Abstract
Background
Measurement of selenium in serum is an important clinical biomarker of nutritional status. The presence of gadolinium (Gd) in samples following administration of the contrast agents used for magnetic resonance imaging (MRI) results in a significant positive bias when using quadrupole inductively coupled plasma mass spectrometry (Q-ICP-MS).
Methods
Three instrumental set-ups were assessed: standard mode with no collision gas and collision cell mode with either a hydrogen:helium mixture or hydrogen. The effect of Gd on the selenium (Se) signal was assessed using external quality assurance (EQA) specimens and internal quality control (IQC) materials, both unspiked and spiked with Gd. Serum previously shown to contain high concentrations of Gd-containing contrast agents were also analysed.
Results
Recoveries of Se in the spiked compared to the unspiked samples were: between 500% and 1300% using standard mode; 100% and 29,000% using collision cell mode with hydrogen:helium mixture; and between 99% and 103% using hydrogen. The use of H2 in the collision cell provided accurate results, indicating that the charge exchange reaction (CER) of Gd2+ with H2 removes this interference. Analysis of patient serum known to contain the Gd contrast agent using the method gave results within the selenium reference range (adults 0.89–1.65 µmol/L). The presence of Gd, as low as 0.2 mg/L, in serum samples causes a positive interference on the measurement of Se by ICP-MS.
Conclusions
Using a CER mode with pure H2 in the collision cell it was possible to fully remove the interference due to Gd2+ from the signal for Se.
Introduction
Selenium is an important nutritional trace element 1 which is routinely measured in a clinical setting. It is incorporated into a number of enzymes with metabolic functions including the synthesis of thyroid hormones and the protection of cell membranes against reactive oxygen species. Selenium also guards against development of some cancers and a reduced incidence of prostate cancer in men taking dietary supplements has been reported. 2 Deficiency of Se may be seen if there is inadequate intake in the diet, if there is poor intestinal absorption and in patients who are on prolonged intravenous nutrition when supplementation of Se is insufficient. Selenium is also toxic if there is excessive exposure, which can occur where the soil, and hence crops, contain naturally high concentrations of this element. More usually, toxicity is associated with occupational exposure or in incidents of deliberate poisoning.
The determination of Se to establish nutritional status is an important measurement in clinical science. Diagnosis of deficiency or toxicity is made by measuring the Se concentration in serum or whole blood. This is usually carried out using inductively coupled plasma mass spectrometry (ICP-MS), which provides suitable limits-of-detection and short- and long-term precision. However, Se is a difficult element to determine by ICP-MS for a number of reasons: it has a high ionization potential (IP), so only a small proportion of Se atoms in the plasma are ionized; the signal for Se is spread over a number of isotopes (m/z 74, 76, 77, 78, 80, 82); and all the analytically useful isotopes, including the most abundant m/z 80, have major argon interferences, which precludes their measurement directly. Removal of these interferences can be achieved by using cold plasma conditions, high resolution magnetic sector instruments, or collision/reaction cells.
In our experience, the most appropriate Se isotope to measure is m/z 78 and the polyatomic interferences at this m/z, which include the argon–argon dimer (Ar–Ar); an argon–calcium polyatomic (Ar–Ca); and the oxides of nickel (Ni–O), can be removed using the conventional collision cell set-up for this instrument, which involves the collision gas mixture of hydrogen in helium (7% vol/vol [v/v] H2) and kinetic energy discrimination (KED). This works by separating polyatomic ion interferences from the isotope ion with which they interfere on the basis of a change in the kinetic energy of the ions resulting from collisions between the ions and the gas in the collision cell of the instrument. Polyatomic ions have larger collision cross-sectional areas than isotope ions of the same mass and so undergo a greater number of collisions as they transit the cell.The result is that at the cell exit, the lower energy polyatomic species can be readily filtered out by applying an appropriate voltage difference between the collision cell and the analyser quadrupole. The isotopes of interest also lose kinetic energy as a result of the collisions they experience, thereby reducing the sensitivity, but overall the dramatic reduction in the polyatomic interference signal results in a much greater signal to background ratio for the interfered analyte, and correspondingly more accurate results with better detection limits. With reactive gases, such as H2, in the cell, other interference removal processes based on charge exchange reactions (CERs) and gas phase chemical reactions also occur which can be beneficial for removing certain interferences, particularly monoatomic ion interferences such as doubly charged ions, as demonstrated in this work.
Gadolinium can interfere with the measurement of Se because it readily forms doubly charged ions with the same m/z value as the Se isotopes such that the Gd2+ ions of isotopes m/z 152, 154, 156, 158, and 160 interfere with the Se isotopes at m/z 76, 77, 78, and 80, respectively. Although the Se isotope at m/z 74 is essentially free from a Gd2+ interference it cannot be used because of its low abundance (0.89%) which under cell conditions makes the signal too low. The formation of doubly charged ions is not a problem in most situations as the manufacturer’s specification for running the plasma for routine use includes setting an upper limit for their formation. This is usually established using the major isotope of an easily doubly-ionized element, such as cerium or barium, so that there is no more than 2–3% of the doubly-ionized ion. It is also unusual to come across Gd in most clinical situations as it is not biologically important and being a rare earth element it is not encountered at significant concentrations in the diet. However, it can be a problem in situations where MRI studies are performed, as Gd is present in a number of the commonly used contrast agents. 3 We have previously described 4 a clinical situation whereby the interference from Gd led to a potential for the mis-interpretation of the nutritional status of Se in patients who had undergone an MRI scan as part of their postoperative gastrointestinal assessment. In this work, we investigated the removal of the Gd2+ interference using collision cell quadrupole ICP-MS (Q-ICP-MS) in the determination of the Se nutritional status of hospital patients.
Materials and methods
Reagents and materials
Single element 1000 mg/L stock solutions traceable to the United States National Institute of Standards and Technology (NIST) were used (Certipur, Merck, UK). Selenium calibration standards containing In as the internal standard were prepared in the range 0.5–4.0 µmol/L and Gd was used to prepare the spiked serum samples. All solutions were prepared in dilute (0.5% v/v) nitric acid (Primarplus, Trace Element Grade, Fisher, UK) using reverse osmosis deionised water (>18 Mohms resistivity). The multielemental tune solution (Multielement Tune A, Thermo Fisher Scientific, Hemel Hempstead, UK) contained Ba, Be, Ce, Co, In, Li, Pb, and U at a concentration of 10 µg/L in nitric acid (2% v/v).
Internal quality control (IQC) and external quality assurance (EQA) materials with assigned values for Se content were used to demonstrate the accuracy of the method. The UTAK BI-Level Trace Element Serum Toxicology Controls (UTAK Laboratories Inc., Valencia, Spain) which are prepared from normal human serum specimens and are designed to generate data that evaluates the results of a test method over the normal (UTAK normal) and elevated range (UTAK high) were used. Serum specimens from the UKNEQAS Trace Element Quality Assessment Scheme (TEQAS) (University of Surrey, Guildford, UK) with assigned values for Se were also used. Calibration standards were run as samples to check the accuracy of the calibration.
Instrumental conditions
All the data were collected using a ThermoElemental X2 series Q-ICP-MS (Thermo Fisher Scientific, Hemel Hempstead, UK) equipped with a hexapole collision cell. The multielemental tune solution was used to tune the instrument for sensitivity, formation of oxide and doubly charged ions, mass accuracy and resolution, so as to achieve the instrument manufacturer’s specifications.
Instrument conditions used to determine Se in serum with the Thermo Scientific X2 series ICP-MS.

Results and discussion
Details of the Se concentrations in each material and the accuracy of the standard mode method using a collision cell filled with 7% H2 in He, and KED. a
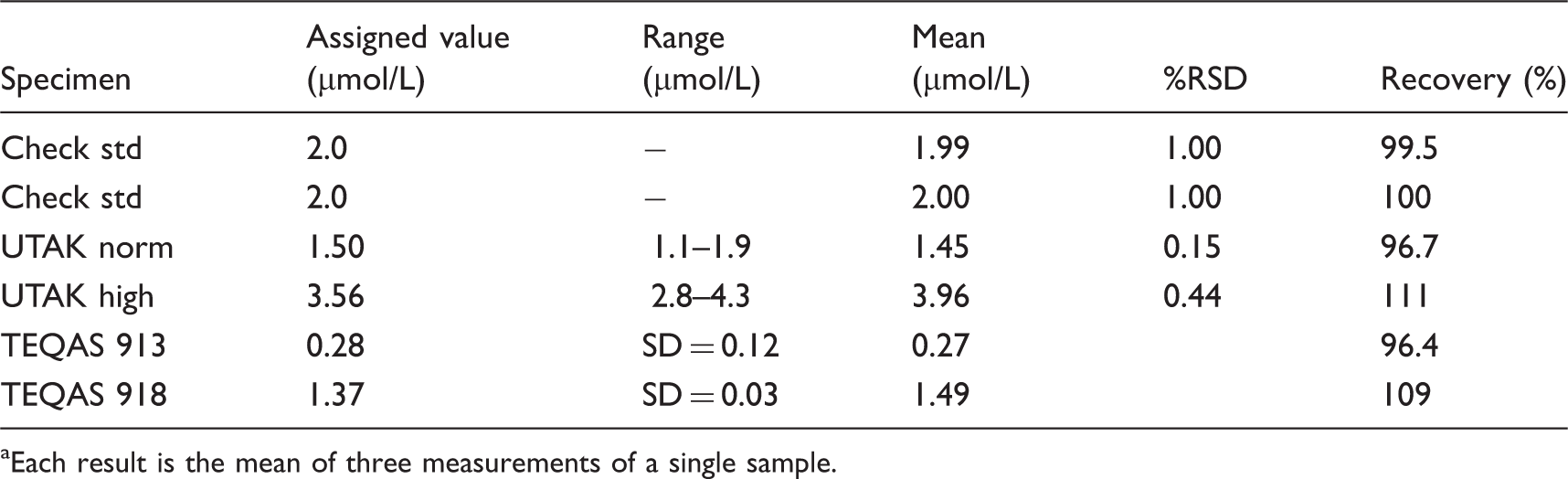
Each result is the mean of three measurements of a single sample.
Investigation of the contribution of Gd2+ to the determined Se concentration in a human serum sample from a normal patient not undergoing MRI imaging. a
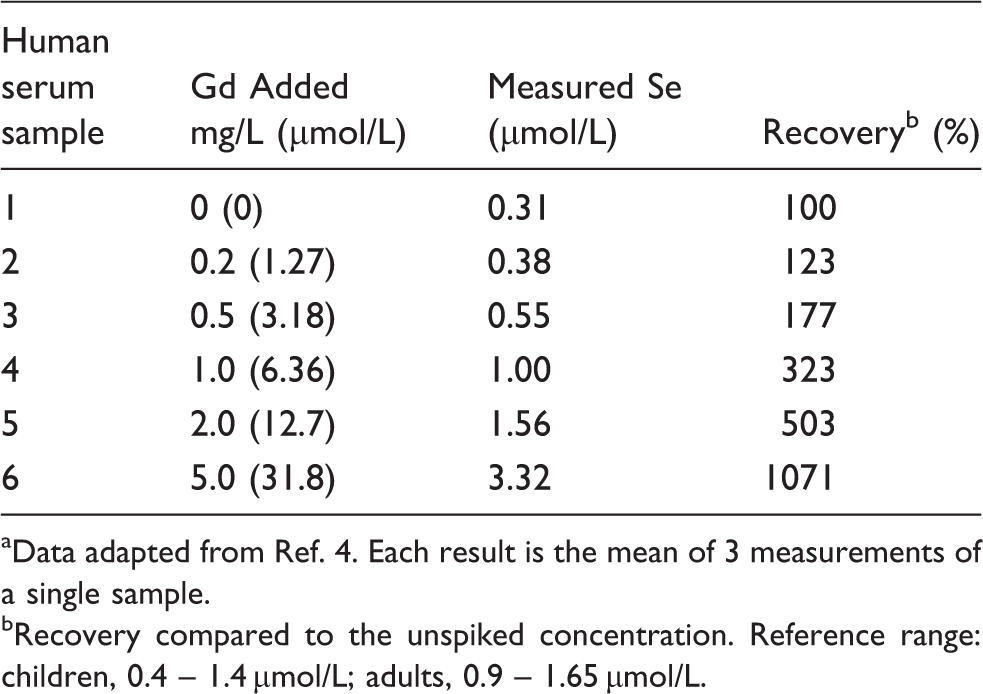
Data adapted from Ref. 4. Each result is the mean of 3 measurements of a single sample.
Recovery compared to the unspiked concentration. Reference range: children, 0.4 – 1.4 µmol/L; adults, 0.9 – 1.65 µmol/L.
Results for the concentration of Se measured in the spiked compared to the unspiked samples. a
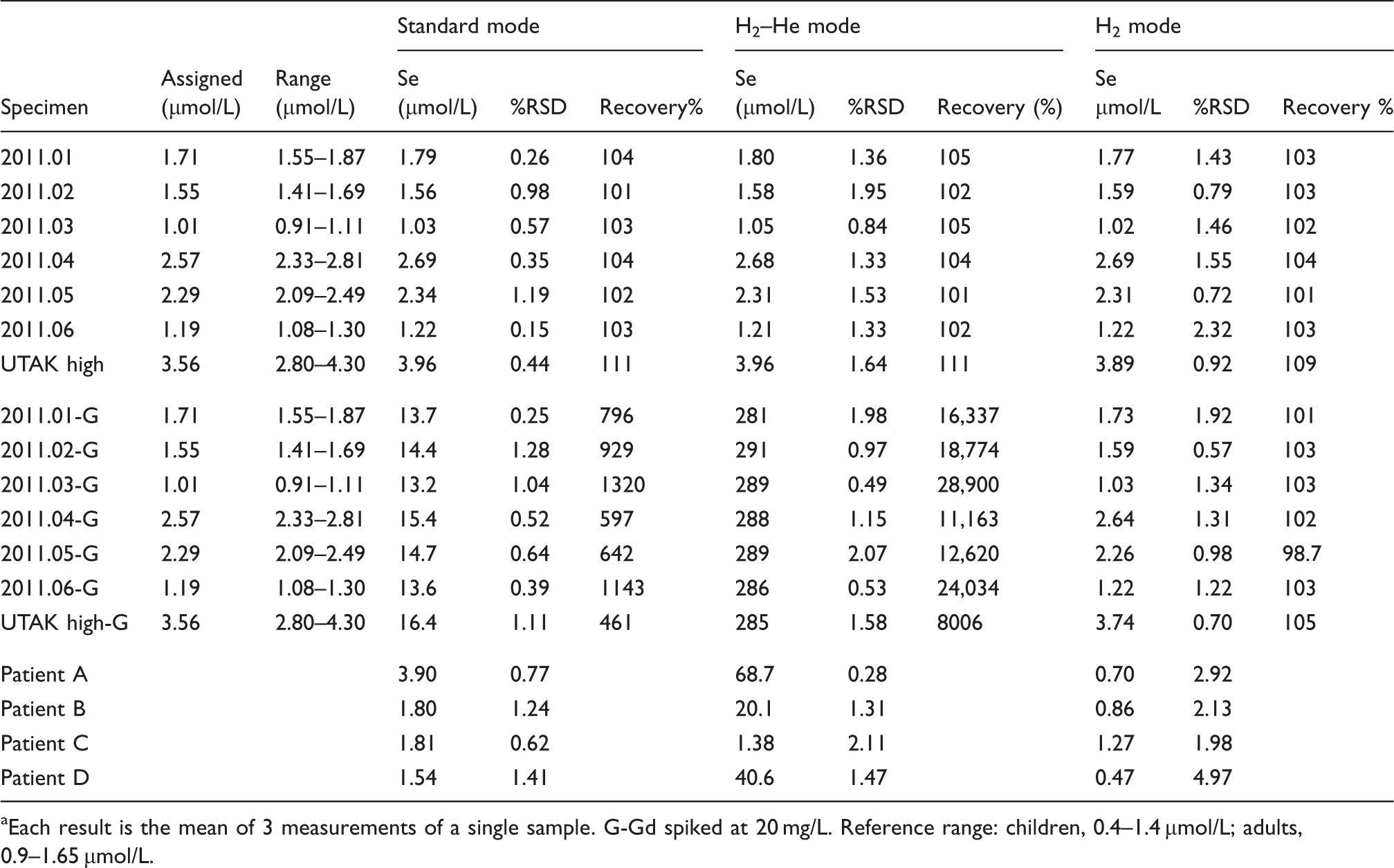
Each result is the mean of 3 measurements of a single sample. G-Gd spiked at 20 mg/L. Reference range: children, 0.4–1.4 µmol/L; adults, 0.9–1.65 µmol/L.
Characterization of the Gd doubly charged interference
The clearance of Gd following its use as a contrast agent has been investigated in healthy subjects 5 and in patients with impaired renal 6 or liver 7 function. Following an intravenous injection of 2 mg Gd per kg body weight to healthy volunteers peak plasma concentrations immediately after dosing were approximately 1570 mg/L (10 mmol/L). 5 Levels were followed for 12 h and showed a linear decay to 15.7 mg/L (100 µmol/L). Measurements were not continued beyond this time and it is not known for how long Gd remains detectable within the circulation.
Interference removal
Multiple charged ions are found at fractions of their nominal mass (m/z), such that the observed mass is equivalent to the nominal mass divided by the charge on the ion; as highlighted in the current situation the Gd2+ ions of isotopes m/z 152, 154, 156, 158, and 160 interfere with the Se isotopes at m/z 76, 77, 78, and 80, respectively. These doubly-charged ions have a smaller formation level probability because the second and higher IPs are larger than the first and are not generally a significant problem unless the source of the multiple charged ion is a relatively easily ionized matrix element of very high concentration. Mass resolution between 2000 and 10,000 is required to overcome these interferences, which is well within reach of magnetic sector ICP-MS. When a magnetic sector instrumentation is not available, then quadrupole instruments with a collision or reaction cell can be used but will be vulnerable to these interferences.
As discussed above, for interference removal purposes, quadrupole instruments are most frequently operated with inert gases in the collision cell with application of KED, but in the case of doubly charged ion interference, this approach is not effective. However, by operating the instrument with a reactive gas such as H2, charge exchange and chemical reactions predominate and these can be extremely effective for interference removal in certain cases. In the case of Gd2+ removal on the isotopes of Se, H2 is an effective gas as it has a lower IP than the second IP of Gd and so can be readily ionized by Gd2+. The result is conversion of Gd2+ to Gd+ (and by chemical reaction, GdH+ as well) and hence removal of the Gd2+ interference from the mass spectrum. For Se, H2 has the added benefit of being highly effective in removing Ar2+ interference and also providing higher sensitivity for Se compared to KED with He or H2/He cell gas mixtures.
The current clinical situation whereby very high concentrations of Gd are present in the serum due to administration of a Gd-containing contrast agent is a scenario where an easily ionized element would be present at a high concentration. It is clear from the results in Table 3 that the presence of 2 mg/L Gd in the serum sample is sufficient to increase the apparent Se concentration to the upper end of the reference range for Se in the adult UK population. With concentrations of 15.7 mg/L present 12 h after administration of the contrast agent, 5 there is clearly a very real potential for the measurement of Se in these patients to appear grossly high due to the presence of Gd2+ in the ICP. The results obtained with the specimens with and without addition of 20 mg/L Gd are shown in Table 4.
All three modes, i.e. standard mode with no cell gas present, KED mode with H2 in He present, and CER mode with pure H2 present, gave accurate results for the unspiked samples. The Se concentrations for all samples were within their assigned or reference ranges. The concentration of Se in the samples covered the range 1.01–3.56 µmol/L, which includes the human reference range for Se. The data show recoveries between 101% and 111%. The precision for these results was also acceptable with %RSD values in the range 0.15–2.32. In comparison, when run using standard mode, the spiked samples gave Se values between 13.2 and 16.4 µmol/L, when the assigned values for these samples ranged between 1.01 and 3.56 µmol/L, indicating the enhancement in the Se signal due to the presence of the Gd2+ ion at m/z 78. A considerably different situation was apparent using the KED mode with a cell containing H2 in He. The apparent Se concentrations in these seven specimens had a mean of 287 µmol/L (SD 3.07, %RSD 1.07), which indicated that the signal for Se was completely masked by the doubly-charged ion signal. The third mode tested using CER and pure H2 gave values in agreement with the assigned or certificate value for Se, despite the presence of the high concentration of Gd. Recoveries were in the range 98.7–105%. It is clear that the CER conditions using pure H2 in the reaction cell overcomes the influence of the Gd2+ ions on the measurement of Se and this mode of operating the ICP-MS should be employed with clinical samples that might contain Gd from Gd-containing MRI contrast reagent.
Patient sample analysis
Four patients’ samples that had previously shown elevated concentrations of Se above the adult reference range and been investigated and shown to contain Gd by demonstrating the presence of the characteristic Gd isotopic pattern were also analysed using the three cell modes (Table 4). Once again only the CER mode with pure H2 in the collision cell provided accurate results, which where all within the adult reference range for Se. The other modes tested gave much higher results, some of which were not consistent with the reference range and indicated excessively high concentrations, particularly using the KED mode with H2 in He as the collision gas.
Conclusion
We have previously demonstrated that Gd, present in human serum samples following the use of Gd-contrast agents for MRI, causes a positive interference on the measurement of Se by ICP-MS. Interference was observed with Gd concentrations as low as 0.2 mg/L. By using a CER mode with pure H2 in the collision cell it was possible to fully remove the interference due to Gd2+ from the signal for Se. In situations where this is not possible, e.g. where the use of H2 is not permitted on safety grounds, a new sample should be requested from the patient after the gadolinium-containing contrast reagent has been eliminated from the body.
Footnotes
Declaration of conflicting interest
SN is an employee of Thermo Fisher Scientific.
Funding
The research was funded internally as part of the instrument validation.
Ethical approval
Not required.
Guarantor
CFH.
Contributorship
CFH and SN conceived the study. AW was involved in protocol development as were CFH and SN. AT researched the literature. All authors contributed to the manuscript preparation, review and approved the final version.
