Abstract
Background
Most errors in the clinical laboratory occur in the preanalytical phase. This study aimed to comprehensively describe the prevalence and nature of preanalytical quality monitoring practices in UK clinical laboratories.
Methods
A survey was sent on behalf of the Association for Clinical Biochemistry and Laboratory Medicine Preanalytical Working Group (ACB-WG-PA) to all heads of department of clinical laboratories in the UK. The survey captured data on the analytical platform and Laboratory Information Management System in use; which preanalytical errors were recorded and how they were classified and gauged interest in an external quality assurance scheme for preanalytical errors.
Results
Of the 157 laboratories asked to participate, responses were received from 104 (66.2%). Laboratory error rates were recorded per number of specimens, rather than per number of requests in 51% of respondents. Aside from serum indices for haemolysis, icterus and lipaemia, which were measured in 80% of laboratories, the most common errors recorded were booking-in errors (70.1%) and sample mislabelling (56.9%) in laboratories who record preanalytical errors. Of the laboratories surveyed, 95.9% expressed an interest in guidance on recording preanalytical error and 91.8% expressed interest in an external quality assurance scheme.
Conclusions
This survey observes a wide variation in the definition, repertoire and collection methods for preanalytical errors in the UK. Data indicate there is a lot of interest in improving preanalytical data collection. The ACB-WG-PA aims to produce guidance and support for laboratories to standardize preanalytical data collection and to help establish and validate an external quality assurance scheme for interlaboratory comparison.
Introduction
Within laboratory medicine, the total testing process (TTP) encompasses every step from test request to receipt of result. 1 The TTP can be divided broadly into three distinct phases. Firstly, the preanalytical phase, which includes every action from test request, through sample collection and transport, registration on the relevant Laboratory Information Management System (LIMS) up to the initiation of specimen analysis. Secondly, the analytical phase, which involves measurement of the required analyte, technical validation of the result and release to the LIMS. Finally, in the postanalytical phase, results are interpreted, clinically authorized and communicated to the requesting clinician. Errors may be introduced in any of these phases, and therefore the term ‘testing error’ cannot solely apply to errors made in the analytical phase or within the laboratory. Laboratories might employ mechanisms to identify and nullify the effects of testing error in real time, but often these are neither recorded nor monitored. 2 The impact of testing errors may be difficult to assess as they may be distant from the cause both spatially and in time, but may include delayed diagnoses, increased demand on resources, longer hospital stays, or inappropriate clinical decision making based on erroneous results. 3
The 2013 Francis report has increased the public focus on quality of care 4 and highlighted the requirement to improve the patient experience through reduction of errors in healthcare provision. In the TTP it has been reported that only 7–13% of errors actually occur in the analytical phase and that the most errors could be classified as either preanalytical (46–68%) or postanalytical (18–47%).5–7
Clinical laboratories in the UK are subject to regular and rigorous quality assessments by official bodies such as Clinical Pathology Accreditation (CPA) soon to be replaced by the United Kingdom Accreditation Service (UKAS). To retain accreditation, laboratories must demonstrate that the analytical phase is monitored through a combination of internal quality control and participation in comparative external quality assurance (EQA) schemes. The relatively low proportion of errors in the analytical phase most likely reflects improvements in analytical quality resulting from standard operating procedures, quality assurance and methodological developments, including increased automation. In contrast, the extra-analytical phases are rarely subject to routine assessments and often utilize a variety of staff groups not necessarily under the managerial control of the laboratory. Traditionally, there has been limited attention on what happens to samples before they reach the laboratory and as a result preanalytical errors may be under-reported and their effects underestimated.8–10 Further, there are no explicit standards requiring regular monitoring of preanalytical error. The move to ISO15189 extends the scope of the laboratory into the extra-analytical phases where quality management system implementations and continual assessment of performance may harvest the biggest quality improvement gains in the TTP. 11
There have been numerous international efforts to raise awareness of the importance of the extra-analytical phase.12–14 Conferences on the preanalytical phase are held by the European Federation of Laboratory Medicine (EFLM) ‘Working group on laboratory errors and patient safety’ (WG-LEPS).12,13 A series of quality indicators were designed by the International Federation of Clinical Chemistry and Laboratory Medicine Working Group for Laboratory Errors and Patient Safety (IFCC-WG-LEPS), 34 of which apply to the preanalytical phase. These indicators are used as the basis for a European wide data collection exercise. The Australian Key Incident Monitoring and Management System (KIMMS) is one of a few EQA schemes for the extra-analytical phase. 14 At present, no UK-based schemes exist, although a pilot scheme is currently in the early stages of development. Preanalytical errors encompass specimen identification and sampling problems, transportation issues and incorrect volume. Any instance where a sample is rejected on the basis that it fails to meet specimen acceptance criteria may also be counted as an error.11,15
As stated by Lippi and Guigi, defined quality indicators can be used to identify defects in the process when implemented as part of a quality improvement plan. 16 This requires the ability to record, monitor and improve performance to assess the quality of service provision. 16 Variations in reported error rates may be introduced by a number of factors such as data collection mechanisms, error classification and difference in accession number assignation. These variations make standardization and benchmarking difficult and must be considered when judging performance against targets or ideals.17,18 Existing practices for collating and monitoring preanalytical errors within laboratories are likely to be laboratory specific and not known on a national level. We, the Association of Clinical Biochemistry and Laboratory Medicine Preanalytical Phase Working Group (ACB-WG-PA), therefore issued a survey to collect these data with a view to standardizing the processes in the future.
Methods
A survey consisting of 12 questions was designed (see Appendix 1) to capture existing steps laboratories in the UK currently take to monitor and record preanalytical errors. The survey aimed to discover what systems and platforms were used, whether preanalytical errors were manually or electronically monitored and recorded, and how they were classified (i.e. per sample or per request). The survey also aimed to gauge the current interest of laboratories for an EQA scheme focussed on the preanalytical phase. The proposed EQA scheme would allow the comparison of preanalytical error rates between laboratories and enable support and education from the ACB-WG-PA.
The survey was sent out on behalf of the ACB-WG-PA to all heads of Clinical Biochemistry departments via Survey Monkey and extracted data were sent back to the group. Data were analysed using simple descriptive statistics.
Results
Overall 104 (66.2%) of 157 laboratories polled responded to the survey representing a good proportion of UK Clinical laboratories. Several laboratories provided multiple responses, which if similar were excluded but were included when significant differences were observed since they may represent different sections of a large laboratory. There were a total of 195 individual responses. With the exception of questions 1 (178 responders) and 9 (137 responders) all other questions were answered by all the responding laboratories.
The data for questions 1, 2, 4 and 5 were trimmed to look at LIMS; request counting mechanism used; and use of indices and analytical platforms (only once per site). All other questions showed a mixed picture across the site and therefore results from all responders were included.
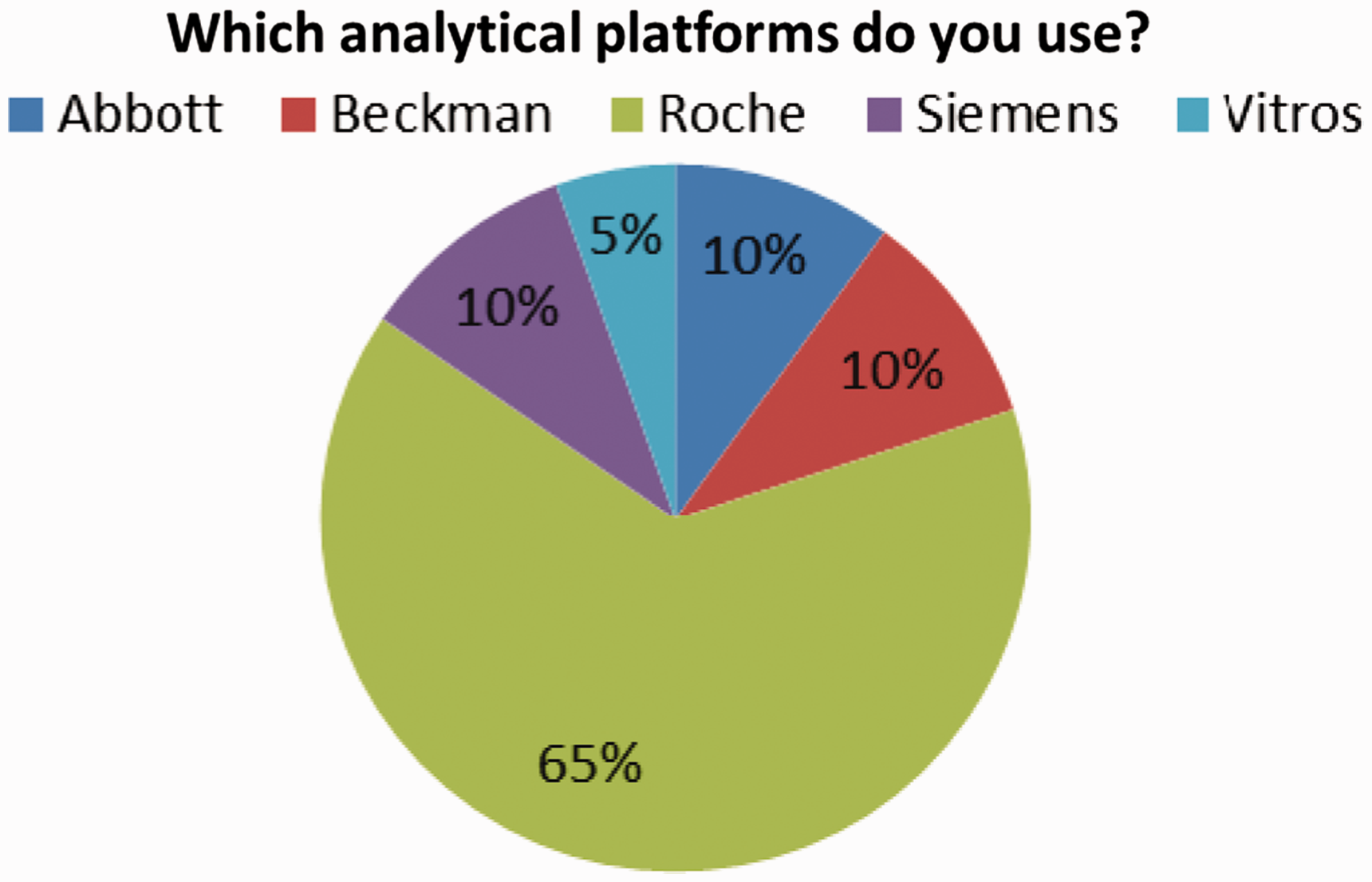
The results for question 1 clearly showed that there are a wide variety of LIMS currently in place in the UK with a few clear market leaders including CliniSys Winpath (18%), APEX (15%), Telepath (12%) and Clinisys Labcentre (9%) (Figure 1). Question 4 complemented these data by looking at the major analytical platforms in use and showed that although Roche is currently the market leader (65%), Abbott (10%), Siemens (10%) and Beckman Coulter (10%) also have significant market share (Figure 2).
Suppliers of laboratory information management systems in the UK clinical laboratories surveyed. Laboratory analytical platforms in use in UK clinical laboratories surveyed.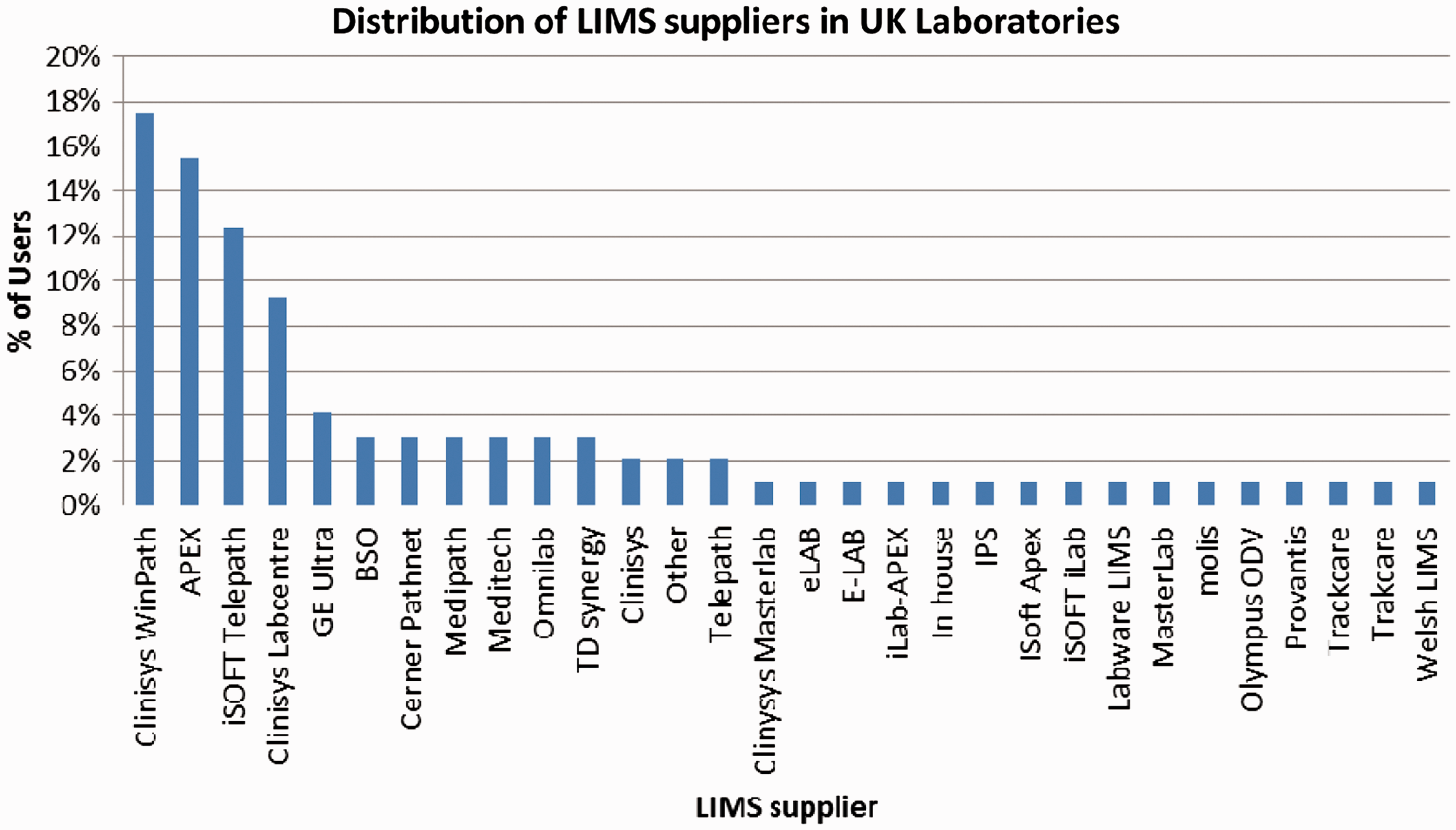
Question 2 illustrated the differences in how requests are counted, with 51% of laboratories using a separate barcode for each sample and 49% using a separate barcode for each request.
Similarly, question 3 queried whether laboratories currently record preanalytical errors manually or electronically, and this showed a 67.5% to 32.5% split, respectively. This also indicates that some laboratories use both methods of data collection and record data both manually and electronically.
Question 5 looked at the use of automated serum indices for haemolysis, icterus and lipaemia. Responses indicate that 20% of laboratories do not use automated serum indices.
As question 6 highlights, 34.5% of laboratories do not routinely monitor any preanalytical quality indicators.
Question 7 looked at what interest there would be in guidance and quality assurance for indicators of preanalytical phase quality. Among respondents, 95.9% expressed an interest in guidance, with 29.4% of these interested in guidance specific to their own LIMS. In response to questions 10–12, 91.2% of laboratories favoured a central location for these guidelines, 72.7% were interested in attending meeting days to share initiatives and 85.6% were interested in e-learning packages.
Question 8 showed that 91.8% of laboratories are interested in the establishment of an EQA scheme to allow interlaboratory comparisons of preanalytical error.
Figure 3 summarizes the responses of those laboratories that measure preanalytical error (70%) to question 9 and highlights that only two markers (booking-in errors and mislabelling of samples) are routinely measured by 50% or more of these laboratories. Also, 30% of laboratories do not monitor any preanalytical markers.
Frequency of reporting of preanalytical markers in UK clinical laboratories surveyed.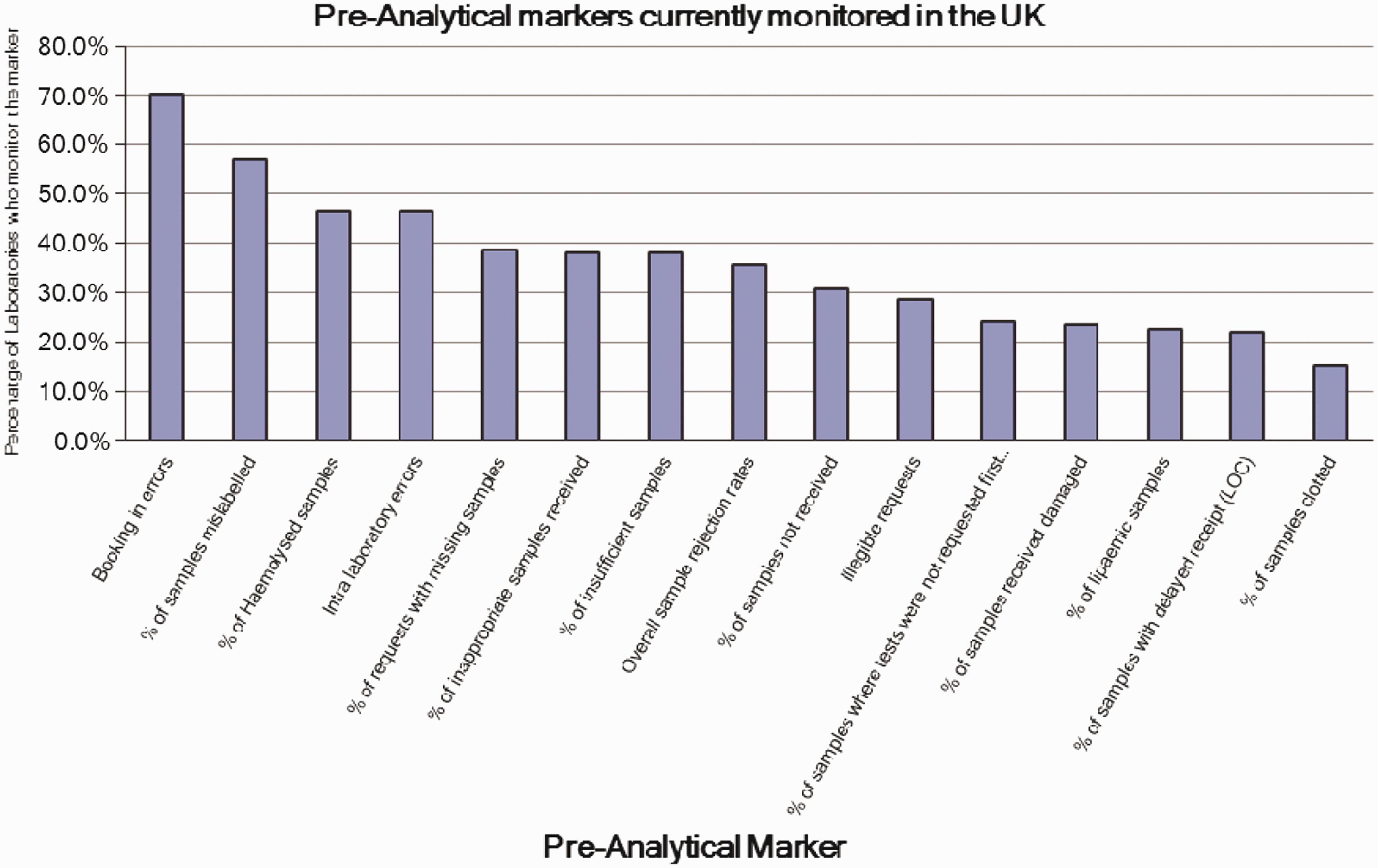
Discussion
The survey received 195 responses made up from 106 distinct sites from 157 polled heads of department. Respondents were geographically evenly spread across the UK and all hospital types were represented, suggesting the survey will accurately reflect the UK situation as regards monitoring and reporting of preanalytical error.
The survey highlights the wide range of LIMS currently in use in the UK and how complex setting up guidelines for collection of preanalytical quality markers will be. Interestingly, there does not appear to be a correlation between LIMS supplier and whether a site monitors preanalytical error, indicating that the drive to collect these markers comes from the laboratory. The results show that there are three or four market leaders in LIMS provision and initial guidance could be targeted to users of these systems, with advice for less frequently used systems to follow.
As with the LIMS, there are several providers of analytical platforms that dominate the market with Roche currently having the market lead followed by Abbott, Siemens and Beckman Coulter. Again, these differences in technology may make producing a standard guidance difficult.
One major issue with data collection is in how laboratories count requests and associated errors. In some system configurations, each sample is given a unique barcode, whereas in other configurations, all samples associated with a request are given individual or duplicate barcodes. Laboratories recording errors as a proportion of the total number of samples rather than the total number of requests will have a lower perceived frequency than those laboratories who count errors as a proportion of total requests. Some laboratories may also count multiple errors on a sample whereas other laboratories may just count this as one sample with errors. In our survey, both counting methods are used with similar frequency, highlighting the need for standardization with respect to counting method when analysing and comparing preanalytical error rates between laboratories.
Two-thirds of laboratories record data on preanalytical errors manually, which could lead to under-reporting of certain errors. The aims of the ACB-WG-PA would be to provide guidance on how to collect the data automatically wherever possible. While this may be difficult initially, in the longer term it will lead to better standardization and data will be easy to audit and compare, providing a good quality indicator for accreditation purposes. Surprisingly, in the developed UK market, was the finding that there are still 20% of laboratories not using automated serum indices meaning they are unlikely to identify, by human judgement, those samples where haemolysis, lipaemia or icterus will cause significant analytical interference. These laboratories risk reporting inaccurate results with potentially serious consequences for patients. Perhaps more worryingly, given the arrival of ISO15189 and the growing literature on preanalytical errors, 30% of the UK laboratories surveyed do not routinely monitor any preanalytical markers whatsoever. Although such laboratories may perform audits on an infrequent basis, the ACB-WG-PA believes that preanalytical quality markers should be routinely monitored and recorded.
Encouragingly, the data show that a wide variety of markers are being monitored by a significant proportion of UK laboratories, and these laboratories can be used to help establish data collection systems. However, there were several causes for concern. Despite the recent Pathology Quality Assurance Review and the Francis report, just two markers are recorded by greater than 50% of laboratories, those for mislabelled samples and for booking-in errors. Perhaps most surprising was that markers of sample quality such as haemolysis, which is well documented to interfere in test results, are monitored by less than 50% of laboratories.
Results highlight that laboratories, on the whole, are aware of the issue of preanalytical error and keen to establish recording and monitoring systems, and perhaps just have not had the guidance or time to do so. The fact that almost all laboratories are interested in the establishment of guidelines is pleasing. The ACB-WG-PA aims to fill this knowledge gap primarily through provision of an online home for preanalytical error guidelines. Online resources can be followed or supported by e-learning packages and possibly one-day meetings, depending on demand. Given the overwhelming interest expressed, the ACB-WG-PA will also work with UK quality scheme organizers to establish an EQA scheme so that laboratories can see how they compare to other laboratories in the UK and further afield. UK labs are used to using EQA schemes to compare their analytical performance with other laboratories as part of their clinical governance framework. Differences in error frequency between laboratories may act as the starting point to investigate the cause and implement changes if required. EQA providers can also support any standardization and harmonization processes. When enough data are collected, standards could establish maximum acceptable frequencies for each error. These could then be monitored to drive the collection of preanalytical markers by bodies such as the Royal College of Pathologists, the Care Quality Commission, Medicines & Healthcare products Regulatory Agency (MHRA) or UKAS. The ACB-WG-PRE are willing to work with these organizations to establish these new Key Performance Indicators.
In conclusion, results of this survey suggest that too few laboratories are collecting regular and comprehensive data on preanalytical error rates. This situation will need to change given the arrival of ISO15189 and the recent Barnes and Francis reports, and laboratories have expressed a desire to get involved. Although many of the causes of preanalytical error maybe outside of the direct control of the laboratory, the laboratory is uniquely placed to provide data and target problem areas to improve the TTP. Improvement in the TTP would serve to improve the quality of specimens, reduce costs, strengthen the credibility of the laboratory and ultimately improve patient care. It is therefore essential that there is a continuous cycle of education and monitoring to ensure that any improvements to the TTP are maintained and built upon. A national EQA scheme comparing laboratories could help to drive this. Looking at the data, it is encouraging that there is almost universal interest in improving the UK situation, and the ACB-WG-PA will aim to produce guidance and support for laboratories to achieve standardized preanalytical marker data collection and to help establish and validate a scheme for interlaboratory comparison of the collected data.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
