Abstract
Background
Selective serotonin reuptake inhibitors (SSRIs) block the serotonin transporter on neurons, but also on platelets, thus decreasing platelet serotonin concentrations in users of SSRIs. Data on plasma-free serotonin concentrations in SSRI users are lacking, while plasma-free serotonin is available for receptor binding and plays a role in several pathophysiological processes. We therefore measured the plasma-free and platelet serotonin concentrations in users of SSRIs and age-matched healthy controls, and we analysed plasma concentrations of the serotonin precursor tryptophan and serotonin metabolite 5-hydroxyindoleamineacetic acid (5-HIAA).
Methods
For this cross-sectional single-centre case control study, participants were recruited at the departments of Psychiatry and General Medicine. High-performance liquid chromatography combined with tandem mass spectrometry (LC-MS/MS) was used to measure plasma-free and platelet serotonin, plasma tryptophan and 5-HIAA concentrations. Preanalytical conditions were optimized by careful blood collection, rapid sample handling, high-speed centrifugation, drug and diet restrictions and age-matched controls.
Results
In 64 SSRI users, median concentrations of plasma-free and platelet serotonin were 10-fold and 14-fold lower, respectively, than in 64 matched controls. Patients using higher dose SSRIs or those with higher affinity for the serotonin transporter had lower plasma-free and platelet serotonin concentrations. Compared with controls, SSRI users had similar median plasma tryptophan concentrations but slightly higher plasma 5-HIAA concentrations.
Conclusion
SSRI users have low platelet serotonin and low plasma-free serotonin. This could not be explained by lower concentrations of its precursor tryptophan, and only partially by increased breakdown to 5-HIAA.
Introduction
Serotonin is synthesized from the essential amino acid tryptophan, mainly in the gut by the enterochromaffin cells and the brain. After uptake by the serotonin transporter, it is metabolized to 5-hydroxyindoleacetic acid (5-HIAA) by monoamine-oxidase A (MAO-A) in the lung and liver. 1 Serotonin is involved in a range of physiological processes including gastro-intestinal motility, vascular tone, metabolic regulation and mood regulation. 2
In the circulation, serotonin is mainly transported by platelets. In healthy individuals, only a small percentage circulates free in the plasma. Plasma-free serotonin is available for binding to receptors, but it cannot cross the blood–brain barrier.3–5 Although plasma-free serotonin is implicated in several pathophysiological processes, little information regarding its concentrations in humans is available. To obtain reliable information about plasma-free serotonin concentrations, it is important to take specific preanalytical conditions into account and use a sensitive analytical method.3,6,7
Selective serotonin reuptake inhibitors (SSRIs) increase synaptic concentration of serotonin by blocking the serotonin transporter on neurons. Platelets also express serotonin transporters; if these transporters are blocked, then platelet serotonin concentrations decline in users of SSRIs. 8 Additionally, platelet serotonin concentration is affected by the affinity of SSRIs for the serotonin transporter, which differs between various SSRIs. 9 Blocking the serotonin transporter could therefore have consequences for clearance of serotonin from the circulation. Despite the importance of circulating free serotonin, there is only scarce information available about plasma-free serotonin concentrations in humans using SSRIs. 10 This study investigated the effect of one type of SSRI (fluvoxamine) on plasma-free and platelet serotonin by high-performance liquid chromatography in combination with fluorometry.
High-performance liquid chromatography combined with tandem mass spectrometry (LC-MS/MS) enables sensitive and specific quantification of plasma-free serotonin concentrations. Using this advanced approach, we analysed plasma-free and platelet serotonin concentrations in users of SSRIs and compared these with concentrations in healthy, age-matched controls. To investigate whether altered serotonin concentrations in SSRI users could be explained by different concentrations of the serotonin precursor tryptophan or by a change in breakdown to the serotonin metabolite 5-hydroxyindoleacetic acid (5-HIAA), we also analysed plasma concentrations of these components.
Methods
Patients
SSRI users were recruited from the Depression and Anxiety outpatient clinic of the University Center for Psychiatry and the Department of General Practice of the University Medical Center Groningen. Patients from the University Center for Psychiatry were recruited in consecutive order when they visited the outpatient clinic. All patients from the general practitioners that were on SSRIs received a letter from their general practitioner to request their participation in the study. As serotonin concentrations are affected by age, 11 age-matched healthy controls were recruited among employees and visitors (mainly family members of patients) of the University Medical Center Groningen. All participants were 18 years or older and gave their written informed consent.
SSRI users were eligible when they had used an adequate SSRI dose for at least six weeks (minimum dose per day: citalopram 20 mg; escitalopram 10 mg; fluoxetine 20 mg; fluvoxamine 100 mg; paroxetine 20 mg; sertraline 50 mg). Exclusion criteria for all participants were use of any other drugs that might interfere with serotonin metabolism, reuptake of serotonin or platelet function. These drugs were: aspirin, antidepressants (apart from SSRIs in SSRI users), antipsychotics, anticonvulsants and anxiolytics. This study was approved by the institutional medical ethics committee and complies with the Declaration of Helsinki. The study is registered in the Dutch Trial Register (NTR3393).
SSRI users were asked to fill out a short questionnaire regarding the SSRI they used and the treatment indication. The inhibitory constants of SSRI were obtained from the Psychoactive Drug Screening Program Ki Database (https://pdsp.unc.edu/databases/kidb.php).
Blood collection and processing
Twenty millilitres of blood were drawn in two 10 mL ethylenediaminetetraacetic acid (EDTA) tubes. A butterfly needle with vacutainer system was used to minimize platelet activation and corresponding release of platelet content. 12 As serotonin-containing foods can affect plasma serotonin concentrations, 13 patients were asked not to eat bananas and walnuts up to 48 h prior to blood collection. Blood was kept at room temperature and immediately transported to the laboratory for processing.
Both 10 mL tubes were processed to obtain platelet-rich plasma (PRP) and platelet-poor plasma (PPP). For PRP, the blood was centrifuged at 120 g for 30 min at room temperature. PRP (0.5 mL) was used to assess platelet count (Sysmex XE-2100). To obtain PPP, the PRP was centrifuged at 1800 g for 10 min at room temperature. PRP and PPP were stored in Sarstedt cups with EDTA plus the antioxidant L(+) ascorbic acid at –80°C.
Serotonin, tryptophan and 5-HIAA analysis
Serotonin, tryptophan and 5-HIAA concentrations were analysed using online sample preparation coupled to isotope dilution liquid chromatography (Symbiosis™ System, Spark Holland, Emmen, the Netherlands) in combination with a XEVO TQ-MS tandem mass spectrometer (Waters, Milford, CO, USA). 13
The concentration of serotonin in platelets was calculated using the following formula
Concentration in platelet fraction = concentration PRP–concentration PPP.
Statistical analysis
We calculated that 63 SSRI users and 63 matched healthy controls were needed to detect a difference of 0.5 standardized effect size with 80% power and a two-sided α = 0.05. For each group, one extra participant was included.
For comparison of plasma-free and platelet serotonin concentrations between patients and healthy controls, Mann-Whitney U tests were used, as data were not normally distributed. To determine the effects of different SSRIs on plasma-free and platelet serotonin concentrations, we analysed their effect in multivariate regression analyses after a log transformation of the serotonin data. The relative SSRI dose was calculated as a reference to the standard dose of the respective SSRI. A P value < 0.05 was considered significant. Statistical analysis was performed with SPSS (version 25). GraphPad (version 6) was used to create the artwork.
Results
SSRI users had a median age of 48 years (range 22–76 years) and their controls had a median age of 54 years (range 20–74 years). Clinical characteristics are shown in Table 1.
Clinical characteristics of SSRI users (n = 64).
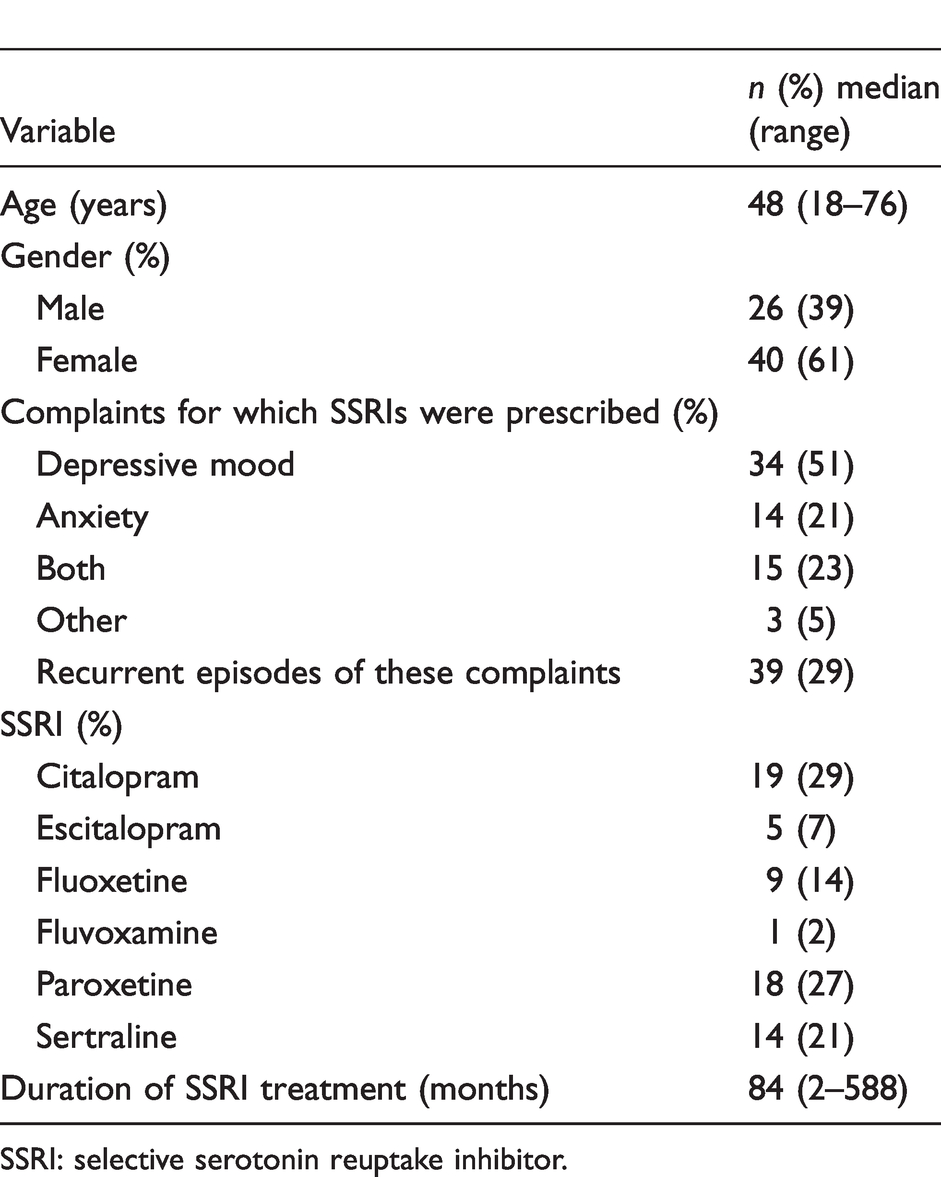
SSRI: selective serotonin reuptake inhibitor.
Median plasma-free serotonin concentrations were approximately 10-fold lower in SSRI users (1.9 nmol/L, range 0.1–38 nmol/L) than in matched healthy controls (19 nmol/L, range 3.8–406 nmol/L, P < 0.001) (Figure 1(a)). Median serotonin concentrations in platelets were 14-fold lower in SSRI users (117 nmol/L, range 11–2539 nmol/L) than in controls (1635 nmol/L, range 459–4523 nmol/L, P < 0.001) (Figure 1(b)). Platelet count did not differ between SSRI users and controls (Supplementary Figure 1).
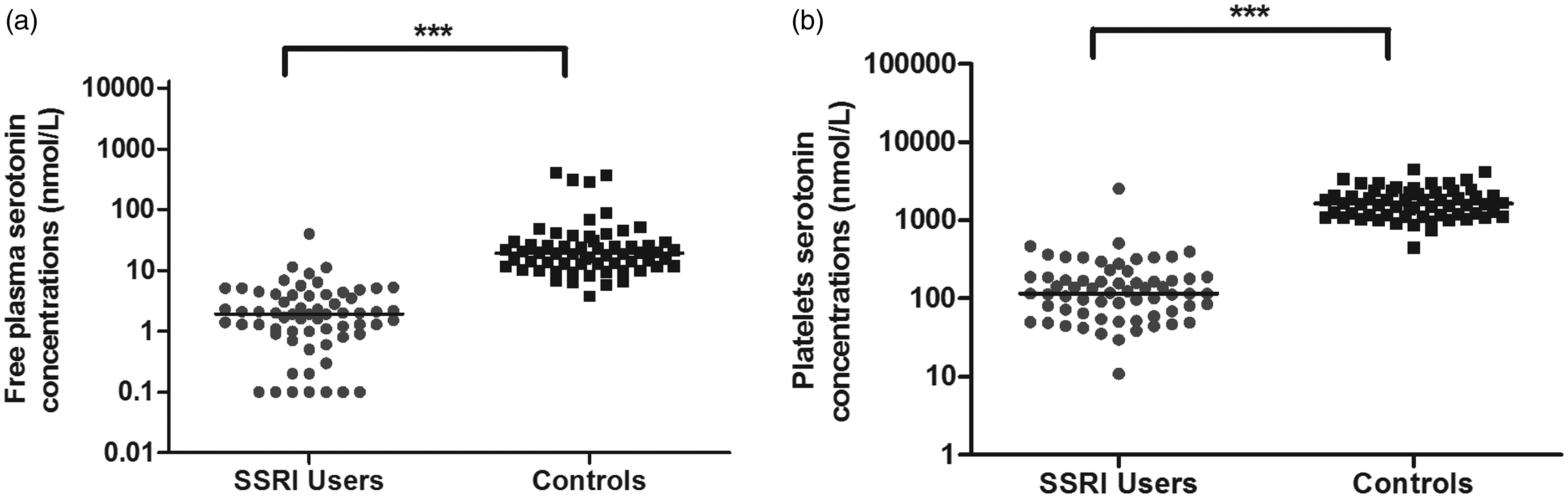
(a) Plasma-free serotonin concentrations and (b) platelet serotonin concentrations in 64 users of selective serotonin reuptake inhibitors (SSRIs) and 64 matched healthy controls. Medians are indicated with a horizontal line, ***P < 0.001.
We investigated potential contributing factors to the low plasma-free serotonin concentrations in SSRI users. We determined whether the plasma-free and platelet serotonin concentrations were associated with the binding affinity of different SSRIs for the serotonin transporter or the relative dose prescribed. A smaller Ki (hence, a higher affinity of the SSRI for the serotonin transporter) and a higher relative SSRI dose were both associated with lower (log transformed) plasma-free serotonin concentration (βki = 0.090, P < 0.05, βdose –0.349, P < 0.001) and platelet serotonin concentration (βki = 0.067, P < 0.001, βdose –0.317, P < 0.001).
To determine whether enhanced breakdown of serotonin or reduced availability of the serotonin precursor tryptophan could be responsible for the very low plasma-free serotonin concentrations, we measured plasma tryptophan and 5-HIAA concentrations. In SSRI users and healthy controls, plasma tryptophan concentrations were similar (Supplementary Figure 2(a)). Plasma 5-HIAA concentrations were slightly higher in SSRI users (median 40 nmol/L, range 25–207 nmol/L) than in healthy controls (median 35 nmol/L, range 19–106 nmol/L, P < 0.05) (Supplementary Figure 2(b)).
Discussion
This is the first study in which plasma-free serotonin was associated with the type and dose of SSRI in a large group of SSRI users. Compared with healthy age-matched controls, we found low platelet serotonin concentrations and low plasma-free serotonin concentrations in SSRI users. This observation could not be explained by lower concentrations of its precursor tryptophan, and only partially by increased breakdown to 5-HIAA. However, the low plasma-free serotonin concentrations could potentially be explained by an alternative mechanism. As serotonin transporters are blocked by the SSRI, the main breakdown route of serotonin in the lung and liver is no longer available. Our results suggest that in SSRI users, serotonin is quickly metabolized via an alternative route. When high affinity-low capacity transporters such as serotonin transporters are blocked by SSRIs, low affinity-high capacity transporters such as organic cation transporters (OCT) and plasma membrane monoamine transporters can take over the transport of serotonin.14,15 In mice, OCT is upregulated in the brains of serotonin transporter knock-out mice. 16 This has not yet been investigated for the liver. However, liver cells do express OCT1 and OCT3 and can effectively take up serotonin. 17 Potentially, breakdown of serotonin via this high capacity OCT route may induce an elementary scavenger mechanism to capture as much plasma-free serotonin as possible in order to diminish its detrimental effects, for example on the cardiovascular system. Selective knock-out of OCT in mice could shed further light on this mechanism.
In this study, LC-MS/MS was used to measure plasma and platelet serotonin concentrations in users of SSRIs, which enabled us to detect very low concentrations. This is because online sample preparation with LC-MS/MS is more sensitive and specific than immunoassays and conventional chromatographic techniques, which are often used for measurement of serotonin. 18 Despite the careful procedures, a potential limitation could have resulted from leakage of platelet serotonin into the PPP, since we did not obtain only the mid-portion of the supernatant after spinning down PRP. 6 However, contamination of serotonin, and the corresponding uncertainty in the measurement, was kept to a minimum, as shown by the very low plasma-free serotonin concentrations that we measured in SSRI users. Sampling patients in a fasting state would also decrease the variation; however, some patients had to travel a considerable distance, and therefore sampling in a fasting state was not an option.
In future studies, serial measurement of plasma-free serotonin concentrations and its metabolites after start of SSRIs might provide insight into the complex processes regarding regulation of serotonin concentrations in plasma in psychiatric patients and the side-effects these patients experience.
Supplemental Material
ACB880567 Supplemental Material1 - Supplemental material for Use of selective serotonin reuptake inhibitors is associated with very low plasma-free serotonin concentrations in humans
Supplemental material, ACB880567 Supplemental Material1 for Use of selective serotonin reuptake inhibitors is associated with very low plasma-free serotonin concentrations in humans by Marloes AM Peters, Martijn van Faassen, Wilhelmina HA de Jong, Grietje Bouma, Coby Meijer, Annemiek ME Walenkamp, Elisabeth GE de Vries, Sjoukje F Oosting, Henricus G Ruhé and Ido P Kema in Annals of Clinical Biochemistry
Supplemental Material
ACB880567 Supplemental Material2 - Supplemental material for Use of selective serotonin reuptake inhibitors is associated with very low plasma-free serotonin concentrations in humans
Supplemental material, ACB880567 Supplemental Material2 for Use of selective serotonin reuptake inhibitors is associated with very low plasma-free serotonin concentrations in humans by Marloes AM Peters, Martijn van Faassen, Wilhelmina HA de Jong, Grietje Bouma, Coby Meijer, Annemiek ME Walenkamp, Elisabeth GE de Vries, Sjoukje F Oosting, Henricus G Ruhé and Ido P Kema in Annals of Clinical Biochemistry
Supplemental Material
ACB880567 Supplemental Material3 - Supplemental material for Use of selective serotonin reuptake inhibitors is associated with very low plasma-free serotonin concentrations in humans
Supplemental material, ACB880567 Supplemental Material3 for Use of selective serotonin reuptake inhibitors is associated with very low plasma-free serotonin concentrations in humans by Marloes AM Peters, Martijn van Faassen, Wilhelmina HA de Jong, Grietje Bouma, Coby Meijer, Annemiek ME Walenkamp, Elisabeth GE de Vries, Sjoukje F Oosting, Henricus G Ruhé and Ido P Kema in Annals of Clinical Biochemistry
Footnotes
Acknowledgments
We would like to thank H. Adema, J. Krijnen, C.P. van der Ley, H.J.R. Velvis and M. Wilkens for the technical assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MAMP was supported by a fellowship of the Junior Scientific Masterclass of the University of Groningen. There was no involvement of the funding source in the study design; collection, analysis and interpretation of data; writing the report or in the decision to submit the article for publication.
Ethical approval
Medical Ethical Committee University Medical Center Groningen (# 2009/171).
Guarantor
IPK.
Contributorship
MAMP, MvF, WHAdJ, CM, AMEW, EGEdV, SFO, ER and IPK contributed to the study planning. MAMP and MvF performed the experiments. MAMP, GB, AMEW, EGEdV, HGR and SFO contributed to the inclusion of patients. MAMP, MvF, WHAdJ, CM, AMEW, EGEdV, SFO, HGR and IPK contributed to analysis and interpretation of the data. All authors contributed to writing the paper and approved the final version for publication.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
