Abstract
Background
The measurement of oestradiol is an integral component for the management of ovarian stimulation for in vitro fertilization. Automated immunoassays offer fast assay times and high throughput, with less sensitivity and specificity. The aim of this study is to optimize the oestradiol assay in patients undergoing ovarian stimulation for in vitro fertilization via comparison of oestradiol values obtained using two immunoassays compared with mass spectrometry.
Methods
Patients undergoing ovarian stimulation were prospectively recruited. Serum samples were analysed with ADVIA Centaur® CP Immunoassay, Abbott Architect i1000® immunoassay and AB Sciex 5500 liquid chromatography-tandem mass spectrometry (LC-MS/MS) systems. Per cent bias was determined for each system to report the average tendency of the values to be larger or smaller than the LC-MS/MS value. Linear regression of total follicular volume and oestradiol was computed.
Results
The ADVIA Centaur® CP assay had a positive bias of 20% compared with LC-MS/MS, while the Architect i1000® had a non-significant, negative bias of 0.3%. With regression fit, a clear, positive relationship was seen between follicular volume and oestradiol. The Architect i1000® assay had a greater correlation (R2 = 0.46) compared with Centaur® CP (R2 = 0.36), when oestradiol values were >1000 pg/mL (3670 pmol/L).
Conclusions
The Abbott Architect i1000® oestradiol assay exhibits greater agreement with LC-MS/MS and exhibited better correlation to follicular volume when oestradiol values are >1000 pg/mL (3670 pmol/L), prompting a change in the clinic’s oestradiol platform. Attention to assay quality assurance via LC-MS/MS can improve the oestradiol accuracy and permit more informed clinical decisions for improved patient outcomes.
Introduction
The measurement of the sex steroid, oestradiol, is an integral component for the diagnosis and management of numerous conditions such as hypogonadism, disorders of puberty, menopause and ovarian response during stimulation via assisted reproductive technology. 1 As various physiological states are associated with a marked range in oestradiol values extending from undetectable to >10,000 pg/mL (36,700 pmol/L), providing accurate oestradiol values at all ends of the concentration spectrum can prove challenging.1–3 This challenge is especially appreciable in ovarian stimulation cycles in which the oestradiol concentrations increase from low concentrations to supraphysiologic concentrations over the course of days.
Historically, metabolites of oestradiol were measured in urine as ketosteroids via colorimetric assays, which lacked sensitivity and specificity secondary to both endogenous and exogenous interferences. The introduction of radioimmunoassays using organic extraction methods prior to quantification provided improved the specificity and sensitivity but were subsequently replaced by automated immunoassays as testing demand for speed and high throughput increased. 2 While the automated immunoassays have provided faster throughput, this has come at the expense of decreased specificity, and the reporting of possible erroneous values due to cross-reactivity with other steroids or interferences.2,4–8 Mass spectrometry (MS)-based assays utilize both extraction and a chromatographic step to separate oestradiol from similar compounds in serum prior to the quantification, thus offering improved sensitivity and specificity. Although considered the gold standard, MS-based assays are associated with higher costs, slower turnaround time, require greater technical skills and are not routinely used in the reporting of oestradiol concentrations for in vitro fertilization (IVF) clinical decision-making.1,9
Oestradiol concentrations, as a measure of follicular recruitment and oocyte maturation, are utilized in the management of the IVF cycle. As a determinant of follicular recruitment and oocyte maturation, clinicians utilize serum oestradiol concentrations to adjust gonadotropin doses, initiate antagonist therapy, time the administration of a ‘trigger’ for oocyte maturation, optimize the number of oocytes reaching maturation simultaneously and determine the risk of ovarian hyperstimulation syndrome. Cumulatively, these decisions affect the pregnancy rates and patient outcomes. Ideally, oestradiol assays used in this clinical practice setting would offer both accurate and quick turnaround of results, with low, if any, cross-reactivity with other steroid compounds. While the automated immunoassays used in most assisted reproductive clinics offer fast assay times and high throughput, this is at the detriment to assay sensitivity and specificity. 10 Multiple case reports have highlighted the instances of erroneous oestradiol reports due to immunoassay interference; a range of compounds such as serum sex hormone-binding globulin, 4 antianimal antibodies,5,6 monoclonal gammopathy, 7 or even unidentified protein compounds 8 have been known to cross-react with oestradiol immunoassays, thus further complicating accurate quantification of oestradiol concentrations during ovarian stimulation.
With more than 14 different immunoassay platforms available for clinical use in determination of oestradiol concentrations, 11 greater emphasis has been placed on consistency and improved analytical performance including precision, linearity and limits of detection. In acknowledgement of this heterogeneity, a recent Endocrine Society Position Statement highlighted these weaknesses in immunoassays and therefore now requires studies reporting on sex steroid assays specifically use MS-based assays. 3 This further underscores that the current assay techniques may provide undesirable variance and offers impetus to standardization of oestradiol assays across clinical laboratories. Unfortunately, there are limited studies and research demonstrating successful techniques for assessing individual laboratory optimization of oestradiol assays in the IVF setting.8,12,13
The aim of this study is to determine the sensitivity and consistency of the oestradiol values during ovarian stimulation at the Family Fertility Center at Texas Children’s Hospital Pavilion for Women and to address the recommendation for MS-based assays for clinical decision-making during an IVF cycle. This collaborative partnership of the reproductive endocrinologists and the clinical pathology team used liquid chromatography tandem mass spectrometry (LC-MS/MS) as a gold standard to assess oestradiol concentrations using the current assay platform (ADVIA Centaur® CP Immunoassay system) and compared it with another available platform (Abbott Architect i1000® Immunoassay system). Furthermore, an attempt was made to evaluate these various assays to biological correlation of total volume of oestradiol-producing follicles as determined via ultrasonography.
Materials and methods
Instrumentation
The studies were performed using oestradiol assays on ADVIA Centaur® CP Immunoassay system (Siemens Corporation, Washington D.C., USA), Abbott Architect i1000® (Abbott Diagnostics, Abbott Park, IL, USA) system and AB Sciex 5500 QTRAP LC-MS/MS system (SCIEX, Framingham, MA, USA). The ADVIA Centaur® CP oestradiol assay is a monoclonal, competitive, chemiluminescent immunoassay that measures oestradiol with a turnaround time of 18 min. The Abbott Architect i1000® oestradiol assay is a monoclonal, delayed one-step immunoassay that uses chemiluminescent microparticle immunoassay (CMIA) technology and has a turnaround time of 28 min. The analytical measurement range for the Abbott Architect i1000® was 10–1000 pg/mL (36.7–3671 pmol/L), with a clinical reportable range of up to 15,000 pg/mL (55,050 pmol/L). If indicated, a 1:5 on board dilution was used and if greater, then a 1:10 manual dilution was employed. The analytical measurement range for the ADVIA Centaur® CP Immunoassay system was up to 2000 pg/mL (7340 pmol/L) with the same clinical reportable range. Quality controls were from each manufacturer and the manufacturer’s instructions were followed for quality control.
All samples for oestradiol by LC-MS/MS were sent to Quest Diagnostics, one of the facility’s reference laboratories for testing. The samples (300 µL each) were spiked with MassChrom® steroids internal standard mix (Chromsystems Instruments & Chemicals, Munich, Germany) and following liquid–liquid extraction using methyl-ter-butyl ether, samples were evaporated, reconstituted in mobile phase and injected on C18 columns on a high-performance liquid chromatography (HPLC) systems (Shimadzu, Tokyo, Japan), followed by spectral acquisition in the negative mode on a ABSciex QTRAP 5500 system. Mass transitions of oestradiol were 271/1/145 and 271.1/143, respectively. The limit of quantitation for oestradiol by LC-MS/MS was 0.02 ng/mL (73.4 pmol/L). The analytical measurement range extended up to 300 pg/mL (1101 pmol/L) and the clinical reportable range was up to 6000 pg/mL (22,020 pmol/L).
Precision
Precision studies on Centaur® CP system were performed using commercially available quality control materials. Intra-assay precision was conducted on two concentrations of control as 20 replicates. Inter-assay precision was determined using two concentrations of control as 20 replicates, three runs per day, over a period of 14 days. Precision studies on Abbott Architect i1000® system were carried out using commercial quality control materials at two concentrations.
Patients and methods
Patients included in our study were women undergoing ovarian stimulation for IVF at the Family Fertility Center. A total of 37 samples from 10 women were collected; demographic information such as age, anti-Mullerian hormone (AMH) concentration, and Society for Assisted Reproductive Technology (SART) diagnosis was recorded. For each stimulation, an individualized antagonist ovarian stimulation protocol was employed (Cetrorelix acetate, Cetrotide®, Freedom Fertility Pharmacy, Byfield, MA, USA, urofollitropin, Bravelle®, Ferring Pharmaceuticals, NJ, USA and menotropin, Menopur®, Ferring Pharmaceuticals, NJ, USA). Follicular number and size were obtained via transvaginal ultrasound during ovarian stimulation monitoring. Total follicular volume was calculated from the greatest follicular diameter in three dimensions using the mean diameter to produce radius of the follicle (volume = 4/3πr3; r = radius of follicle). Three-dimensional follicular volume was chosen due to a demonstrated stronger correlation with the number of mature oocytes retrieved compared with 2D follicular diameter. 14 The serum samples were collected in a red top serum tube via a peripheral blood sample taken within 1 h of the ultrasound measurement. When two or more follicles measured greater than 18 mm and in conjunction with oestradiol values, oocyte maturation was triggered with 10,000 units of Novarel® (chorionic gonadotropin, Ferring Pharmaceuticals, NJ, USA). All clinical decisions were made in conjunction with transvaginal ultrasound imaging and oestradiol concentrations as reported by the Centaur® CP, which was the standard platform used in our laboratory.
Analytical comparisons
Comparison studies were performed using oestradiol assays on ADVIA Centaur® CP, Abbott Architect i1000® and AB Sciex 5500 LC-MS/MS systems. Samples were analysed on ADVIA Centaur® CP system within 1–2 h of collection, and this value was used for clinical decision-making. The samples were then aliquoted and stored frozen (−80℃) until testing on the Abbott Architect i1000® and LC-MS/MS system. All samples were frozen for a period from several days up to one month, but then tested on all platforms from aliquoted serum thawed at the same time. A subset of samples that represented different stages of ovarian stimulation from lower to higher oestradiol concentrations (n = 15) were chosen to be analysed on the LC-MS/MS in addition to the Centaur® CP and Architect i1000® platforms. Per cent bias was calculated as the difference between the Centaur® CP or the Architect i1000® oestradiol value from the LC-MS/MS value divided by the LC-MS/MS value. Furthermore, coefficient correlation (r) was determined for the oestradiol to total follicular volume at all oestradiol values as well as in the subset of oestradiol values >1000 ng/mL (3670 pmol/L). The EP Evaluator Data Innovation (EE 10) program was used for the analysis of results for comparison studies. The significance between data obtained from the different platforms was assessed by ANOVA and post hoc tests on GraphPad Prizm.
Results
Intra-assay and inter-assay precision studies for the ADVIA Centaur® CP and Abbott Architect i1000® Estradiol assays.
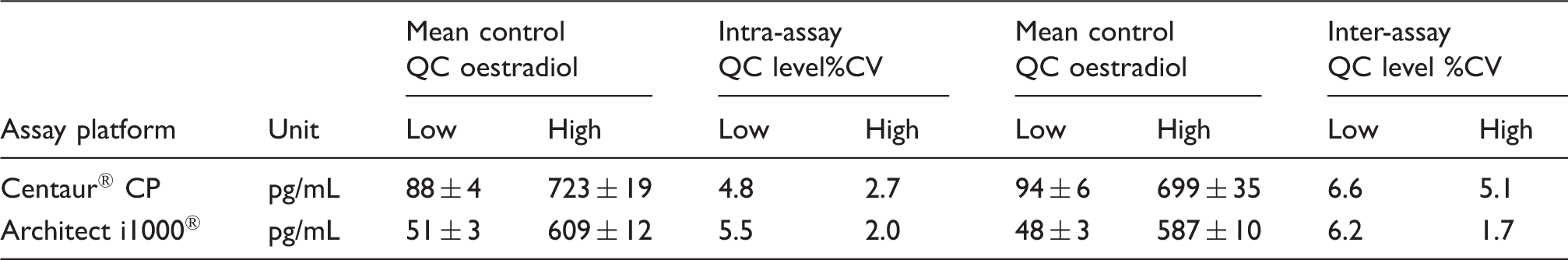
Note: Concentrations are provided as mean ± standard deviation. International System of Units (SI Units) conversion factor of 1 pg/mL = 3.67 pmol/L.
Oestradiol concentrations were obtained for 37 samples from 10 women undergoing ovarian stimulation within the same two-week period. For our patients, the mean age was 34.5 (standard deviation ± 2.9) years and the mean AMH value was 2.4 ng/mL (standard deviation ± 1.4). The SART diagnoses consisted of polycystic ovarian syndrome (n = 3), infertility due to male factor, such as low sperm counts or abnormal sperm function, (n = 3) and unexplained infertility (n = 4). A mean of 14.1 (standard deviation ± 7.26) oocytes were retrieved, of which an average of 12.6 (standard deviation ± 7.29) oocytes were mature. Comparison studies were carried out on all 37 patient samples using both the Centaur® CP and Architect i1000® analyzer and on 15 samples by all three assays (Centaur® CP and Architect i1000® and LC-MS/MS).
The results of comparison studies are shown in Figure 1. Comparison between Centaur® CP oestradiol values and Architect i1000® oestradiol (Figure 1(a)) revealed the Centaur® CP oestradiol had a mean positive bias of 20%, when all oestradiol values were analysed (n = 37). The mean bias of Centaur® CP oestradiol was 18% when comparison analysis was performed on samples with oestradiol concentrations lesser than 1000 pg/mL (3670 pmol/L), n = 16 (data not shown). As demonstrated in Figure 1(b), the bias was 20% when the analysis was performed on samples with higher concentrations of oestradiol (>1000 pg/mL [3670 pmol/L], n = 21).
(a) Comparison studies between ADVIA Centaur® CP and Abbott Architect i1000® oestradiol assays for all oestradiol concentrations (n = 37); (b) Comparison studies between ADVIA Centaur® CP oestradiol assay and Abbott Architect i1000 on the subset of oestradiol samples with concentrations >1000 pg/mL (n = 21). The dotted line indicates mean bias value. International System of Units (SI Units) conversion factor of 1 pg/mL = 3.67 pmol/L.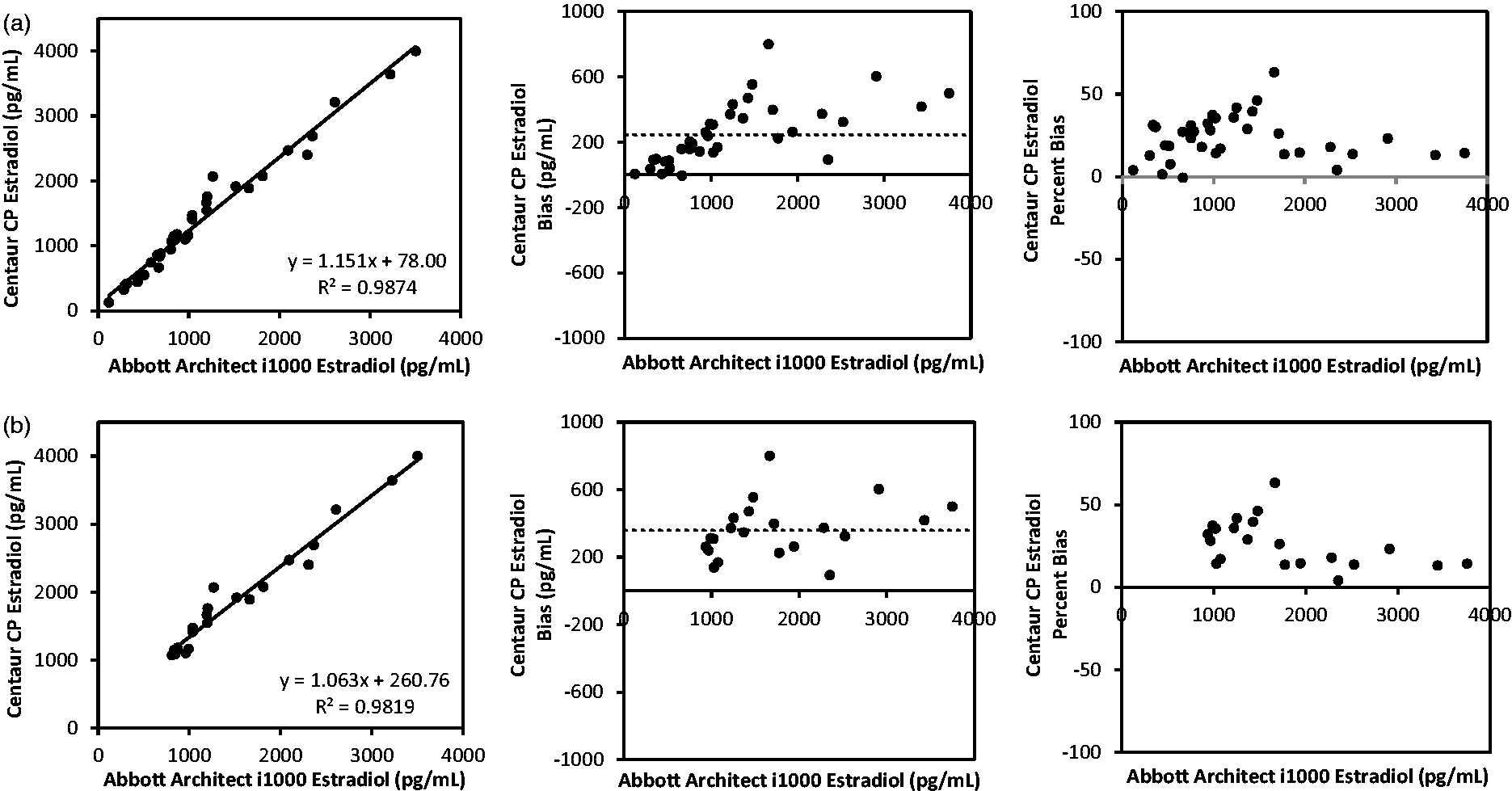
The second portion of the study included analysis of selected samples (n = 15, oestradiol ranging from 124 pg/mL to 3641 pg/mL [455.1 pmol/L to 13,362.5 pmol/L]) that were also analysed using the gold standard LC-MS/MS in order to determine per cent bias of both Centaur® CP and Architect i1000® oestradiol tests, as shown in Figure 2. These results demonstrated that the Centaur® CP oestradiol assay had a significant positive bias of 20% (Figure 2(a)), when compared with LC-MS/MS. When the Architect i1000® oestradiol assay was compared with LC-MS/MS, it revealed a non-significant negative bias of 0.3%, (Figure 2(b)). The per cent bias of each assay was consistent in both lower and higher ranges of oestradiol for both platforms. The Abbott Architect i1000® had a greater correlation coefficient (R2 = 0.9944) compared with the ADVIA Centaur® CP (R2 = 0.9843); while both correlation coefficients are high, the Abbott Architect i1000® oestradiol assay demonstrated a greater degree of correlation with the LC-MS/MS.
(a) Comparison studies between ADVIA Centaur® CP and Tandem Mass Spectrometer (LC-MS/MS) oestradiol assays (n = 15); correlation coefficient R = 0.9843 and bias of 20%. (b) Comparison studies between Abbott Architect i1000® and Tandem Mass Spectrometer (LC-MS/MS) oestradiol assays (n = 15); correlation coefficient R = 0.9944 and bias of −0.3%. The dotted line indicates mean bias value. International System of Units (SI Units) conversion factor of 1 pg/mL = 3.67 pmol/L.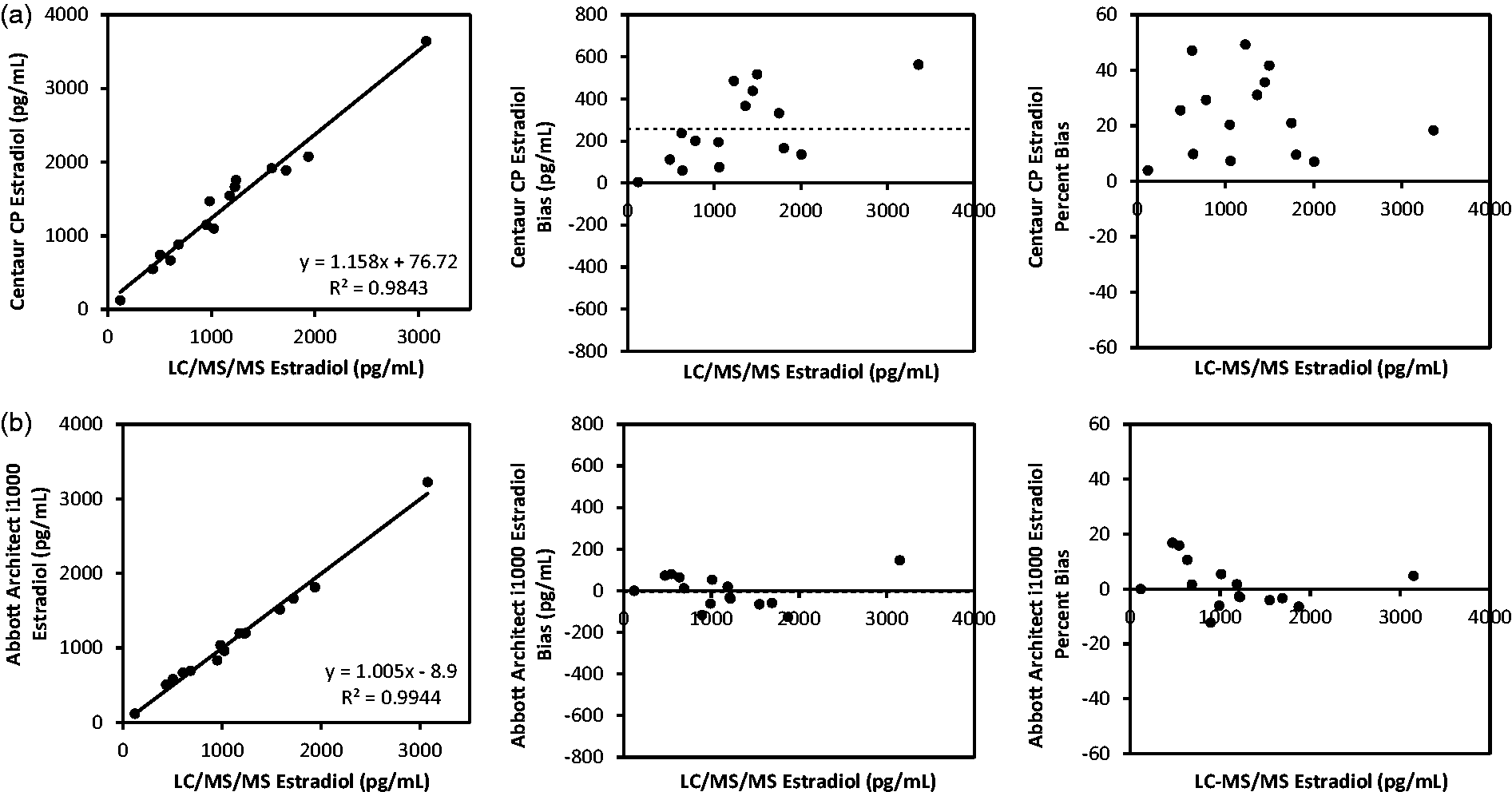
Lastly, the biological correlation between oestradiol concentration and total follicular volume for both platforms was determined via regression fit (RF) comparison to total follicular volume ratio. On the day that the serum was obtained for oestradiol concentration, the total volume of the follicles on each ovary was determined. The Architect i1000® (Figure 3(a)) and Centaur® CP (Figure 3(b)) correlation coefficients were nearly identical when the entire range of oestradiol concentrations were included in the analysis (R2 = 0.61 and 0.62, respectively). However, the RF analysis revealed that at oestradiol concentrations >1000 pg/mL, the Architect i1000® oestradiol assay had a greater correlation to the total follicular volume (R2 = 0.46) compared with Centaur® CP (R2 = 0.36) (Figure 3(c) and (d), respectively).
Regression fit analysis for the (a) Centaur® CP oestradiol immunoassay concentration and (b) Abbott Architect i1000® oestradiol immunoassay concentration compared with the total follicular volume including patient samples (n = 37); (c) Centaur® CP oestradiol immunoassay and (d) Abbott Architect i1000® oestradiol immunoassay concentration compared with the total follicular volume including patient samples with oestradiol concentrations >1000 pg/mL (n = 21). International System of Units (SI Units) conversion factor of 1 pg/mL = 3.67 pmol/L.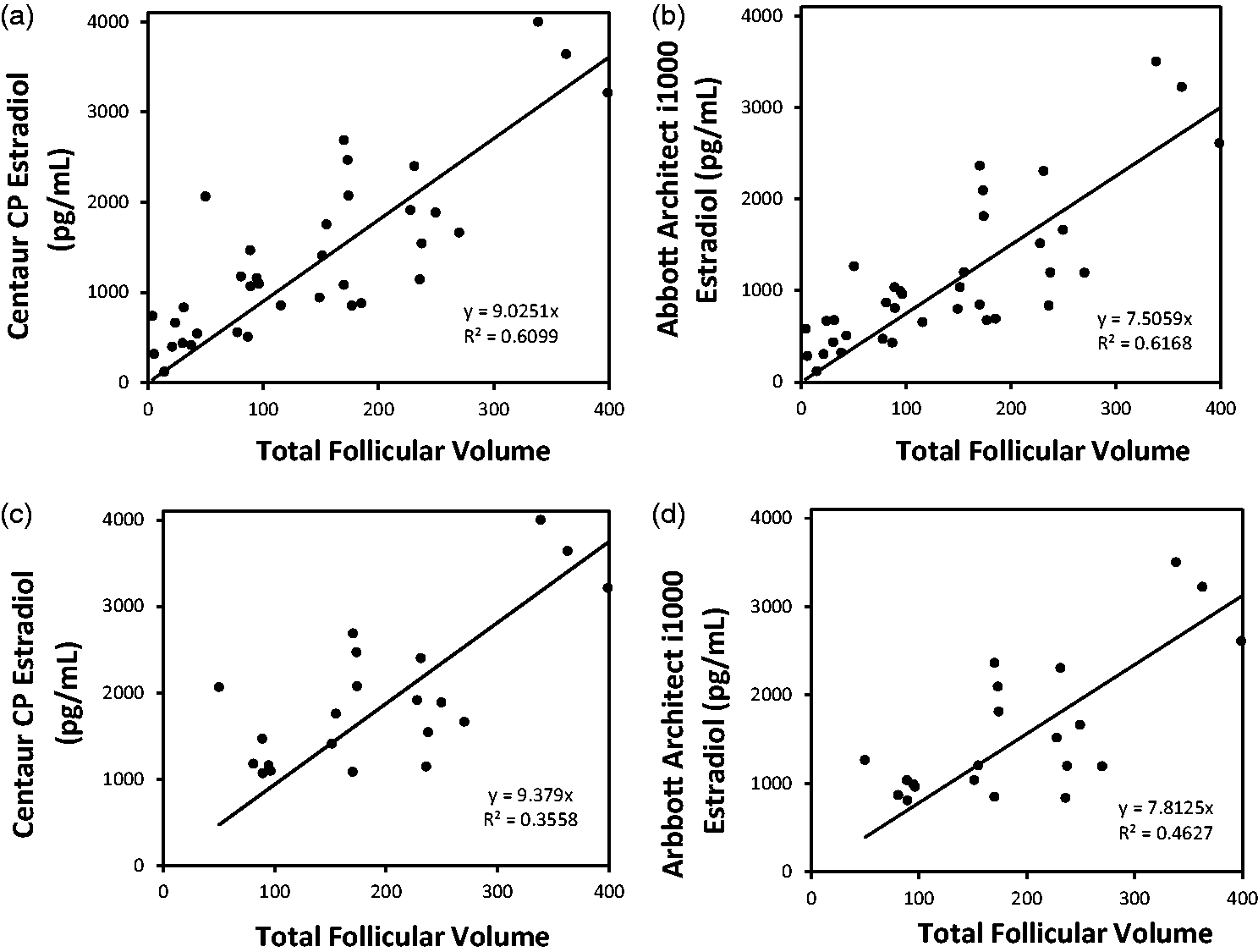
Discussion
Summary of findings
This study highlights the importance of evaluating the oestradiol assays used in the IVF setting to monitor the patients by collaboration of the IVF team with the clinical pathology team. Comparison studies showed that the Architect i1000® oestradiol assay exhibits an excellent agreement with LC-MS/MS at all ranges of oestradiol studies which is consistent with results of similar utility and validation studies.15–17 Similarly, our findings concur with the evaluation studies using ADVIA Centaur® CP system and comparison study with LC/MS/MS in which a 19.5% bias was observed. 18 Additionally, it is important to highlight that although the precision results in which %CV for inter-assay and intra-assay studies are within the normally accepted range, these values are derived from standards provided by the manufacturer; therefore, a reassuring %CV value may not correspond to accuracy when compared with LC-MS/MS.
As well as determining the superior concordance of Architect i1000® oestradiol assay with an LC-MS/MS, we further demonstrated that when the oestradiol concentrations were compared with the total follicular volume, a better correlation was seen with Architect i1000® compared with the Centaur® CP assay when oestradiol concentrations were >1000 pg/mL (>3670 pmol/L). The variation of oestradiol concentrations compared with follicular volume measurements may reflect that only follicles of >10 mm were used to calculate volume, and as such, the smaller, unmeasured follicles may be contributing to oestradiol. Additionally, determining the total volume by using greatest diameter in three dimensions may not accurately reflect the volume of follicles that have complex shapes and are not just spherical. Oestradiol assay accuracy and consistency at values >1000 pg/mL (>3670 pmol/L) are crucial, as clinicians often make critical decisions such as the administration of human chorionic gonadotropin (hCG) trigger injection at this oestradiol range. The administration of hCG trigger prematurely or a delay of trigger based on an inaccurately reported elevated oestradiol concentrations can have cumulative effects on patient outcomes and a clinic’s success rates. Using an assay that provides a greater than 20% overestimation bias of oestradiol concentrations, a reproductive programme may change its determinants in defining medication protocols to decrease gonadotropins and/or routinely administer hCG prematurely. Henceforth, a programme may observe changes ranging from oocyte recruitment patterns to embryo formation and pregnancy outcomes when oestradiol values shown significant deviation from LC-MS/MS. Subsequently, these cumulative clinical decisions may adversely affect the outcomes and pregnancy rates. Clinicians should be aware that changes in assay platforms by an endocrine laboratory might go unreported to an IVF programme, and thus, might contribute to unknown causations for alterations in programme outcomes.
Discussion of clinical use
An individual clinic’s ability to accurately determine oestradiol concentrations is important during monitoring of ovarian stimulation, response to gonadotropins and patient outcomes during in vitro fertilization cycles. Given the wide range of serum oestradiol concentrations observed during an IVF stimulation cycle and the desire for quick results and large volume throughput, obtaining accurate values continues to prove challenging. Historically, clinicians rely on serum oestradiol values in combination with follicular measurements via ultrasonography for ovarian stimulation management, trigger for oocyte maturation and providing prognostic information for IVF outcomes.19–21 More so, programmes have created algorithms relying on information such as the ratio of oestradiol to number of follicles on day of trigger22,23 and the oestradiol to oocyte ratio to determine timing for oocyte maturation and in reporting oocyte recovery rates and clinical pregnancy rates.22,24,25
As clinicians combine the laboratory oestradiol values and ultrasound imaging for patient care and clinical decision-making, ensuring the accuracy and consistency of this data remains paramount. 26 LC-MS/MS and gas chromatography mass spectrometry (GC/MS) techniques have better specificity and sensitivity compared with immunoassays and are the recommended modality of measurement27–30; however, the short analysis time and high throughput make immunoassays desirable for clinical use. Notably, LC-MS/MS is considered a gold standard and the laboratory participates in College of American Pathologists (CAP) proficiency testing. It is thus critical that the clinicians are aware of possible variations in assay accuracy when relying on immunoassays.
This study further highlights the need for clinical awareness in evaluation of oestradiol assays, supporting the recent Endocrine Society statement encouraging MS for reliable oestradiol measurement. 1 From this co-operative collaboration of the IVF and Clinical Pathology teams, we conclude that the Abbott Architect i1000® oestradiol assay displayed a good precision and better agreement with LC-MS/MS, in addition to providing a better correlation with biological measurements of total follicular volume. As a result of these findings, the platform for oestradiol assays was changed from currently used ADVIA Centaur® CP to Abbott Architect i1000®, in order to provide more accurate and consistent oestradiol values for enhancement of patient care. For clinicians, acute awareness of assay quality assurance via MS can improve the oestradiol accuracy and provide better informed clinical decisions for improved patient outcomes. Finally, these findings are reflective of the importance of a close, working relationship between clinicians and pathologists
Limitation and future studies
Although this study only addresses the process and outcomes of a single IVF center working to optimized the oestradiol assay, this work highlights the importance of acute clinical awareness and knowledge of these assays and various platforms available. Even though the research was conducted at an individual clinic with a small number of samples, the process by which collaboration, prospective serum collection and comparison of immunoassays to the gold standard of LC-MS/MS is applicable to a wide range of practice settings. Future studies that include the changes in clinical outcomes after the institution of a new oestradiol assay will be more forthcoming in providing information on the effects of the oestradiol assay optimization on patient care and outcomes.
Footnotes
Acknowledgements
The expertise and collaboration with the Baylor Family Fertility Center and Texas Childrens Clinical Pathology Laboratory are greatly appreciated.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by ChingNan Ou fellowship in Clinical Chemistry.
Ethical approval
The study was approved by the Baylor College of Medicine Institutional Review Board (protocol H-39094).
Guarantor
SD.
Contributorship
Conceived and designed the experiments: MP, NA, WG, PZ and SD. Performed the experiments: MP, NA. Analysed the data: MP, NA, SD. Contributed to the writing of the manuscript: MP, NA, WG, PZ and SD.
