive rate can translate into considerable number of delta check flags that need to be acted upon. Over time, this can reduce efficiencies and risks alarm fatigue in the laboratory staff.
To compensate for the higher probability of flagging in panel tests, the delta check limits for each analyte can be set wider (i.e. detecting larger differences, more specific errors). However, the reduction in the false-positive rate is small, as the number of tests in the panel (i.e. the number of hypothesis testing) remains the same. A high overall specificity can only be achieved when one considers multiple flags as a true positive.
There are several important limitations to this study. Firstly, the data were derived from a paediatric inpatient population and may require further validation in other populations (such as outpatient, primary care, specialist clinics, adults) to generalize the findings. The neonatal age group is under-represented in this study and should be examined in a larger cohort. Secondly, the design of the simulation of this study only addresses the wrong blood in tube scenario. The suggested approach may perform differently for other causes of failed delta checks, such as drip arm contamination, haemolysis or wrong tube type, particularly where these factors affect a smaller number of analytes. Additionally, the wrong blood in tube error may not be truly random since they are more likely to occur in the same physical location (e.g. the same ward), and therefore may display similar result characteristics among the patients.
In practice, a laboratory staff are likely to continue reviewing any flags in a panel of tests, and if the difference is very large (e.g. multiple times greater than the delta check limits), then closer investigation is warranted. On the other hand, the results of this study also indicate that if multiple delta check limits are flagged, then it is highly probable that there is an error in the total analytical process that should be investigated before reporting. In other words, do not ignore a sample that has multiple delta check flags. The number of flags that is considered to be significant is dependent on the delta check limits and the number of tests within the panel. The
Supplemental Material
ACB904749 Supplemental Material - Supplemental material for An approach to optimize delta checks in test panels – The effect of the number of rules included
Supplemental material, ACB904749 Supplemental Material for An approach to optimize delta checks in test panels – The effect of the number of rules included by Rui Zhen Tan, Corey Markus and Tze Ping Loh in Annals of Clinical Biochemistry
Footnotes
Contributorship
RZT and TPL researched literature and conceived the study. CM extracted the data. RZT developed the statistical modelling and performed the analysis. RZT, CM and TPL wrote, edited, reviewed and approved the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was undertaken as a part of an effort to optimize the delta check rules and was exempted from ethics approval.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Guarantor
TPL.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
