Abstract
Background
Point-of-care testing has been developed to provide rapid test results. Most published studies focus on analytical performance, neglecting its impact on patient outcomes.
Objective
To review the analytical performance and accuracy of point-of-care testing specifically planned for immunoassay and to evaluate the impact of faster results on patient management.
Methods
A search of electronic databases for studies reporting immunoassay results obtained in both point-of-care testing and central laboratory scenarios was performed. Data were extracted concerning the study details, and the methodological quality was assessed. The analytical characteristics and diagnostic accuracy of six points-of-care testing: troponin, procalcitonin, parathyroid hormone, brain natriuretic peptide, C-reactive protein and neutrophil gelatinase-associated lipocalin were evaluated.
Results
A total of 116 scientific papers were analysed. Studies measuring procalcitonin, parathyroid hormone and neutrophil gelatinase-associated lipocalin reported a limited impact on diagnostic decisions. Seven studies measuring C-reactive protein claimed a significant reduction of antibiotic prescription. Several authors evaluated brain natriuretic peptide or troponin reporting faster decision-making without any improvement in clinical outcome. Forty-four per cent of studies reported analytical data, showing satisfactory correlations between results obtained through point-of-care testing and central laboratory setting. Half of studies defined the diagnostic accuracy of point-of-care testing as acceptable for troponin (median sensitivity and specificity: 74% and 94%, respectively), brain natriuretic peptide (median sensitivity and specificity: 82% and 88%, respectively) and C-reactive protein (median sensitivity and specificity 85%).
Conclusions
Point-of-care testing immunoassay results seem to be reliable and accurate for troponin, brain natriuretic peptide and C-reactive protein. The satisfactory analytical performance, together with an excellent practicability, suggests that it could be a consistent tool in clinical practice, but data are lacking regarding the patient outcomes.
Background
The technology of point-of-care testing (POCT) aims to generate appropriate test results quickly, thereby improving clinical outcomes and reducing associated costs. 1 The effect of POCT should therefore be evaluated in terms of correct diagnosis, improvement of therapeutic strategies and patient outcomes in the healthcare setting. A test implicates a benefit if its result leads to appropriate action. Few studies have been conducted to evaluate the effect of the results obtained in POCT on clinical outcomes, and, usually indicate the decrease of turn-around-time (TAT) as a critical factor in determining positive effect on patient management. 2 As evaluated in a previous survey, most studies evaluated surrogate outcomes only, which do not directly impact clinical outcomes. 3
The implementation of POCT is successful when the performed assay is helpful for medical decision-making and does not need additional testing for confirmation from a central laboratory. 4 New technologies in POCT should deliver to clinicians rapid and reliable test results, and high analytical and diagnostic performance, to reduce the incidence of false-negative or false-positive results which could lead to unjustified medical interventions. 5 Several studies, evaluating the benefit of POCT in emergency department (ED), reported a significant decrease of TAT, but this effect did not lead to shorter length of stay (LOS) 6 or to drive the decision-making process. 7 Furthermore, the main advantages of POCT may be the early and appropriate support to diagnosis and treatment, but there are few studies evaluating how POCT results influence clinical decisions.
The aim of this research is to systematically revise the current state of six immunometric tests performed in POCT: brain natriuretic peptide (BNP), C-reactive protein (CRP), neutrophil gelatinase-associated lipocalin (NGAL), procalcitonin (PCT), parathyroid hormone (PTH), and cardiac troponin (Tn) and assess the impact of POCT on patients’ management.
Methods
We reported this review according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (Prisma) guideline. 8
Eligibility criteria
Studies were included if they met the following criteria: (i) randomized controlled trial (RCT), prospective cohort, case-control or diagnostic test accuracy studies; (ii) specimens analysed by POCT and laboratory instruments; (iii) at least one indicator of patient outcomes.
Identification of studies
We performed a comprehensive systematic literature search and scanned the reference lists of articles. This search was applied to Medline (1995 – February 2016) and adapted for Embase (1995 – February 2016) to collect all potentially relevant scientific papers published in English. Databases were searched using the following search terms: POCT, BNP, CRP, NGAL, PCT, PTH and Tn. Duplicate articles were removed from the search results.
Selecting published studies
One reviewer examined the search results, screened the titles, abstracts and reference lists of identified articles and evaluated the eligibility of the individual studies. The full texts of the selected articles were obtained and examined for inclusion. If there was doubt about the inclusion of a study, eligibility was discussed by two authors.
Assessment of methodological quality
The Cochrane Collaboration risk of bias tool for cohort studies was adapted, and three questions were considered appropriate for the current study.
Was the study design adequate for the objective of the study? Were outcome data adequately reported? Where results discussed adequately?
Outcomes were classified as ‘adequate’ or ‘inadequate’ based on the quality of information reported. ‘Adequate’ was considered when the study design was randomized or prospectively designed, included sufficient information about diagnostic data (sensitivity [Sn] and specificity [Sp], positive and negative predictive values [PPV and NPV]) or analytical results (value of coefficient of variation, test ‘r’, mean and standard deviation) and the discussion supported the reported study results. ‘Unclear’ was assigned to the question if the information reported in the articles was incomplete.
Data extraction and analysis
One author extracted information on the general features of each study using a standardized extraction form, and a second author checked the data. The details included: (a) characteristic of the study: authors, reference, study design; (b) characteristics of patients (age, sex, number of sample for each patients group); (c) data about analytical performance of the test; (d) diagnostic accuracy outcomes: Sn, Sp, PPV and NPV; (e) patient important outcomes: number of myocardial injuries, infections or re-interventions, recurrence of hyperparathyroidism and renal failure; (f) information about clinical impact of POCT: antibiotic prescription, time to clinical decision, TAT, LOS.
Results
Study selection
Our literature search identified 519 references. Duplicates and irrelevant references excluded 100 records. After the initial screening of the abstracts, 176 studies proved to be eligible for inclusion and their full texts were analysed in more detail. A further 64 studies were excluded because they: (i) had no comparison with laboratory ( The literature flow diagram.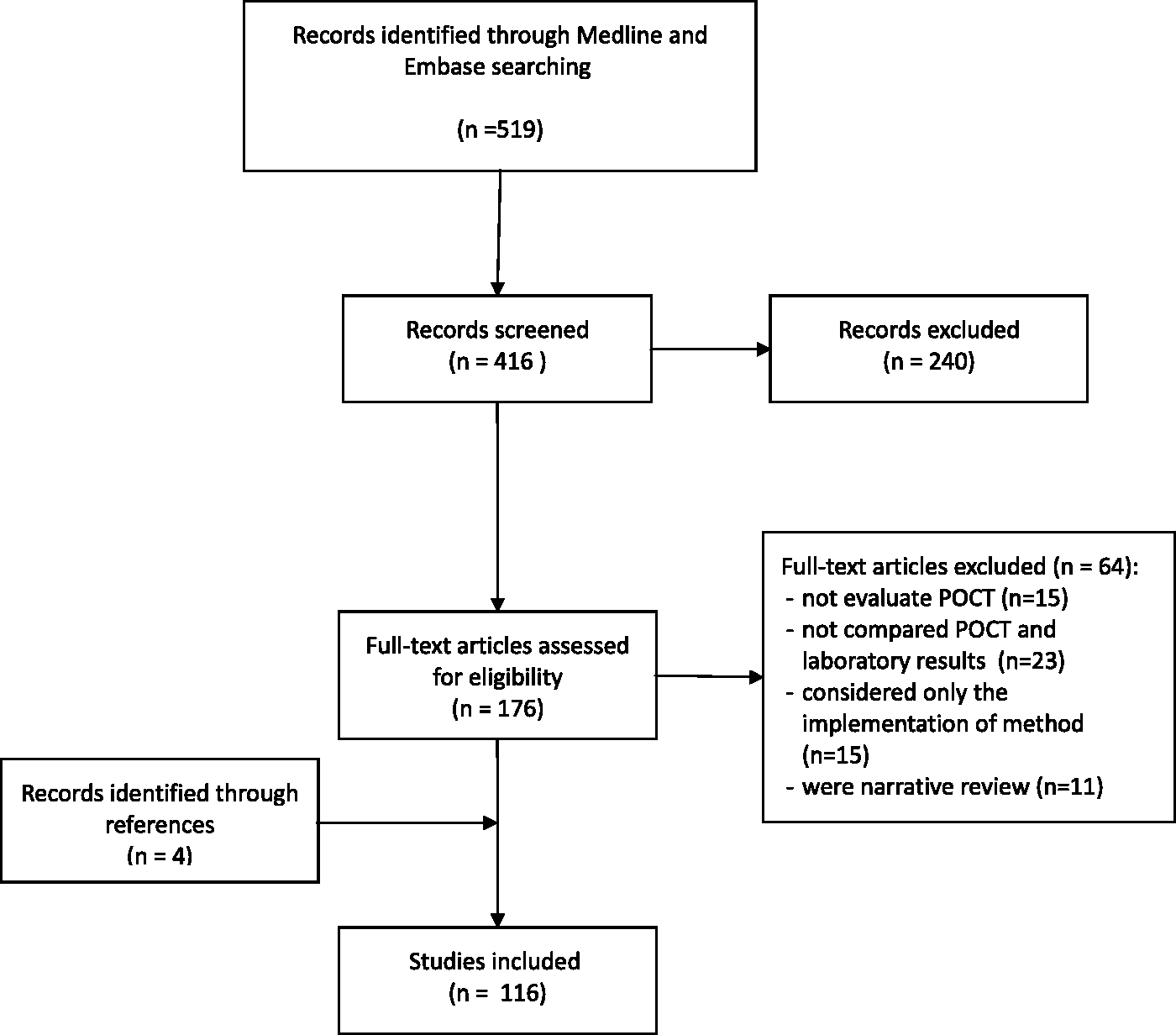
Study characteristics
Characteristics of included studies.

BNP: brain natriuretic peptide; CRP: C-reactive protein; NGAL: neutrophil gelatinase-associated lipocalin; PCT: procalcitonin; PTH: parathyroid hormone; Tn: troponin; CK-MB: creatine kinase-myocardial band; Myo: myoglobin; ED: emergency department; RCT: randomized controlled trial; CCU: critical care unit.
Most studies were performed in ED (
Methodological quality
Overall, we evaluated most studies as adequate in terms of methodological quality (Table 1). Most studies (
Evaluation of clinical impact of POCT
Number of studies evaluating specific outcomes.
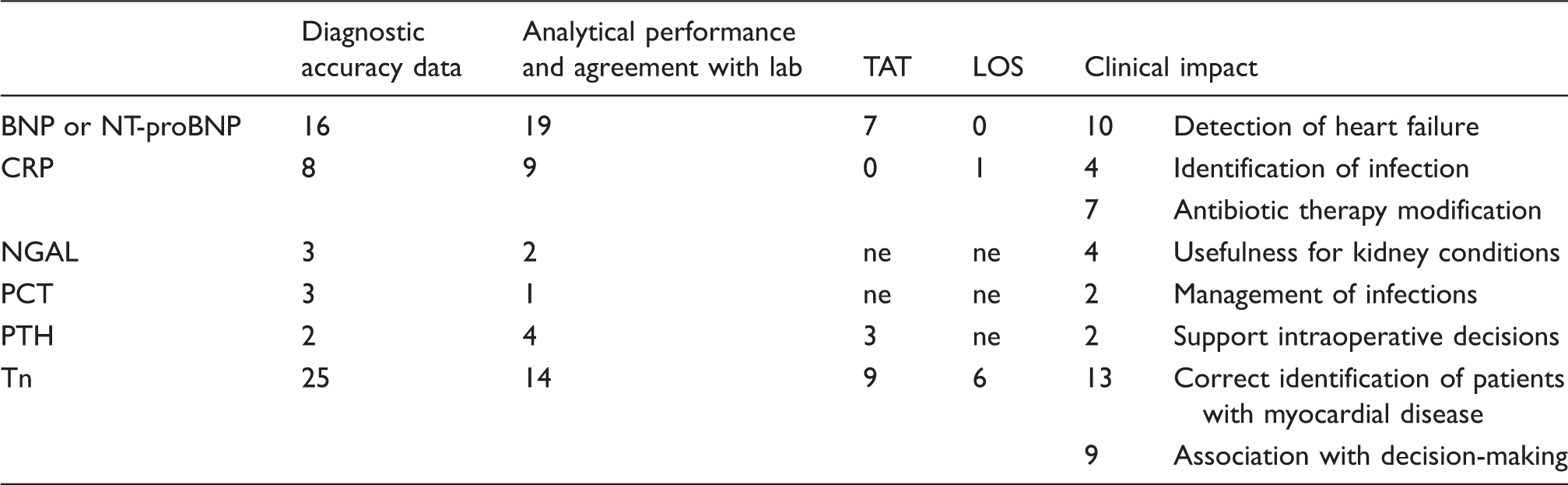
BNP: brain natriuretic peptide; CRP: C-reactive protein; NGAL: neutrophil gelatinase-associated lipocalin; PCT: procalcitonin; PTH: parathyroid hormone; Tn: troponin; TAT: turn-around-time; LOS: length of stay; ne: not evaluated.
Seven studies (28%) dedicated to CRP evaluated the antibiotic therapy modification when CRP was determined by POCT compared with the same test performed in a central laboratory. In particular, three studies40,43,59 reported a significant reduction of antibiotic prescription in POCT group compared with the control group. Instead, three studies reported a significant prediction of antibiotic prescription using CRP in POCT (odds ratio was 0.2, 48 8.949 and 1.6, 50 respectively). Moreover, four studies (16%) evaluated CRP in the identification of patients with bacterial infections. Of these, one study 60 proved that the POCT test successfully identified patients with bacterial or viral infection, two studies51,52 reported a good PPV for bacterial aetiology and one study 55 reported no difference between the intervention and the control group in infection prediction.
Four authors,12,17,65,66 reported in the discussion section that the POCT testing of NGAL is an effective early biomarker for acute kidney disease, providing important information regarding renal perfusion status.
One study 70 suggested the implementation of PCT as a helpful biomarker for antibiotic decision-making in ED.
Two studies about PTH72,73 reported that faster results timely reported to the surgical team, improved patient outcome and care.
Half of the studies about Tn (
Nineteen studies (17%) reported a significant decrease in TAT using POCT technology compared with tests performed in a central laboratory. Seven studies evaluated the effect of POCT on LOS reduction, concluding that generally POCT applied to Tn testing in ED reduces LOS. However, a comparison with a control group did not result in any significant difference. Furthermore, it is worthy to note that the decreased TAT did not correspond to any decrease in LOS.
Analytical performance
Forty-nine studies evaluated the analytical performance of BNP (
Diagnostic accuracy
Twenty-five studies on Tn (54%), 16 on BNP (52%) and 8 on CRP (30%) evaluated the diagnostic accuracy of POCT. Tn in POCT was proven to be effective in identifying patients with cardiac disease: Sn ranged from 39% to 100% and Sp ranged from 57% to 100%. In these studies, the PPV ranged from 20% to 100% and the NPV from 31% to 100% for predicting the presence or the absence of cardiac disease. Likewise, the measurement of BNP or pro-BNP in POCT showed a good accuracy: Sn ranged from 67% to 100% and Sp from 29% to 99%, the PPV ranged from 72% to 95%, and the NPV from 84% to 100%. The diagnostic accuracy was also acceptable for CRP with Sn ranging from 84% to 100% and Sp from 79% to 95%.
Discussion
In our review, we explored the clinical usefulness of POCT for immunoassay for BNP and proBNP, CRP, PCT, PTH, NGAL and Tn. The advantages and limits of these biomarkers determined by POCT were analysed in terms of patients’ management.
A total of 116 scientific papers were included, 49 of these studies (44%) reported analytical data, showing satisfactory correlations between results obtained through POCT instruments and those in a central laboratory setting. Forty-nine studies evaluating the diagnostic accuracy of Tn, BNP and CRP, reported sensitivity and specificity data that show an acceptable precision level of test results for the correct and prompt diagnosis of myocardial injury or infection.
Moreover, we investigated the possible clinical impact of POCT testing in 51 studies. Five studies performed on PCT, PTH and NGAL were available on this issue and reported a limited impact on diagnostic decisions. Seven studies evaluating CRP showed a significant decrease of antibiotic prescription, also proven in a previous systematic review. 121 Several studies evaluating BNP or Tn highlighted the usefulness of rapid tests especially in ED. However, there was insufficient evidence defining an improvement in clinical outcomes. In ED, where POCT for immunoassay is commonly applied, the availability of early and reliable diagnostic biomarkers for cardiac disease is especially useful in the provision of timely and accurate AMI diagnosis, 122 thereby reducing the incidence of inappropriate diagnosis. 123 Despite the rapid TAT, early POCT had a lower sensitivity compared with hs-cTn performed in a central laboratory, which is more effective in excluding AMI in patients accessing ED with higher accuracy. 124 Our analysis confirms that POCT for cardiac markers does not seem to significantly improve the clinical outcomes, as concluded by a previous systematic review. 125
The usefulness of POCT should be strongly associated with the requirement of a technology able to provide appropriate results together with optimal analytical performances to support clinical decision in the light of patients’ characteristics. Most evaluated studies simply evaluated the decrease of TAT, suggesting that the decrease of time elapsed from test request and release of results could produce an advantage to patients’ management.5,6,17,74,77,87,89,93,95 Available studies evaluated scientific dimensions only, which are considered surrogate outcomes, whereas an effective evaluation should include clinical outcomes and the relevant impact of timely results on patients’ management. 3
It seems logical that the reduction of time between test request and result availability facilitates early treatment intervention and clinical actions, thereby improving the disease management. However, the time to clinical action also depends on factors such as doctors’ experience, elaboration and interpretation of data or the need for further investigations, clinical organization, other technology and facilities present in each clinical setting. Thus, for patients with suspected deep venous thrombosis, acute coronary syndrome or acute appendicitis, POCT significantly reduced TAT but was proven to have a limited effect on time to action.126,127 Furthermore, some authors showed as the usefulness of POCT in faster decision-making, influencing treatment and further care, 128 supporting clinical decisions and stratifying patients, 5 but only in specific environments when access to care is limited or in case of environmental disasters.129,130
The literature evaluation showed that POCT is part of a healthcare system in which the patient is the central point. Our systematic review confirms that POCT is characterized by good agreement with central laboratory system, and analytical and diagnostic accuracy. However, our research showed that, although POCT is widely adopted, its impact on time to clinical decision and patient management has not been consistently investigated. Evaluation of patient outcome is an important issue in the assessment of POCT effectiveness in clinical practice. Studies evaluating the impact of tests on patient outcomes remain partial, and consequently, this systematic review cannot offer suggestions or strong recommendations. Studies evaluating the impact of POCT in terms of patient outcome are indispensable for harmonization. In the process of harmonization, the issue of request pertinence was debatable, and the agreement in terms of interpretation of results, analytical performance and reference values for POCT was reached, but evidence about the improvement in the quality of healthcare after TAT reduction are not available.
Moreover, while interpreting the results of this review, some aspects should be considered. The included studies were performed in heterogeneous setting (especially in primary care or ED) and enrolled heterogeneous patients. Further, different POCT technologies were used, different cut-offs were considered and different operators carried out the tests in different conditions and organizational contexts. All these aspects contribute to generate inharmonious results and this renders the generalization of the data presented in the different research difficult.
In conclusion, POCT for immunometric markers offers several clinical advantages: faster test results with minimal analytical engagement. They have a potential to improve patient outcome, although this aspect is inadequately recorded in current clinical studies.
To introduce POCT in daily practice, the instrument should be adequate to guarantee, in a short TAT, the requested analytical and diagnostic performance, to avoid misleading results, to identify and classify patients according to the risk of injury 122 and to guarantee the best clinical outcomes.
It is recommended that future publications on this topic include the impact on clinical decisions and patient outcomes.
Footnotes
Acknowledgements
The authors wish to thank Johanna Chester who assisted with an editorial evaluation of this review.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was not required because this work is a review of biomedical literature.
Guarantor
VP.
Contributorship
All the authors have accepted responsibility for the entire content of this submitted manuscript and approved submission. VP conceived and designed the study. VP and TT wrote the protocol. VP designed and implemented the search strategies. VP selected studies, assessed validity, and extracted data. VP entered and analysed the data. All authors interpreted the data, prepared the full review and contributed to its revision, interpretation of results, and approval.
