Abstract
Background
Major surgery predisposes to endothelial glycocalyx injury. Endothelial glycocalyx injury associates with cardiac morbidity, including spontaneous myocardial infarction. However, the relation between endothelial glycocalyx injury and the development of perioperative myocardial infarction remains unknown.
Methods
Fifteen perioperative myocardial infarction patients and 60 propensity-matched controls were investigated in this prospective study. The diagnosis of perioperative myocardial infarction was based on repeated cardiac troponin T measurements, electrocardiographs and recordings of ischaemic signs and symptoms. We measured endothelial glycocalyx markers – soluble thrombomodulin, syndecan-1 and vascular adhesion protein 1 – and an inflammatory marker, namely interleukin-6, preoperatively and 6 h and 24 h postoperatively. We calculated the areas under the receiver operating characteristics curves (AUCs) to compare the performances of the different markers in predicting perioperative myocardial infarction. The highest value of each marker was used in the analysis.
Results
The interleukin-6 concentrations of perioperative myocardial infarction patients were significantly higher preoperatively and 6 and 24 h postoperatively (P = 0.002, P = 0.002 and P = 0.001, respectively). The AUCs (95% confidence intervals) for the detection of perioperative myocardial infarction were 0.51 (0.34–0.69) for soluble thrombomodulin, 0.63 (0.47–0.79) for syndecan-1, 0.54 (0.37–0.70) for vascular adhesion protein 1 and 0.69 (0.54–0.85) for interleukin-6.
Conclusions
Systemic inflammation, reflected by interleukin-6, associates with cardiac troponin T release and perioperative myocardial infarction. Circulating interleukin-6 demonstrated some potential to predict perioperative myocardial infarction, whereas endothelial glycocalyx markers did not.
Introduction
Major surgery causes sympathoadrenal activation and systemic inflammation, exposing the organ system to ischaemia-reperfusion (I/R) injury. In addition, extensive perioperative fluid management may contribute to endothelial injury. The vascular endothelium represents the main organ affected by systemic inflammation, I/R injury and hypervolemia, leading to endothelial activation and endothelial glycocalyx (EG) injury.1–5 After early endothelial distress, circulating EG components (syndecan-1) and endothelial cell integral membrane proteins (soluble thrombomodulin [sTM] and vascular adhesive protein [VAP-1]) are released into plasma and have been suggested to be sensitive markers of endothelial damage and potentially imminent organ injury. 6 Pro-inflammatory cytokine IL-6 is central in acute-phase reactions caused by infection or tissue trauma. 7
The important role of EG injury has been established in numerous critical illnesses, including sepsis and acute kidney injury (AKI), and it is associated with increased mortality.8–10 Furthermore, EG injury associates with cardiovascular morbidity, including acute coronary syndrome (ACS).11–13 However, no data exist on the association of EG injury with perioperative myocardial infarction (PMI).
The objective of this study was to investigate the relation of EG injury and systemic inflammation with perioperative myocardial injury/infarction, evaluated by serial measurements of EG markers and IL-6. We hypothesized that acute inflammation and EG injury are associated with perioperative cardiac troponin T (cTnT) release and PMI.
Methods
Study design
This was a laboratory substudy of a prospective, observational single-centre study investigating the incidence of major perioperative cardiac events in patients undergoing systematic perioperative ischaemia screening, conducted at the Meilahti Hospital, Helsinki, Finland, in 2014. 14 Ethical approval for the study (no. 11/13/03/02/2014) was provided by the Ethics Committee of the Department of Surgery at Helsinki University Hospital, Helsinki, Finland, on 30 January 2014. All patients or their next of kin signed a written informed consent before enrolment. In brief, we included 385 consecutive patients, aged 50 years or older, undergoing non-cardiac (vascular, thoracic or gastrointestinal) surgery between 3 April and 19 June 2014. Organ transplantations and hepatic surgery were excluded. In this substudy, we included all PMI patients with available baseline and follow-up blood samples (n = 15), as well as four propensity-matched controls for each PMI patient (n = 60). The variables used for matching were age, sex, main medical history, medications and intraoperative data.
Patient management and data collection
The procedure for obtaining the patients’ medical history, demographics, medication, physiological and surgical data has been explained in detail previously. 14 A blood sample for cTnT and an electrocardiograph (ECG) were obtained five times: preoperatively, 6 h from the conclusion of the operation, and on the first, second and third postoperative morning, or until discharge. Together with cTnT, we took 5 mL of EDTA plasma for a future analysis of endothelial EG markers and IL-6. Possible ischaemic signs and symptoms were obtained from the patients’ charts and medical records filled in daily by the treating clinical staff. Based on repeated cTnT measurements, ECGs and clinical observations, a cardiologist (JV) blinded to the EG marker and IL-6 analyses confirmed all PMI diagnoses. The diagnosis of PMI was defined as a rise and/or a decline in cTnT, with at least one value above the upper reference limit (14 ng/L), and either ischaemic ECG changes or other ischaemic features present (symptoms of cardiac ischaemia, cardiac imaging evidence of myocardial infarction, autopsy findings of acute or healing myocardial infarction), as recommended. 15
Blood samples and enzyme-linked immunosorbent assay analyses
Our focus was on perioperative EG injury, which is why only blood samples taken preoperatively and 6 and 24 h after surgery were analysed. cTnT concentrations were analysed immediately after sampling from a vein or an arterial catheter. We used a high-sensitivity fifth-generation troponin T assay (electrochemiluminescense immunoassay [ECLIA]; Roche Diagnostics International Ltd, Rotkreuz, Switzerland) for the analyses. The limit of detection (LoD), analytical range, 10% coefficient of variation level and 99th percentile cut-off point were 5 ng/L, 5–10,000 ng/L, 13 ng/L and 14 ng/L, respectively. Plasma samples were centrifuged at 22 °C at 3170 r/min for 10 min within 3 h of sample collection. The centrifuged samples were stored in Mekamini™ tubes (Mekalasi Oy, Nurmijäri, Finland) at –80°C until all samples were collected for analysis. The biomarkers of EG injury (sTM, syndecan-1 and VAP-1) and the activation of the inflammatory system (IL-6) were analysed at the Minerva Foundation Institute for Medical Research, Helsinki, Finland, with commercially available immunoassays according to the manufacturers’ recommendations: sTM (Quantikine™ ELISA, R&D Systems Europe, Ltd, Abingdon, UK), LoD 0.0078 pg/L, intra- and inter-assay coefficient of variation (CV) 2.3%–3.6% and 5.7–8.0%); syndecan-1 (Human sCD138 [syndecan-1] ELISA Kit, Nordic BioSite AB. Täby, Sweden), LoD 0.0049 ng/L, intra- and inter-assay CV 6.2% and 10.2%, respectively; VAP-1 (Quantikine™ ELISA, R&D Systems Europe, Ltd, Abingdon, UK), LoD 0.000024 ng/L, intra- and inter-assay CV 1.5%–2.4% and 4.5%–4.8%, respectively; IL-6 (Quantikine™ ELISA, R&D Systems Europe, Ltd, Abingdon, UK), LoD <0.0007 pg/L, intra- and inter-assay CV 1.6%–4.2% and 3.3%–6.4%, respectively. In one patient, the preoperative IL-6 value was under the LoD. All other endothelial and inflammatory markers were above the LoD.
Outcomes
To explore the association of EG injury and systemic inflammation, as reflected by IL-6, with cTnT release and PMI.
Statistical analyses
The assumption of a clinically significant two-fold increased level of laboratory parameters (sTM, syndecan-1, VAP-1, IL-6) in PMI patients compared with non-PMI patients and of a standard deviation equal to the difference would require a sample size of at least 10 patients and 40 controls (with a significance level of 0.05 and a power of 80%). Based on these assumptions, we considered a total sample size of 75 patients (15 PMI patients and 60 propensity-matched non-PMI patients) to be adequate for comparisons between the groups. To match controls with PMI patients, we calculated a propensity score for each patient based on demographic and medical data. The variables included in the propensity matching are presented in supplemental digital content (S) Table 1. In brief, the variables included age, sex, main medical history, medications and intraoperative data. Four propensity-matched controls were selected for each PMI patient.
Characteristics of the PMI patients and propensity-matched controls.
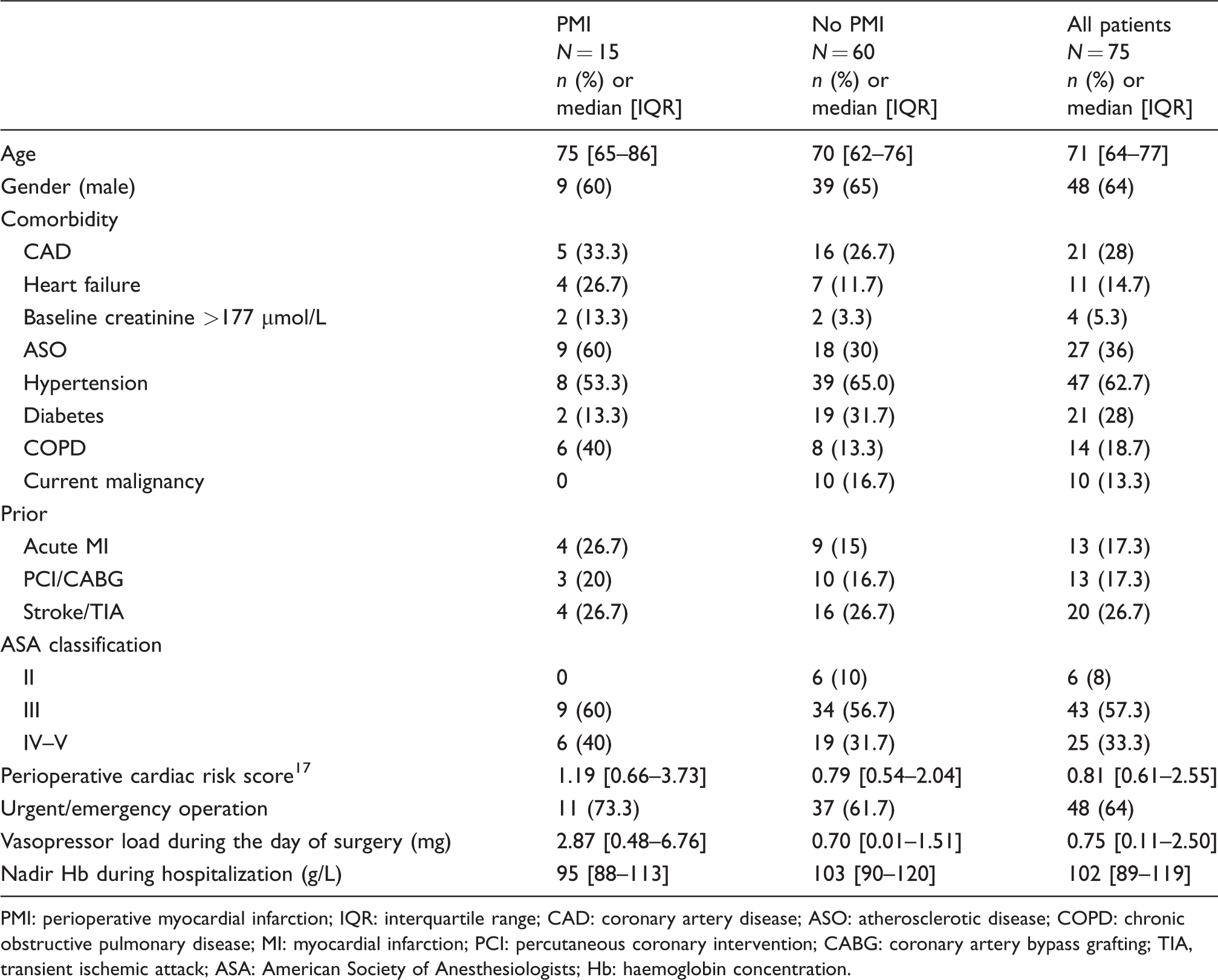
PMI: perioperative myocardial infarction; IQR: interquartile range; CAD: coronary artery disease; ASO: atherosclerotic disease; COPD: chronic obstructive pulmonary disease; MI: myocardial infarction; PCI: percutaneous coronary intervention; CABG: coronary artery bypass grafting; TIA, transient ischemic attack; ASA: American Society of Anesthesiologists; Hb: haemoglobin concentration.
We present continuous variables as medians with interquartile ranges (IQRs) and dichotomous variables as absolute numbers with percentages. The comparisons of continuous variables and frequencies were made using the Mann-Whitney U-test and Fisher’s exact test, respectively. We calculated the Hodges-Lehmann estimator with 95% CIs to measure the magnitude of differences between PMI patients’ and non-PMI patients’ preoperative, 6 h and 24 h postoperative values, as well as the highest values of EG markers and IL-6. We calculated the areas under the receiver operating characteristics curves (AUC) with 95% CIs. The highest plasma concentrations of EG markers and IL-6 were used as test variables. We defined an AUC of 0.5–0.75 as poor, 0.75–0.9 as good and >0.9 as excellent, as suggested. 16 A two-sided P-value of <0.05 was considered significant. Statistical analyses were performed with SPSS version 23 (SPSS; Chicago, IL, USA).
Results
With regard to PMI, a total of 75 patients (15 with PMI and 60 matched controls) were included in the present study (Figure 1). In order to assess the generalizability of the current results, we compared the baseline characteristics of the primary study patients with all plasma samples taken to those with lacking plasma samples. The results are presented in Tables S1 and S2. With the exception of the differences in the numbers of urgent/emergency operations, the use of vasopressor support, the American Society of Anesthesiologists class and perioperative cardiac risk, 17 the patients were comparable in terms of baseline characteristics.
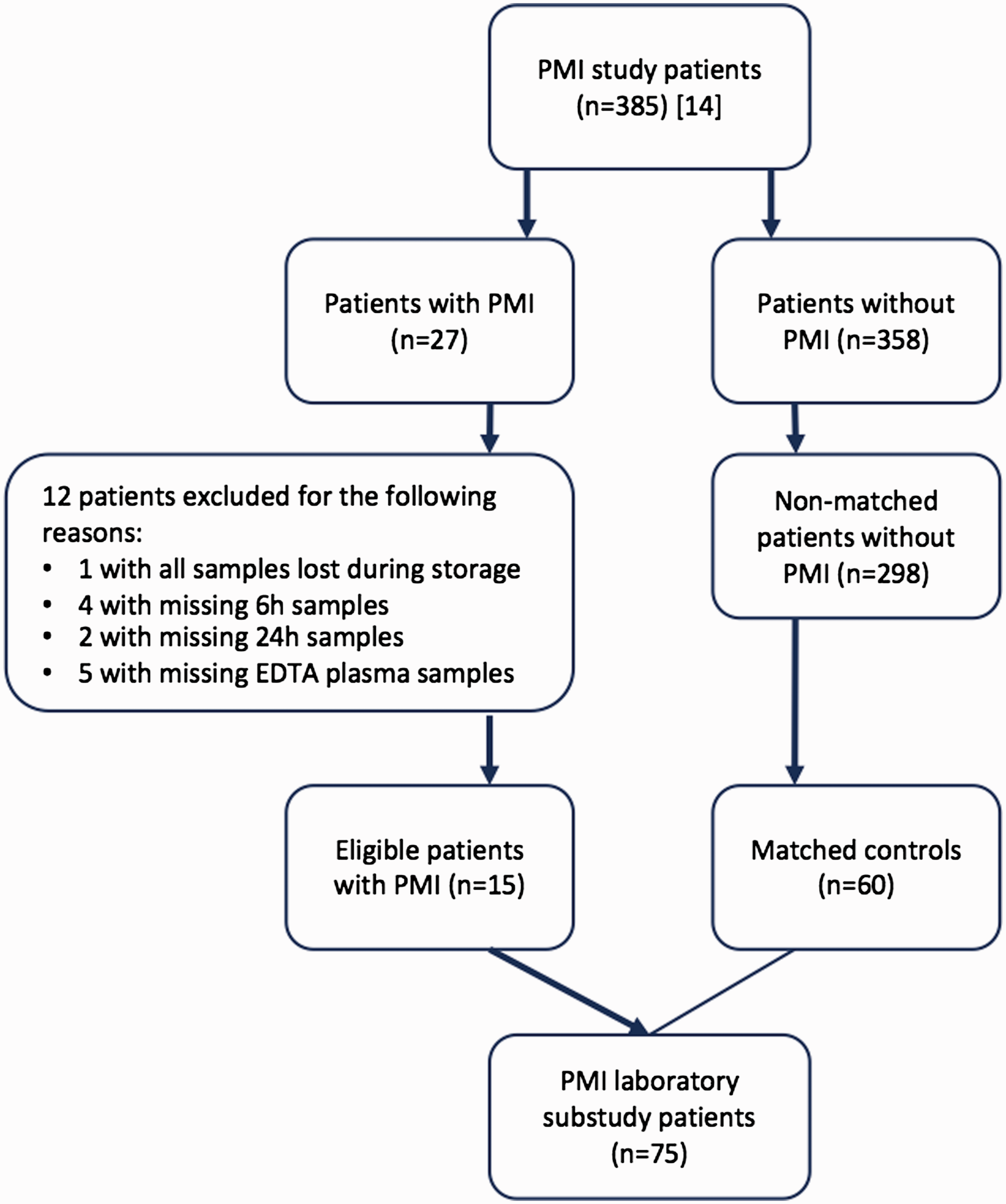
Study flowchart.
PMI
The demographics, medical history and preoperative clinical findings of the PMI patients and their propensity-matched controls are summarized in Table 1. The 90-day mortality of PMI patients was 20%, compared with the 8.3% among non-PMI patients.
Table 2 presents the median values of EG markers and IL-6 in patients with and without PMI. The PMI patients had higher IL-6 concentrations during the whole perioperative 24 h follow-up, but the sTM, syndecan-1 and VAP-1 concentrations were comparable in patients with and without PMI at all time points.
The median values of EG markers and IL-6 measured in PMI patients and propensity-matched controls.
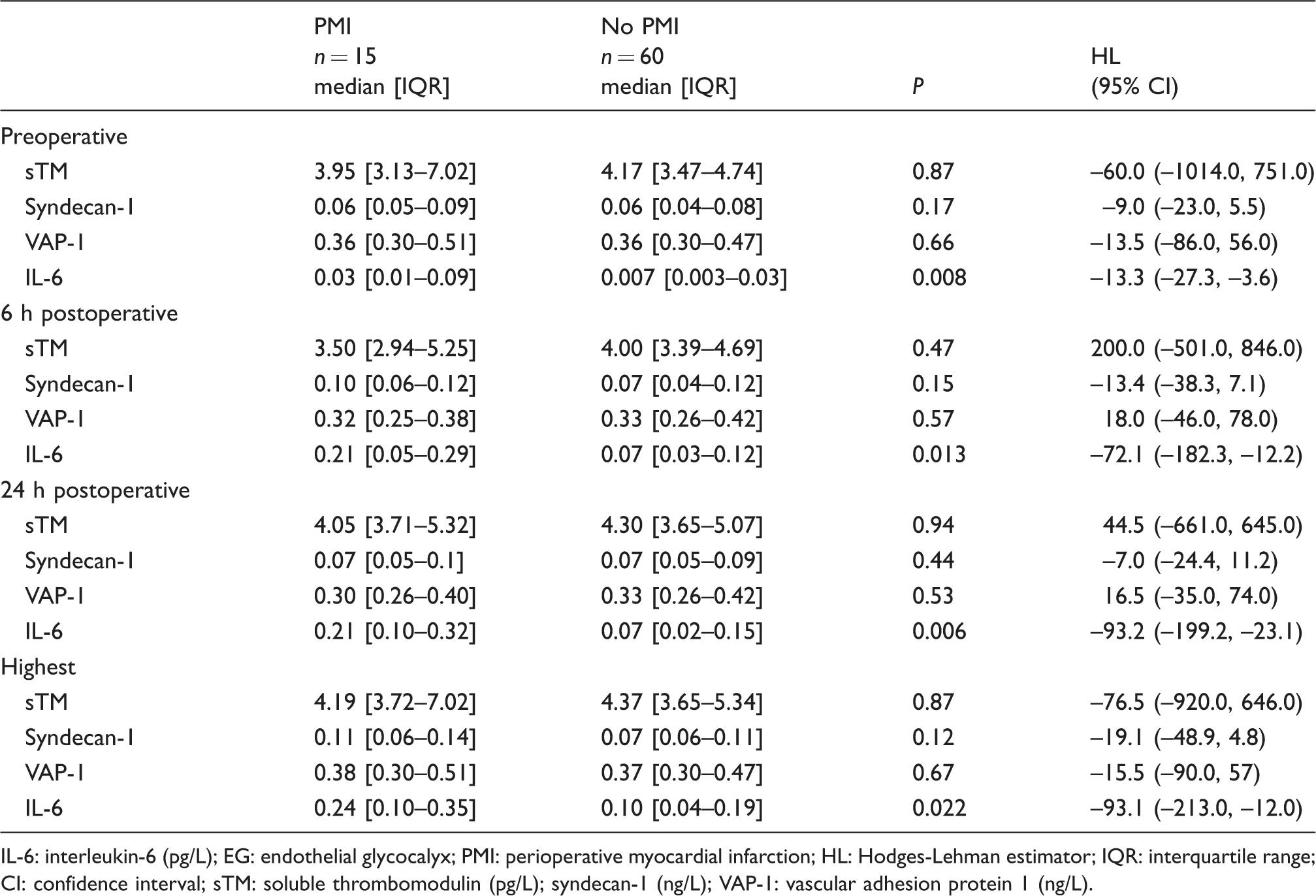
IL-6: interleukin-6 (pg/L); EG: endothelial glycocalyx; PMI: perioperative myocardial infarction; HL: Hodges-Lehman estimator; IQR: interquartile range; CI: confidence interval; sTM: soluble thrombomodulin (pg/L); syndecan-1 (ng/L); VAP-1: vascular adhesion protein 1 (ng/L).
The association of systemic inflammation and EG injury with perioperative cTnT release
Figure 2 illustrates the kinetics of the EG markers and IL-6 in patients with and without PMI. The IL-6 concentrations of PMI patients were significantly higher across all time points – P = 0.002, P = 0.002 and P = 0.001, respectively. However, the sTM, syndecan-1 and VAP-1 concentrations did not differ between PMI and non-PMI patients.
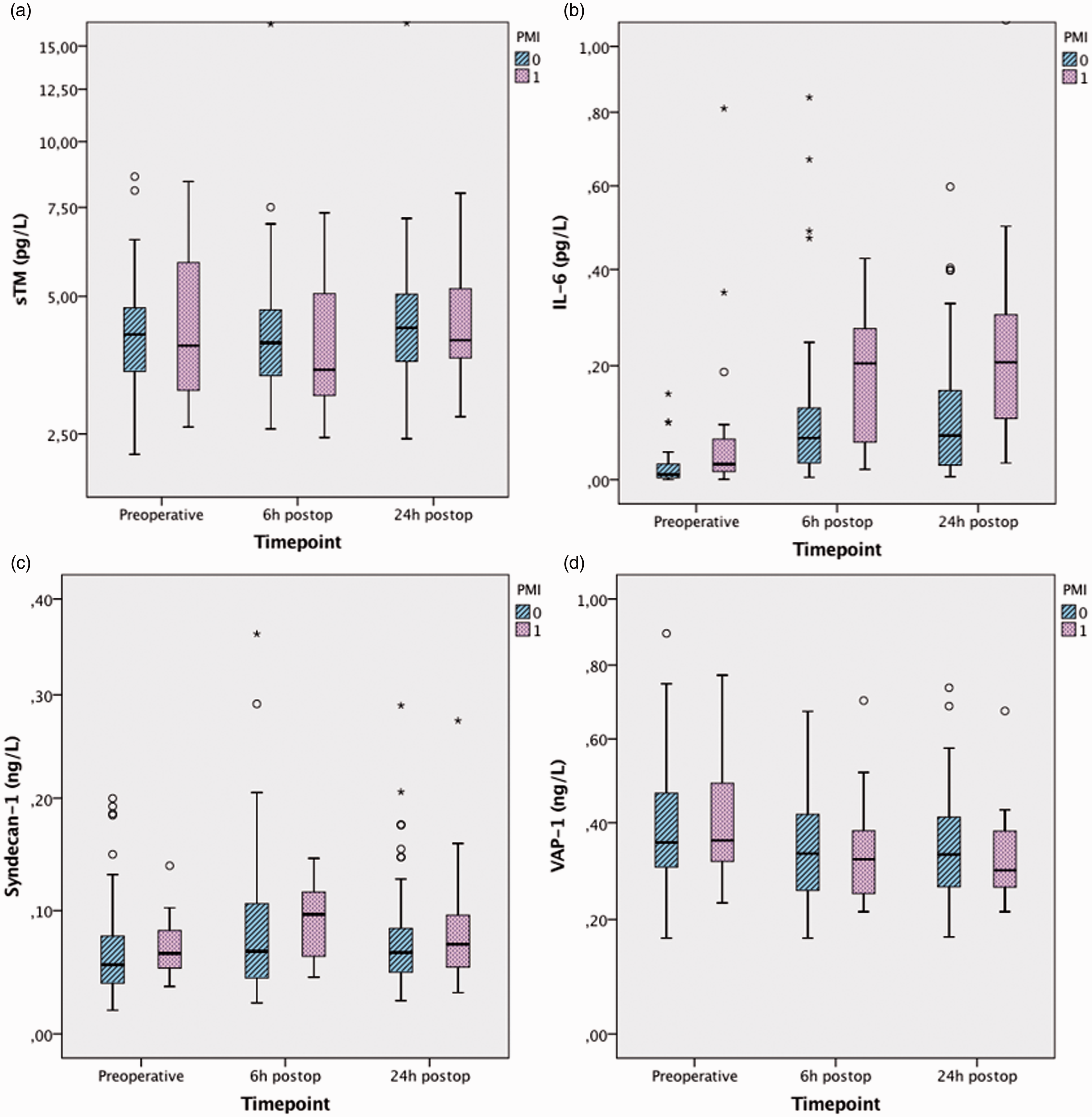
The kinetics of endothelial glycocalyx markers and interleukin-6 in PMI and non-PMI patients. Non-transformed values are presented on a logarithmic scale. Bars represent medians with interquartile ranges (IQRs). T-bars represent values 1.5 times greater than the IQR.
We tested the potential correlations between cTnT, IL-6 and EG markers in PMI and non-PMI patients with Spearman’s rho. The correlations are presented in Tables S3A and S3B. The preoperative IL-6 concentration had a positive correlation with preoperative, 6 h postoperative and 24 h postoperative cTnT in PMI patients, P = 0.05. Furthermore, in PMI patients, the 24 h postoperative sTM correlated positively with preoperative, 6 h postoperative and 24 h postoperative cTnT (P = 0.05). In non-PMI patients, the sTM and cTnT concentrations correlated positively preoperatively (P < 0.001), 6 h postoperatively (P = 0.001) and 24 h postoperatively (P < 0.001).
The association of systemic inflammation and EG injury with PMI
Figure 3 is a graphic representation of the performance of the EG markers and IL-6 in predicting PMI. The results of the analysis are summarized in Table 3. None of the EG markers were predictive of PMI when the highest values were analysed. IL-6 predicted PMI with an AUC of 0.69 (95% CI 0.54–0.85), P = 0.02. The best cut-off value for IL-6 was 0.238 pg/L, with a sensitivity of 0.53, specificity of 0.85 and positive and negative likelihood ratios of 3.56 (95% CI 1.65–7.65) and 0.55 (95% CI 0.32–0.95), respectively.
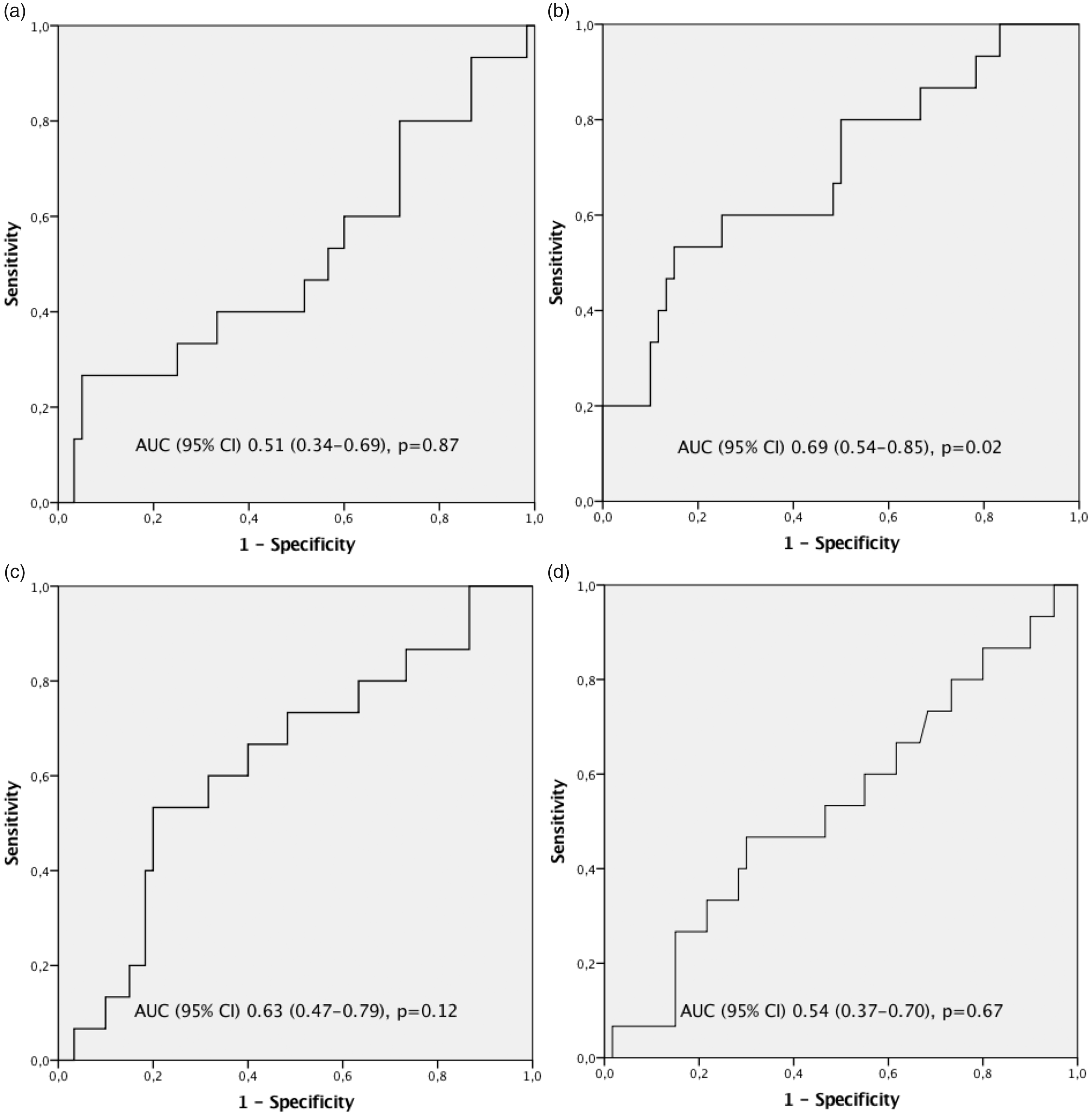
The highest values of the EG markers and IL-6 in predicting PMI. A: sTM, B: IL-6, C: syndecan-1, D: VAP-1.
The highest values of EG markers (sTM, syndecan-1 and VAP-1) and IL-6 in predicting PMI.
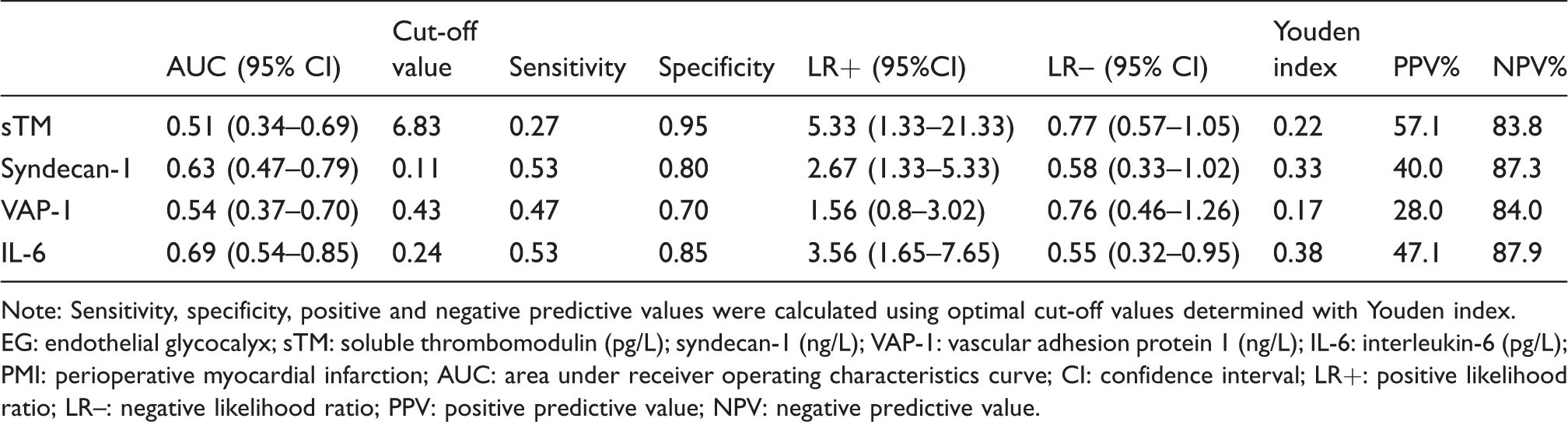
Note: Sensitivity, specificity, positive and negative predictive values were calculated using optimal cut-off values determined with Youden index.
EG: endothelial glycocalyx; sTM: soluble thrombomodulin (pg/L); syndecan-1 (ng/L); VAP-1: vascular adhesion protein 1 (ng/L); IL-6: interleukin-6 (pg/L); PMI: perioperative myocardial infarction; AUC: area under receiver operating characteristics curve; CI: confidence interval; LR+: positive likelihood ratio; LR–: negative likelihood ratio; PPV: positive predictive value; NPV: negative predictive value.
Discussion
In this prospective laboratory study, we demonstrated that high IL-6 concentrations during the perioperative period were associated with cTnT release and PMI. Furthermore, a high perioperative IL-6 concentration may be predictive of PMI, whereas EG markers (sTM, syndecan-1 and VAP-1) were not.
The vascular endothelium is an active paracrine, endocrine and autocrine organ that is crucial in regulating vascular tone and maintaining vascular homeostasis. Moreover, previous observations indicate that endothelial dysfunction may represent a key early step in the development of atherosclerosis and that it is also involved in plaque progression and the occurrence of atherosclerotic complications.18,19 According to current knowledge, endothelial dysfunction may play a critical role in the development of ACS, as a dysfunctional endothelium causes a pro-inflammatory state that contributes to enhanced coronary plaque vulnerability. 20 It also has a decreased ability to produce nitric oxide, resulting in vasospasm and impaired coronary arterial flow. 20 Furthermore, endothelial dysfunction is characterized by a reduced anticoagulatory potential of the endothelium, an increased endothelial production of pro-coagulation mediators and an increased vasoconstriction due to platelet-derived mediators, 11 resulting in a thrombogenic vascular environment.
Several studies have demonstrated the occurrence of EG injury in medical conditions characterized by sympathoadrenal activation, systemic inflammation and I/R injury.8,10,21,22 Ostrowski et al. investigated patients with an acute ST segment elevation myocardial infarction (STEMI), admitted for primary percutaneous coronary intervention (pPCI) and demonstrated that these patients presented with high plasma concentrations of circulating sTM, syndecan-1, adrenalin and noradrenalin. The authors showed that high circulating sTM was associated with myocardial injury as defined by high troponin I (TnI) release, and a similar tendency was seen in the current study (Tables S3A and S3B). Moreover, high syndecan-1 concentrations were associated with short- and long-term mortality and heart failure. 10 Sustained EG injury associates with the severity of post-cardiac arrest syndrome (PCAS), as was recently demonstrated by Bro-Jeppesen et al. 22 The authors showed that baseline IL-6 concentrations were associated with EG activation and endothelial cell injury in patients with severe myocardial damage. 22 However, to the best of our knowledge, no previous study exists concerning the association of endothelial injury with PMI.
The markers used to demonstrate EG activation and injury vary extensively between the studies. The different markers detected in circulating plasma represent the different phases of the endothelial activation and injury.
Firstly, we investigated thrombomodulin (TM), an integral membrane protein expressed on the surface of endothelial cells. It has a critical role as a cofactor in the protein C pathway that is important in regulating coagulation and inflammation. sTM circulates in plasma and is found in increased concentrations in connection with various diseases, such as cardiovascular disease and diabetes, and in ischaemic or inflammatory endothelial damage. 23 We found that the sTM concentrations correlated with cTnT release, especially in patients with no PMI. However, circulating sTM was not predictive of PMI.
Secondly, we measured syndecan-1, an endothelial cell transmembrane protein, which exists in the form of proteoglycans. Previous animal studies suggest that the shedding of EG is associated with a significant increase in circulating syndecan-1,24,25 and this also appears to occur in humans during I/R injury. 9 In accordance with these findings, we observed a rise in syndecan-1 concentrations when preoperative and 6 h postoperative values were compared. Like sTM, syndecan-1 was not predictive of PMI.
Thirdly, we studied VAP-1, which belongs to a family of semicarbamizide-sensitive amine oxidases (SSAOs) and is expressed in endothelial cells. VAP-1 plays an important role in controlling neutrophil adhesion to the endothelium and neutrophil extravasation from blood to tissues. Increased VAP-1 activity has been observed in critically ill patients with septic shock, 26 and elevated concentrations have also been described in obese and diabetic persons. 27 According to our findings, VAP-1 did not associate with perioperative cTnT release or myocardial infarction. In addition to EG markers, IL-6 was measured as a pro-inflammatory cytokine produced mainly by macrophages and monocytes at an inflammatory site. IL-6 is a main stimulator in acute-phase reactions following an infection or tissue trauma but also plays an important role in the development of chronic inflammation. 7 According to the current findings, a perioperative rise in IL-6 is associated with PMI and might be predictive of the complication.
The superiority of any endothelial marker in demonstrating endothelial injury in the clinical setting and in predicting the patients’ outcome has not been demonstrated. In a perioperative setting, Rehm et al. demonstrated that plasma syndecan-1 and heparan sulphate concentrations rose in patients undergoing procedures predisposing them to either global or regional ischaemia followed by reperfusion, whereas intercellular adhesion molecule-1 (ICAM-1) and vascular cell adhesion molecule-1 (VCAM-1) concentrations remained unchanged. 9 This finding led the authors to the conclusion that the circulating components of EG may be more sensitive markers of EG injury than integral membrane proteins. Furthermore, Rehm et al. observed a high interindividual variation in EG shedding, which is in broad agreement with our results. The differences in surgical procedures, perioperative management and the patients’ medical condition are a plausible explanation for the variation.
Despite the great emphasis on perioperative care during the past years, PMI remains a severe complication, with an in-hospital mortality of 12%–40%.28–31 Perioperative hemodynamic instability, leading to oxygen demand/supply disparity, as well as blood flow stagnation and thrombus formation are regarded as major factors in the development of PMI. 32 Therefore, most trials have been targeted towards preventing these adverse factors. However, despite numerous large-scale clinical studies,33–35 efficient and safe methods to prevent PMI are still lacking, and it is likely that we remain unaware of the exact pathophysiological mechanisms of PMI. Considering the physiological disturbances – with impaired vasodilatation, enhanced plaque vulnerability and pro-thrombotic state in particular – caused by a dysfunctional endothelium, endothelial injury seems a logical contributor in the development of PMI. Furthermore, several studies have demonstrated an association between the presence of coronary endothelial dysfunction and ischaemic symptoms or imaging findings in individuals without clinically significant coronary artery disease.36–39 This might explain the fact that perioperative cardiac complications are sometimes hard to predict and may occur in patients with relatively clean coronary arteries. 40
To the best of our knowledge, this was the first study to suggest that EG injury may play a role in the pathophysiology of PMI, and this finding opens the door for possible future therapeutic trials. An interesting report published by Manchurov et al. demonstrated that remote ischaemic preconditioning (RIPC) before pPCI clearly improved the endothelial function in patients with an acute myocardial infarction. 41 Whether similar effects can be achieved in surgical patients and whether this is beneficial in preventing perioperative cardiac complications and improving perioperative outcome could be a subject of future clinical trials. In order to conduct these trials, though, larger observational studies on the association between EG injury and perioperative cardiac complications are needed.
The current study has some important limitations. Firstly, because of the missing blood samples, we were not able to include all consecutive patients of the primary study and, unfortunately, had to exclude 12 PMI patients. The current results should be interpreted taking this into consideration. However, as presented in Tables S1 and S2, patients with and without all blood samples available were comparable regarding their baseline characteristics. Additionally, we matched controls for the PMI patients using a propensity score and increased the statistical power by including four controls for each PMI patient. Secondly, this was an observational study, not allowing an evaluation of the causal relationship. Finally, the assessment of diagnostic ability (using positive likelihood ratios) was based on cut-off values derived from the same study because previous values for PMI patients did not exist in the literature. Despite the small sample size, there was a fairly strong correlation between cTnT, IL-6 and EG markers in 15 PMI patients. Thus, our findings should be considered as first hypothesis-generating preliminary evidence regarding the potential association between EG injury and PMI.
Conclusion
Systemic inflammation, reflected by IL-6, associates with cTnT release and PMI. Furthermore, IL-6 demonstrated some potential to predict PMI, whereas EG markers did not. These findings should be considered preliminary and need to be confirmed or refuted in larger future studies.
Supplemental Material
Supplemental Material1 - Supplemental material for The association of endothelial injury and systemic inflammation with perioperative myocardial infarction
Supplemental material, Supplemental Material1 for The association of endothelial injury and systemic inflammation with perioperative myocardial infarction by Aino Ollila, Leena Vikatmaa, Juha Virolainen, Sara Nisula, Päivi Lakkisto, Pirkka Vikatmaa, Ilkka Tikkanen, Maarit Venermo and Ville Pettilä in Annals of Clinical Biochemistry
Supplemental Material
Supplemental Material2 - Supplemental material for The association of endothelial injury and systemic inflammation with perioperative myocardial infarction
Supplemental material, Supplemental Material2 for The association of endothelial injury and systemic inflammation with perioperative myocardial infarction by Aino Ollila, Leena Vikatmaa, Juha Virolainen, Sara Nisula, Päivi Lakkisto, Pirkka Vikatmaa, Ilkka Tikkanen, Maarit Venermo and Ville Pettilä in Annals of Clinical Biochemistry
Supplemental Material
Supplemental Material3 - Supplemental material for The association of endothelial injury and systemic inflammation with perioperative myocardial infarction
Supplemental material, Supplemental Material3 for The association of endothelial injury and systemic inflammation with perioperative myocardial infarction by Aino Ollila, Leena Vikatmaa, Juha Virolainen, Sara Nisula, Päivi Lakkisto, Pirkka Vikatmaa, Ilkka Tikkanen, Maarit Venermo and Ville Pettilä in Annals of Clinical Biochemistry
Supplemental Material
Supplemental Material4 - Supplemental material for The association of endothelial injury and systemic inflammation with perioperative myocardial infarction
Supplemental material, Supplemental Material4 for The association of endothelial injury and systemic inflammation with perioperative myocardial infarction by Aino Ollila, Leena Vikatmaa, Juha Virolainen, Sara Nisula, Päivi Lakkisto, Pirkka Vikatmaa, Ilkka Tikkanen, Maarit Venermo and Ville Pettilä in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
We want to thank research assistants Riikka Kosonen and Sanni Perttunen for their valuable assistance with the ELISA analyses.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by clinical research funding no. TYH 2014239 from the University of Helsinki and Helsinki University Hospital (VP, LV) and by grants from the Sigrid Juselius foundation (VP) and the Finnish Society of Angiology (LV).
Ethical approval
Ethical approval for the study (No. 11/13/03/02/2014) was provided by the Ethics Committee of the Department of Surgery at Helsinki University Hospital, Helsinki, Finland, on 30 January 2014.
Guarantor
VP
Contributorship
AO, LV and VP contributed to the design of the study. AO conducted literature searches, gathered clinical data, wrote the first draft and edited the final version of the paper. JV made all the PMI diagnoses. PL was responsible for the laboratory analyses. AO performed all statistical analyses, and SN acted as a consultant. All of the authors contributed to the critical review of the article and approved the final version.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
