Abstract
Background
The introduction of troponin assays with higher analytical sensitivity and enhanced performance has produced new challenges for both laboratory and clinician in the optimal investigation of patients with cardiovascular disease. After some years of collective experience with this new generation of assays, this survey aimed to assess the level of consensus that exists regarding their application.
Methods
A questionnaire was designed, based on a review of published evidence and current opinion, to obtain information on a number of key areas relating to troponin analysis and reporting and was circulated to lead laboratory consultants across the UK and Ireland.
Results
Completed questionnaires were received from 94 laboratory contacts. Sixty per cent of those who responded had implemented a high-sensitivity troponin assay, with the Roche Cobas troponin T high sensitivity assay the most widely used. It is evident that some confusion remains regarding the definition of high-sensitivity assays and there was considerable variation in practice, even among those using the same manufacturer’s assay.
Conclusions
There is a need for greater consensus in the approach to the clinical utilization of troponin assays with improved sensitivity and it is important that laboratories are fully aware of the capabilities of their assay and provide useful guidance to users. On the basis of survey findings and the existing evidence base, a number of recommendations have been proposed to improve current practice and enhance patient safety.
Introduction
The inclusion of troponin in the essential criteria defining acute myocardial infarction (AMI) in 2000 represented something of a coming of age for cardiac biomarkers. 1 Since then there has been continuous development, with regular revisions of the clinical definition of AMI paralleling refinements in the specification and performance of troponin T (TnT) and Troponin I (TnI) measurement.2–4 Troponin has now largely displaced the more traditional cardiac markers, and although there are several novel candidates that show some promise, none have gained a significant foothold in routine practice. Troponin methodology is continuing to push the boundaries towards yet untapped potential.
It was the clinical demand for improved analytical performance around the 99th percentile of the ostensibly healthy population that led commercial companies to develop troponin assays with enhanced analytical sensitivity capable of measuring significantly lower concentrations. The desire was for assays capable of ≤10% coefficient of variation (CV) at the 99th percentile, which would in turn allow quantitative results to be obtained for a significant proportion of the population distribution. 5 A number of more sensitive troponin assays have been developed in recent years, all with different capabilities, and the term high sensitivity (hs) was introduced to enable users to distinguish these from previous generations. The TnT-hs assay from Roche Cobas was the first commercial assay of this new generation to become widely available and has since been followed by sensitive troponin I applications from a number of manufacturers.6–9 As developments in this field have continued, manufacturers have introduced different terminologies to describe their particular assay. This has led to some confusion, prompting the development of the Apple scorecard 5 to benchmark assay performance.
Implementation of these new troponin assays with increased analytical sensitivity has significantly challenged conventional thinking around the application of troponin testing in clinical practice and we are still coming to terms with the implications. Initially, the introduction of sensitive troponin assays raised many more questions than anyone was equipped to answer. Many clinical laboratory services were faced with the not insignificant task of overseeing a transition to the new assays and ensuring that clinical users were as aware as possible of the implications and impact on patient investigation and care. Although there were welcome initiatives to provide consensus guidance for some assays at the time, 10 this was a step change almost as significant as the original introduction of troponin more than two decades earlier, necessitating a reengineering of our investigative strategies that would take time to bed in.
So now, with the benefit of some years’ collective experience with the new generation of troponin assays, we sought to survey laboratory services across the UK and Ireland. We aimed to obtain an insight into the current application of cardiac markers and assess levels of consensus, alongside the ever-growing literature, regarding the use of troponin assays in clinical practice. Here, we report on the results of this survey, conducted under the auspices of the National Clinical Biochemistry Audit Group (NCBAG) of the Association for Clinical Biochemistry and Laboratory Medicine. We also make a number of recommendations supported by the current evidence base, which we believe would improve current practice. We acknowledge the valuable contributions and feedback from the membership of the NCABG and the local Northern Ireland Regional Audit Group.
Methods
Summary of the key themes relating to troponin analysis that were surveyed.

Results
Completed surveys were received from 94 individual laboratory contacts from across the UK and Ireland either online or by post. This covered 92 individual Hospital Trusts and 20 different pathology networks. We estimate the response rate at better than 54% and broadly representative of current practice across the UK and Ireland. Only one response was counted when two or more labs were using the same assay as a unified laboratory service. However, the difficulty of establishing this is perhaps illustrated by the fact that while 30% of respondents were part of a regional pathology network, only 16% of these networks had a harmonized approach to the use of troponin. Here, we present the survey results on the key troponin themes surveyed.
Defining hs-troponin assays
The survey aimed to establish the number of labs now using a troponin assay with improved sensitivity. Users were asked if their current assay met the hs criterion as set by Apple & Collinson, 2012 (Imprecision ≤ 10% CV at the 99th percentile),
11
and how this had been established. Eighty per cent of respondents stated that they had an hs assay, defined by meeting the above imprecision target, although only 22% of these had conducted an in-house confirmation of the manufacturer’s product insert claims. A breakdown of the number of respondents who reported as having an hs assay and the specific assays noted as hs or non-hs is shown in Figure 1. The most common reason given by those who were not currently using an hs assay was that it was not an option under their current managed service contract.
Chart illustrating the number of hs (dark shading) and non-hs (light shading) assays reported and the individual assays reported in each group. Some assay types were reported under both hs and non-hs by different respondents.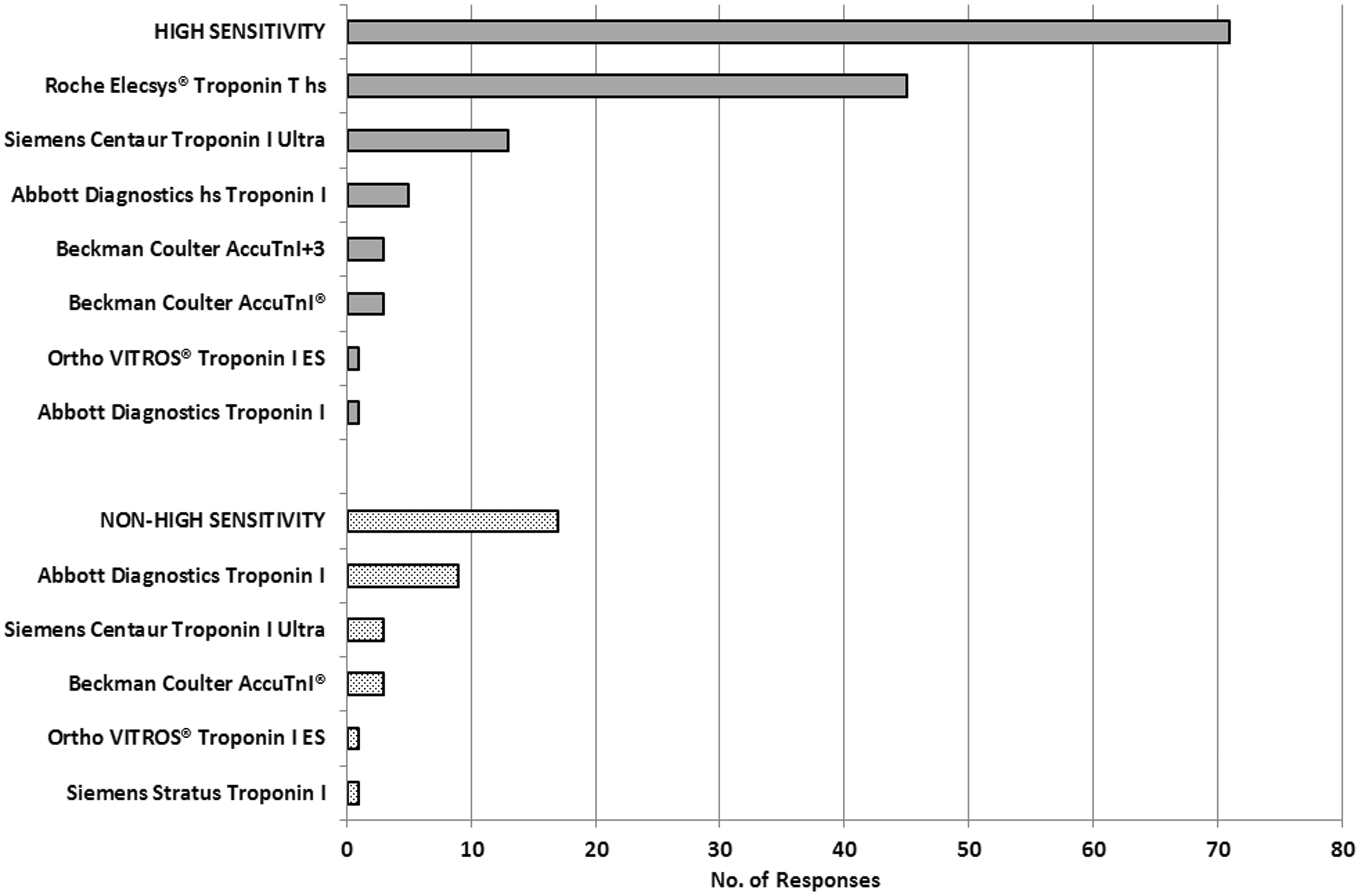
Assay parameters
IFCC: International Federation of Clinical Chemistry and Laboratory Medicine; hs: high sensitivity; NICE: National Institute for Health and Care Excellence; CV: coefficient of variation; hsTnI: high-sensitivity troponin I; N/A: not available.
Apple Scorecard designations – 1. Acceptance criteria: Guideline acceptable (99th percentile CV ≤ 10%), clinically usable (99th percentile CV > 10 to ≤20%), not acceptable (99th percentile CV > 20%); 2. Designation: Level 1 (contemporary) – <50% measurable normal values below 99th percentile, Level 2 (first-generation hs – 50 to <75% measurable normal values below 99th percentile), Level 3 (second-generation hs) – 75 to <95% measurable normal values below 99th percentile, Level 4 (third-generation hs) – ≥95% measurable normal values below 99th percentile. 5
Beckmann AccuTnI + 3 noted as an hs assay in the recent NICE diagnostics guidance. 13
The values for limit of detection (LOD), lowest reportable result and the 99th percentile are obviously assay-dependent. However, it will be noted that even within assay types, there was a variety of levels submitted for these parameters and there may have been some misunderstanding over what was being asked.
Variations in troponin between males and females and the effect of increasing age on troponin concentrations have been discussed in the literature.21–23 Few laboratories were using gender-specific decision limits and most (n = 62) had not considered it. Of all current manufacturers, only Abbott Diagnostics (hsTnI assay) quote a separate 99th percentile cutoff for male and female. No laboratories reported using age-related reference values.
Quality assurance
Strategies for internal quality control (IQC) procedures varied. Of those who responded (n = 72), 46% run IQC once a day, 31% twice daily, with 23% quoting three times or more per day. Commonly, independent suppliers were used for IQC material at three levels, though there was a significant variation in the target concentrations. Of the Roche Cobas TnT-hs users who responded, only three labs were running a quality control sample with a troponin concentration of ≤14 ng/L (the 99th percentile). The majority (n = 18) were using a concentration above 20 ng/L for a low IQC level and 12 labs were running a low IQC containing a TnT concentration between 15 and 20 ng/L. There was a similar pattern among TnI assay users, with only some laboratories running IQC at or close to the 99th percentile.
Samples, assay interference and critical limits
Several sample types are suitable for troponin testing including plain clotted, serum-separator tube (SST), lithium heparin (Li-Hep) and EDTA tubes and most respondents stated that they routinely accept more than one sample type. Some labs stated that they only accept a single sample type, commonly SST or Li-Hep, from particular wards. Fifty-two per cent of respondents advise users against interchanging different samples types within a patient series although 38% did not provide any such guidance.
Laboratories were also asked how they dealt with haemolysed, icteric or lipaemic samples for troponin analysis. Reviewing samples for haemolysis was common and response data for how labs deal with samples breaching indices limits is shown in Figure 2.
Breakdown of the responses received for how laboratories deal with troponin samples that breach indices limits. A summary of the comments noted under other practice is shown.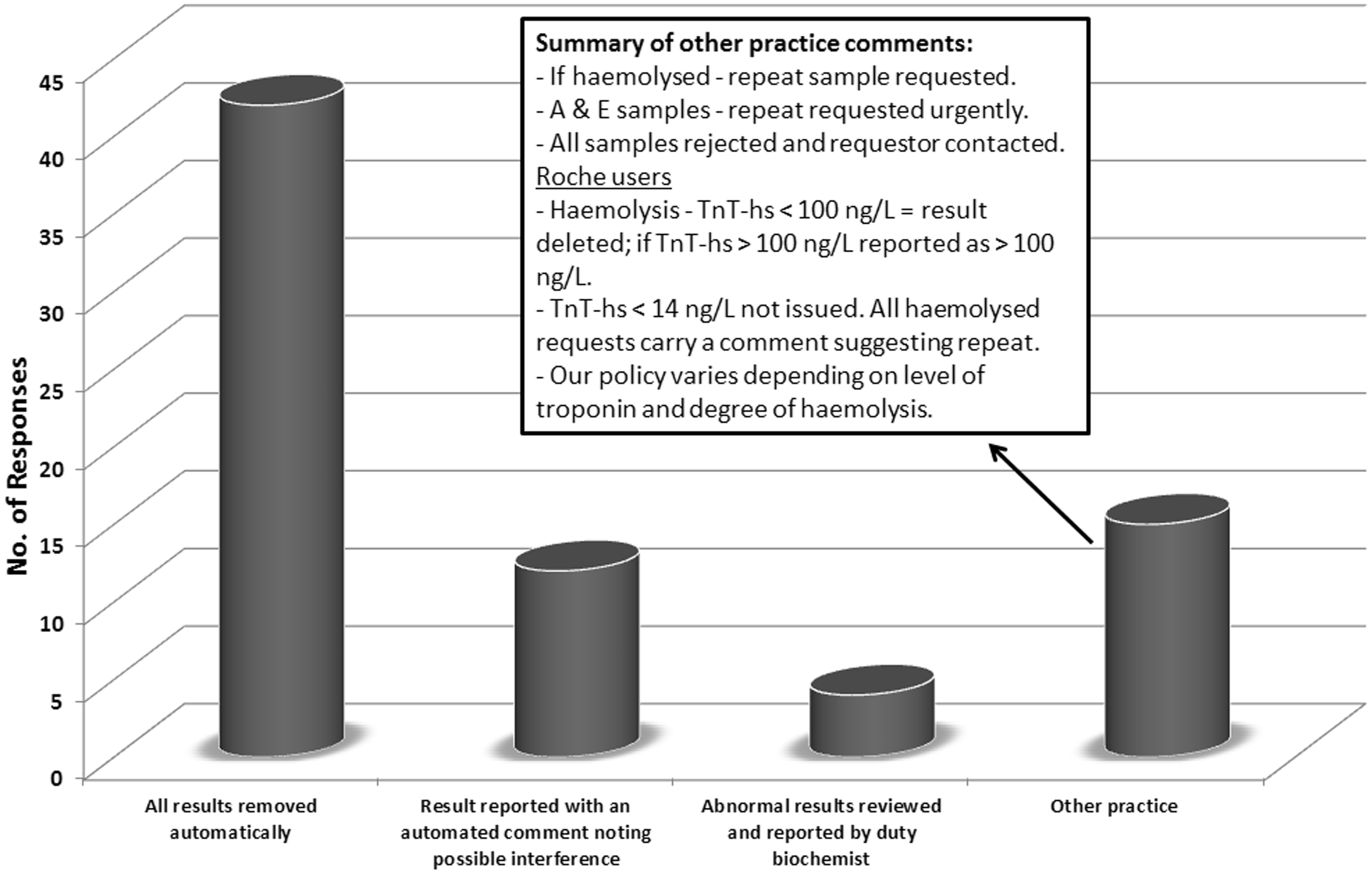
Summary of responses obtained for survey questions asking about reference change values, change criteria and critical limits quoted for each assay (number of responses in brackets).
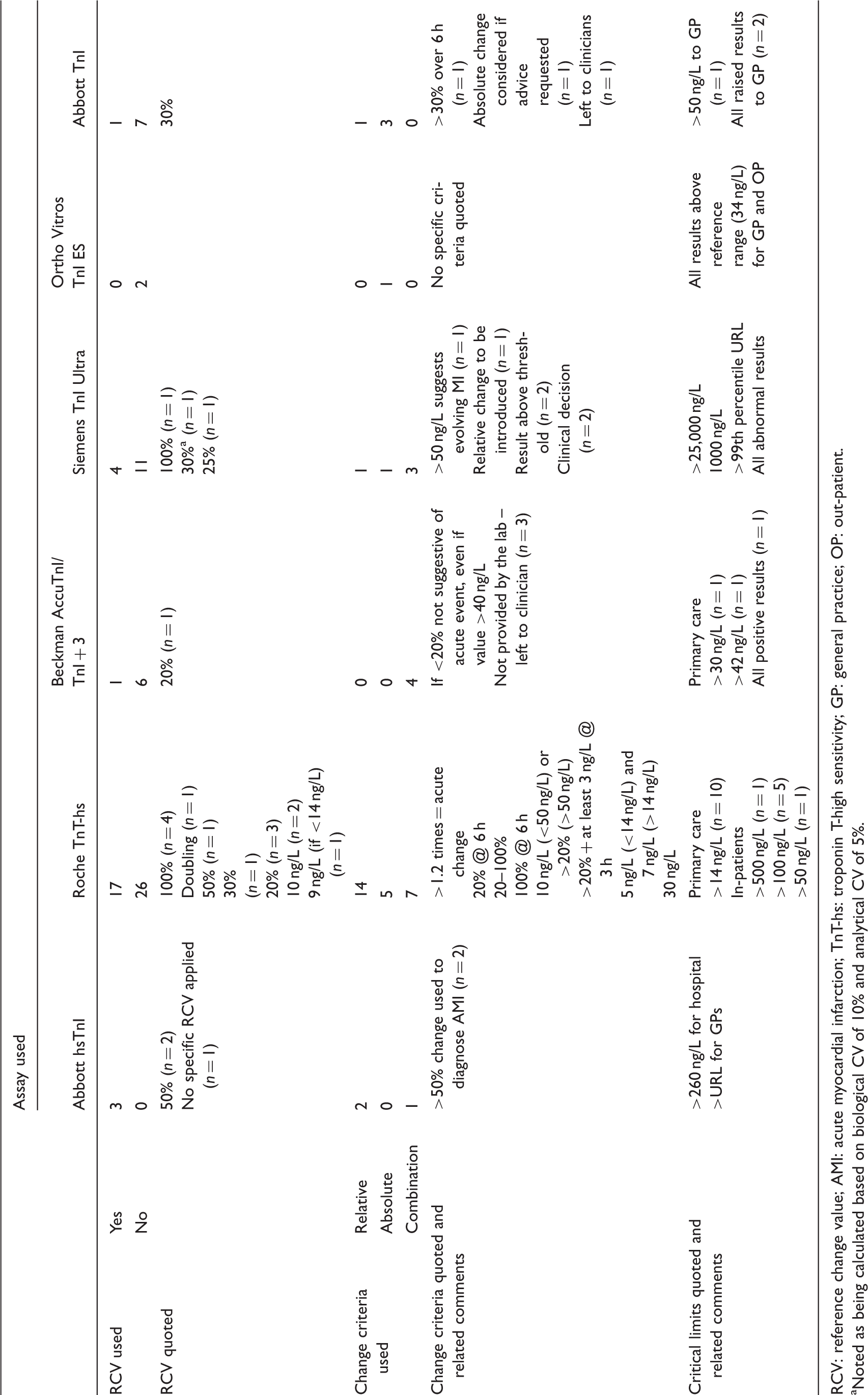
RCV: reference change value; AMI: acute myocardial infarction; TnT-hs: troponin T-high sensitivity; GP: general practice; OP: out-patient.
Noted as being calculated based on biological CV of 10% and analytical CV of 5%.
Workload, turnaround goals and point-of-care testing (POCT)
The majority of laboratories noted that troponin test numbers had increased after the introduction of an hs assay.
Laboratories were asked about their turnaround time (TAT) goal from sample receipt. The majority (n = 58) quoted the current Royal College of Pathologists (RCPath) key performance indicator (KPI) for TAT of <1 h from reception in the laboratory. A further 22 labs quoted <90 min to 2 h and some (n = 6) considered requests non-urgent and quoted a target of up to 6 h.
If individual laboratory services are unable to deliver troponin results within 1 h, then it has been suggested that a POCT option should be considered. 24 Laboratories were asked about the availability of POCT assays for cardiac markers on their hospital site. The majority of respondents (n = 69) had no POCT assays for cardiac markers available. When asked what cardiac markers additional to troponin I or T were provided by the laboratory brain natriuretic peptide (BNP) or N-terminal proBNP (NT-proBNP) was prevalent, a small number (n = 6) indicated they still had a creatine kinase-MB (CK-MB) assay in current use. One lab had an assay for heart fatty acid binding protein in operation, but no other new biomarkers were recorded.
Primary care
The survey asked how labs deal with troponin requests from primary care. The most common approach was to treat troponin samples sent from primary care as routine workload. A number noted the difficulty in blocking analysis of a sample once it arrives in the laboratory, indicating that this was a desirable option for them, and some explicitly stated that troponin testing was inappropriate for primary care. Others acknowledged that when presented with a patient with suspicion of acute coronary syndrome (ACS), the preferred course of action is assessment at an emergency department (ED), though there is a case for testing on patients in remote locations with mobility issues who are assessed as relatively low risk. Most laboratories noted that abnormal troponin concentrations (>99th percentile) would be communicated without delay.
Testing strategy
The admission sample is universally regarded as the baseline for assessment of dynamic changes in troponin. We asked what recommendations laboratories make regarding the collection of the second troponin sample to assess the likelihood of an acute event. This generated a variety of responses, illustrated in Figure 3. The majority of labs using an hs-troponin assay (n = 32) recommend resampling at ≥6 h from the admission sample, with only 12 quoting a shorter protocol. A number of respondents (n = 5) with a sensitive assay were still recommending waiting 12 h before collecting a second sample.
Summary of responses for timing of second sample and what assays are involved.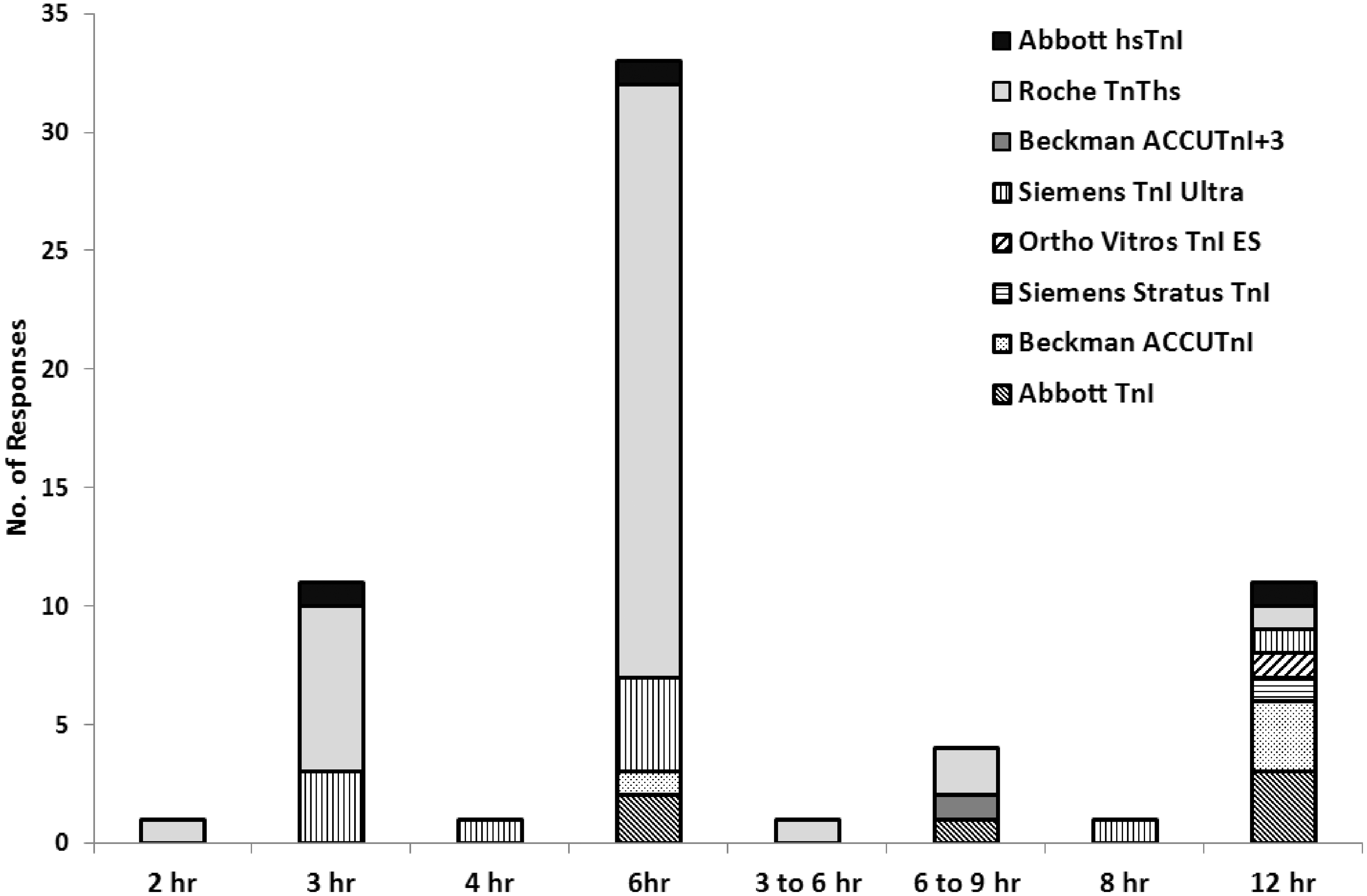
Interpretation
Thirty per cent of users quoted use of a reference change value (RCV), expressed as a percentage, to assess the clinical significance of a change in troponin concentration with time. 25 In addition, many use a relative ‘delta’ value (%) or an empirically derived absolute (concentration) change to differentiate acute from chronic causes of troponin elevation. This was a particular area of variation in practice even between those laboratories using the same troponin assay and analytical platform. A summary of responses to these questions is shown in Table 3.
Rule-out testing
Laboratories were asked if they quoted a troponin concentration for assisting with the rule out of ACS. The majority of respondents (n = 62) did not quote a decision limit for rule out. However, a number of Roche TnT-hs users (n = 7) quote admission values of <14 ng/L as ‘low risk’ or ‘negative’, whereas three labs quoted their lowest reportable value (LOD or LOB) as a rule-out value.
Reporting and education
The final part of the survey sought to identify what information laboratory services were providing to assist users with the application of troponin testing and the interpretation of results. A summary of responses is shown in Figure 4. In addition, a number of laboratories offer education on the appropriate use of troponin with 53% having guidelines available in their lab handbook or on a local intranet. A small number highlight this on their reports or refer users to a ward protocol. Twenty-seven per cent offer teaching sessions to medical and nursing staff, and 31% of those who responded were involved in clinical teams. Comments for this section noted that a number of labs have established links with their cardiology and EDs and have provided guidance and training to a number of different groups. Some have conducted clinical audits with cardiology teams and others noted that although they had no formal meeting arrangements, they would discuss individual cases as requested.
Summary of responses on report information, added comments or advice.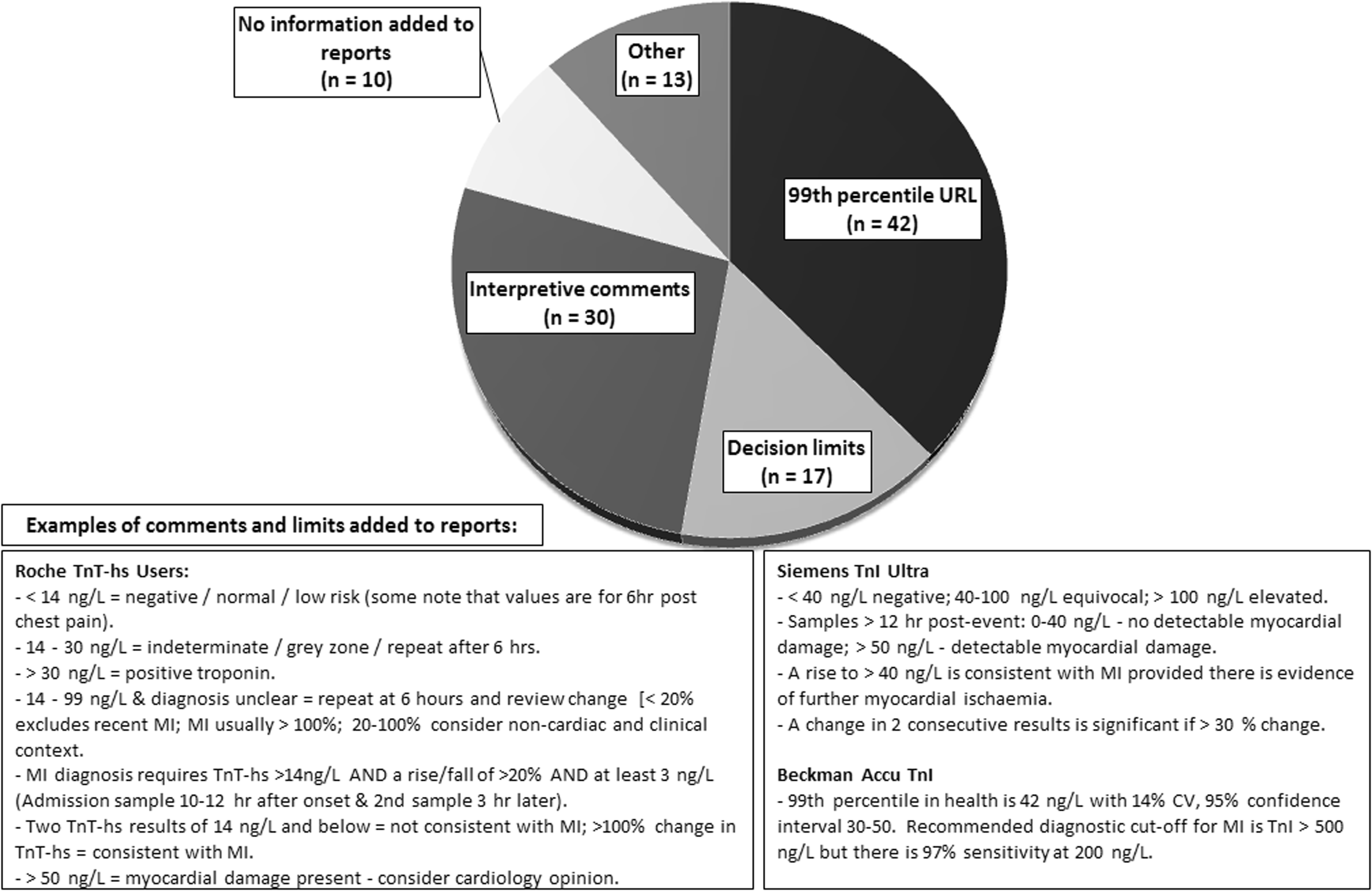
Discussion
Higher sensitivity varieties of troponin assays are now prevalent in our clinical biochemistry laboratories. The measurement of troponin is integral to the investigation of ACS, and although the diagnosis of AMI ultimately remains a clinical decision, 26 laboratories have a vital role to play in this decision-making process. A good understanding of assay characteristics is essential to the provision of sound guidance to clinical users on rational testing and interpretation of results.
This survey has highlighted that there is a significant degree of variation in how troponin assays are applied, even within laboratories operating within the same regional pathology network. It was evident from the survey responses that some of the sites within the same network were using the same assay, although other variations may be linked to different assay providers under separate managed service contracts. However, there remains scope for significant improvements and a more consistent evidence-based approach to the use of troponin. Based on survey responses and published evidence, we make a number of recommendations related to the key troponin themes reviewed.
Defining hs-troponin assays
Variations in the branding and marketing of the latest troponin assays have led to a lack of clarity regarding terminology and assay performance. The assay trend is towards hs and reliable quantitation of troponin in an ever greater proportion of the population distribution. This now includes the ability to quantify troponin in at least 50% of a healthy population. 12 It is evident that some confusion remains regarding how to define troponin assays and in particular when assays can be regarded as hs. The survey asked laboratories if their current troponin assay operated with a CV of ≤10% at the 99th percentile and so would be deemed as hs. In hindsight, users should have also been asked to indicate assay performance in line with the ability to detect troponin in greater than 50% of a healthy population (i.e. ≥Level 2 on the Apple Scorecard 5 ), which is now the accepted definition of an hs assay. Variations in the responses returned, and displayed in Figure 1, reflect the fact that a number of current troponin assays struggle to meet the imprecision standard. Some laboratories had conducted their own imprecision evaluations, whereas others had answered according to the quoted manufacturer data.
Table 2 summarizes the reporting limits and 99th percentiles quoted for the various assays from survey responses alongside published limits for each assay and how each is scored under the International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) and Apple criteria. Assays must be designated as Level 2 or above using the Apple scorecard to meet the current IFCC hs standard. The Abbott hsTnI assay easily meets this target. On the other hand, there have been varying reports for the Roche Cobas TnT-hs regarding the hs-level designation.6,15,27 The recent National Institute for Health and Care Excellence (NICE) diagnostics guidance
13
notes this assay as meeting the Level 2 designation. The same report also lists the Beckman Coulter Accu TnI + 3 as an hs assay, though no data are quoted for the troponin detection rate in a reference population. It is desirable that all laboratories providing a service to acute medicine should use an assay that approaches current hs criteria (≥Level 2). Recommendation 1: Where possible, laboratories should provide a hs assay that is designated as Level 2 or above on the Apple scorecard.5
Establishing assay parameters and quality assurance
There were variations in the various assay parameters quoted among users of the same troponin assay, particularly the 99th percentile, LOD, limit of quantitation (LOQ) and limit of blank (LOB). The LOB is the highest apparent analyte concentration expected to be found when replicates of a blank sample containing no analyte are tested. The LOD is the lowest analyte concentration that can be reliably distinguished from the LOB and the LOQ is the lowest concentration of analyte that can be quantitatively determined with acceptable imprecision and bias (goals predefined).18,28 It has been proposed that, to improve analytical robustness, the LOB of a troponin assay should be ∼5-fold lower than the 99th percentile limit calculated for the same assay. 18
Regardless of the assay in use, all laboratories should be fully aware of their own assay’s performance characteristics in order to provide appropriate guidance to service users. Within the Roche Cobas TnT-hs group, it is apparent that some use the LOD (5 ng/L) and others the LOB (3 ng/L) as the lowest reportable result. Consensus guidance when the Roche TnT-hs assay was first introduced recommended reporting TnT-hs values down to the LOB. It was thought that this could still provide useful clinical information despite poor imprecision at this low level.
10
This continues to be seen as a pragmatic approach for most laboratories. Recommendation 2: Laboratories should be familiar with assay performance including LOQ and assay designation.
The 99th percentile is the critical cutoff to aid the diagnosis of AMI, which should be quoted for all hs-troponin assays. There has been much discussion about how this should be established.29,30 Recent guidance produced by the IFCC Task Force on the clinical applications of cardiac biomarkers states that the 99th percentile for each assay could be obtained from either manufacturer information or peer-reviewed literature. 12 However, it is evident that various 99th percentile values have been quoted in the literature for the same assay in different studies (Table 2). The importance of population selection, which includes screening for underlying influential comorbidities, has been highlighted in a number of studies.15,16,29 Laboratories should carefully review the strength of evidence from manufacturer information and published data, for their particular assay to establish the 99th percentile to quote on their laboratory report. A value of 14 ng/L for TnT-hs (Roche Cobas) has been corroborated in a number of reference studies15,16 for a mixed population and has been adopted widely.
It is desirable that all laboratories verify assay imprecision at concentrations equal to and under the 99th percentile, although, our survey shows that very few labs currently do this, either formally or by running IQC at that level. The variation noted in the IQC protocols in use may reflect individual assay performance, since internal QC procedures should always be tailored to maximize error detection and minimize false rejection. Regardless, a target troponin concentration at or around the 99th percentile, using material from a third-party source, should be employed as a matter of routine.
31
Recommendation 3: Laboratories should use third-party IQC at or near the 99th percentile decision limit. Recommendation 4: Laboratories should verify assay imprecision at concentrations near the 99th percentile decision limit.
There was a general consensus among users regarding the reporting of hs-troponin concentrations as whole numbers (ng/L). The small number of laboratories using alternative units should aim to adopt ng/L for result reporting.
20
Recommendation 5: Laboratories should report hs-troponin as ng/L.
A number of studies have shown clear differences for both TnT and I concentrations between the male and female distributions.23,29 Despite this, and as reflected in the survey findings, only one assay manufacturer, at the time of survey, provides gender-related reference values. The risk of a misclassification error using a single cutoff has been highlighted.32,33 More manufacturers should be encouraged to produce gender-related data for their assays and significant differences between male and female reference limits should be identified so that users can take account.
Age-related reference values may also be important as the 99th percentile can differ significantly with age, particularly in the male population. 23 Therefore, the 99th percentile for the general population could be inappropriately low for older patients or even those with a history of coronary artery disease. Ideally, we require an age- and gender-related reference value for each assay although it would take significant resources to provide these data.
Sampling variables
The quality and type of blood sample may be of significance in the interpretation of results. Assays are now generally flexible in the samples that can be analysed; however, there may be small differences with regard to results obtained. Best practice would suggest that interpretation of serial troponin changes should be on a consistent sample type, as already implemented by some laboratories.
Haemolysis is a common interference encountered in samples sent for troponin analysis and has been shown to cause a negative interference in the Roche TnT-hs and Beckman Coulter Accu-TnI assays34,35 and cause falsely elevated troponin values in the Ortho Vitros TnI assay.
34
A number of laboratories surveyed do not report troponin results that breach the set haemolysis limit and many request an urgent repeat sample. Clearly, it is necessary to avoid reporting potentially erroneous results, however the risk of missing or delaying reporting a clinically significant result should be considered. It is possible to devise a sensible algorithm for reporting troponin results in the presence of haemolysis, similar to that put forward previously by Lyon et al.
36
where TnT-hs concentrations (>100 ng/L) in haemolysed samples are reported, with an appropriate comment added. Some sites have adopted such an approach, but any protocol devised should clearly take account of individual lab assay characteristics and be verified. Recommendation 6: Laboratories should have a protocol in place to deal with haemolysed samples.
TAT and POCT
Most labs surveyed are in line with the current recommendation of reporting results within 1 h from sample receipt. However, from April 2015, the RCPath recommendation is a more exacting 1 h from sample collection and the standard is currently set at 90% compliance.
37
This will represent a significant challenge for most laboratories. If labs are unable to deliver troponin results within 1 h from blood draw, then it has been suggested that a POCT option should be considered.
24
Few POCT troponin assays were noted to be in use across the sites surveyed and no current POCT TnT or I assays approach the analytical performance of the latest lab-based hs-troponin assays.
38
Nonetheless, laboratories should provide a suitable troponin testing service which may, of necessity, involve the use of alternative analytical platforms in order to process requests in the most timely manner. Recommendation 7: The laboratory troponin service performance should be assessed against current relevant professional quality assurance recommendations (key performance or assurance indicators).
Primary care and result communication
How laboratories deal with troponin requests from primary care is an area of interest and survey responses show variation in practice across different locations. Requests for troponin testing from the community should be restricted to circumstances where this is considered a necessary or prudent option as opposed to referral of the patient to the nearest Accident and ED. It has been suggested that the only appropriate use of troponin testing in general practice is for the patient presenting a significant number of hours or even days after chest pain. 39 Clearly, a symptomatic patient should never have referral delayed pending a troponin result. If it is considered necessary to rule out ACS for patients with mobility or ED access issues, or when a significant time has elapsed following onset of symptoms, provision should be made to enable timely analysis and telephone communication of any result meeting an agreed alert threshold.
A further KPI set by the RCPath requires urgent results to be communicated to a responsible clinician as soon as available, typically within 1 h.
37
There are variations across laboratories regarding the definition of a critical troponin result, which is related to the source of request. A number of respondents state that any result above the 99th percentile should be communicated urgently if received from primary care. The situation for secondary care is less clear cut where it may be considered prudent to differentiate between the need for direct urgent communication to busy acute units who are actively investigating and treating ACS patients and that for other sources. This is a matter for local agreement. Recommendation 8: Laboratories should have an agreed protocol in place for communication of results that require urgent action.
Testing strategy
The recommended time interval between serial samples varied considerably between laboratories. The enhanced analytical performance of latest troponin assays offers the opportunity to reduce the time required to detect a change. It is now evident that a clinically significant change can be reliably detected within a much shorter time span than the 12 h or more of previous assay generations. The question is, how far can this be taken? Guidelines produced by European Society of Cardiology state that patients presenting with suspicion of AMI should have samples collected on admission and 3 h later for hs-troponin analysis.17,24 This approach has also been validated in a number of other studies.40–42 Furthermore, the recent NICE diagnostics guidance notes that the Abbott hsTnI and Roche Elecsys TnT-hs assays are recommended for use with ‘early rule-out protocols’, which typically include a blood sample at initial assessment and a second blood sample taken after 3 h.
13
This would seem to be a sensible option for hs-troponin assays at present, although it is notable that even shorter intervals have been proposed in the literature. It remains important that any troponin change within 3 h is interpreted in the appropriate clinical context. Recommendation 9: A 3-h testing interval should be adopted for evaluating the initial hs-troponin change (delta) in the appropriate clinical context.
Interpretation
Those using a sensitive troponin assay continue to face the challenge of how to best differentiate those with an elevated troponin due to acute cardiac damage, requiring further urgent referral, from those patients with chronic elevations of troponin. Reference change and absolute change values have been advocated to improve specificity for detecting AMI and a diverse range of change criteria have been quoted in the literature. These are specific to individual assays and this area continues to evolve. This survey has highlighted that there remains significant variation in practice regarding the use of change values even among users of the same troponin assay. In hindsight, the questions relating to this section of the survey could have been better constructed as there may have been some misunderstanding regarding what was being asked. The use of a RCV, derived from biological and analytical variation data, can assist with rule out. Change limits should take account of RCV data available but can be set at a higher (delta) level to increase diagnostic specificity.
There is difficulty in quoting an optimal troponin delta for a particular assay that can be applied to all patients to assist with ruling-in ACS, as troponin changes can be influenced by presenting concentration, presenting population (i.e. cardiovascular disease (CVD) vs. healthy) and time window elapsed since chest pain onset. 42 However, laboratories should aim to provide guidance to users on when a troponin change becomes significant and highlight the importance of reviewing such changes in concert with the clinical context. One approach is to provide guidance on when a troponin change breaches that explained by biological and analytical variation alone, and so becomes potentially significant, by defining the assay RCV. A number of different RCVs, based on biological variation, have been quoted in the literature for hs-troponin assays and variations have largely been related to the different populations studied.43–46 Laboratories should assess analytical imprecision across different troponin concentrations and review RCV data published for a particular assay, along with imprecision data, as a first-level evaluation of the differences between two results.
To improve specificity, additional delta criteria could be employed to highlight those patients at higher risk. Initial guidelines for the Roche TnT-hs assay recommended using a 100% relative change in TnT-hs to help differentiate an TnT-hs increase caused by AMI and so reduce the number of nonacute referrals. 10 Subsequent studies have quoted relative change (%) values for different hs troponin assays ranging from 20% to 250%. 42 In our own lab, we quote an RCV of 20% and advise that any change above this is potentially clinically significant and the greater the delta percentage, the more significant it is likely to be. A 100% change has a positive predictive value approaching 100% for MI. 10
Recently, absolute changes in troponin (measured in ng/L) have been promoted as having superior diagnostic accuracy for the identification of MI.40,47 Absolute deltas of 5, 7 and 9.2 ng/L have been quoted for the Roche TnT-hs assay
42
and some have suggested a combined application of absolute and relative changes.
48
This is an area that is likely to progress and it has been noted that many of the change values published, based on receiver operating characteristic curve analysis, require prospective validation for each assay and population. It is imperative that labs highlight that serial troponin changes must always be viewed alongside the clinical picture. ACS is dynamic and evolving by definition and patients may present at any stage. There will be occasions when further blood sampling is required (e.g. at 6 h post admission) where the clinical picture remains unclear from first 3-h interval.
17
Recommendation 10: Laboratories should provide advice on the significance of incremental changes in serial troponin results based on RCV data and appropriate relative or absolute change values.
Rule-out testing
An area of significant interest, particularly for EDs, is how hs-troponin data can be reliably used to assist with ruling out AMI with a view to more efficient decision making. A number of Roche TnT-hs users quote values of <14 ng/L as ‘low risk’ or ‘negative’. Some studies have looked at using TnT-hs < 14 ng/L at presentation to assist with rule out,
49
although better negative predictive values for rule out of ACS are obtained using TnT-hs less than the LOD (5 ng/L)
50
or LOB (3 ng/L).
51
Despite the obvious attraction for a single TnT-hs cutoff to assist with AMI rule out, it is important that users remain alert to the risks of this approach, especially in patients presenting within the first 3 h of chest pain.
50
Laboratories should be cautious about quoting TnT-hs results as normal, negative or low risk without linking advice to users about the importance of considering the full clinical picture. In the appropriate clinical setting, low or undetectable hs-troponin concentrations at presentation could be used to assist rule out, but in the absence of suitable clinical information it is prudent, if not essential, to follow-up with a second sample. If the second result remains under the 99th percentile, the negative predictive value (NPV) approaches 100%.
24
In our lab, this has been highlighted by two individual cases where the TnT-hs at presentation was <3 ng/L, but significantly increased at 3 h above 14 ng/L. In both cases, the patient presented early to the ED and was eventually diagnosed with having an AMI (personal observation). The recently published NICE diagnostics guidance on the use of hs-troponin assays for early rule out of AMI has concluded that although the LOB and LOD show promise as cutoff thresholds in early rule-out strategies, the implications of introducing these into practice are currently uncertain. Recommendation 11: Laboratories should be cautious about quoting hs-troponin ranges as ‘normal’ or ‘low risk’ without appropriate clinical context.
Education and guidance
It was evident from the survey responses that many laboratory services have good links with clinical colleagues and provide guidance on how to interpret troponin results. However, in some cases, there is still much uncertainty over troponin results from more sensitive assays and how results can be applied in particular clinical pathways. Laboratory professionals should be encouraged to continue to foster close links with clinical colleagues. There is still a steep learning curve and the lab has an important role to play regarding assay application. It is crucial that there is adequate training and advice available for new members of medical staff who may have previously worked in an area using a different troponin assay. A number of laboratories run teaching sessions for medical and nursing staff and this is to be encouraged. Recommendation 12: There should be continuous dialogue between laboratory professionals and clinical colleagues regarding the appropriate use of their troponin assay.
Conclusions
Our survey has highlighted the significant variation in approach to troponin testing across the Biochemistry Services of the UK and Ireland. Similar concerns were noted following a European Pilot Survey in 2006 and in follow-up ‘letters to the Editor’,52–54 which questioned the persistence of nonevidence-based behaviour prevalent in many clinical laboratory services and a failure to engage effectively with their clinicians.
List of recommendations based on survey responses and published evidence.

* Level 2 as defined by the Apple scorecard.5 LOQ: limit of quantitation; IQC: internal quality control; RCV: reference change value.
Footnotes
Acknowledgements
We thank the members of the Northern Ireland Regional Audit Group and NCBAG for their input into the survey questionnaire. We would also like to thank Tom Hutchinson for his assistance in organizing circulation of the survey, analysing coverage and coordinating responses. We also thank all those who took time to complete the questionnaire.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Ethical approval
Not applicable.
Guarantor
PA.
Contributorship
GM and PA researched literature, conceived the idea for the survey and developed the survey questionnaire. Input was obtained from the Northern Ireland Regional Audit Group and the National Clinical Biochemistry Audit Group (NCBAG). GM wrote the first draft of the manuscript. Both authors reviewed and edited the manuscript and approved the final version.

