Abstract
Background
Little is known of the vitamin D status of young infants and toddlers and its determinants in West Europe. The prevalence and determinants of vitamin D deficiency of children aged 6–48 months in the centre of the Netherlands (52°N) is investigated.
Methods
In a cross-sectional population study, randomly recruited infants and toddlers (n = 150) were studied using an online questionnaire and a physical examination either in late summer (n = 52) or in late winter (n = 98). Vitamin D analysis was performed by capillary blood sampling using dried bloodspots plus LC-MS/MS.
Results
In late winter, 32% of the children were vitamin D deficient (<50 nmol/L 25OH vitamin D3) with 5% severely deficient (<25 nmol/L). In late summer, 2% were deficient. The odds of vitamin D deficiency were higher in children aged 24–48 months, for those not using formula milk and those not adhering to the supplementation guidelines.
Conclusion
One-third of Dutch infants and toddlers were found to be vitamin D deficient in late winter. Suggested strategies for raising the vitamin D status may include improving the adherence to supplementation, a sensible sun exposure or the use of fortified foods. Special attention is needed for the children aged 24–48 months.
Keywords
Introduction
In the past decades, vitamin D deficiency seems to have become pandemic at all ages and the numbers of laboratory request for 25OH-vitamin D have increased enormously in many countries. Part of these requests is questionable 1 and the discussion when supplementation is needed has not ended yet. In children, vitamin D deficiency has re-emerged as an important clinical theme.2–4 The prevalence of vitamin D deficiency in Europe in infants and toddlers (<48 months) varies from 8 to 45%.4–8 Although symptoms of rickets are rarely seen in Western societies, 2 deficiencies are also associated with muscle weakness and hypotonia, decreased immune response, upper respiratory infection risk and other non-skeletal disorders of childhood.3,4
Vitamin D status is determined mainly by sun exposure or skin colour, and less by consumption of (fortified) foods and supplements.3,4 We point out that food in general, including cow's milk, is not fortified in the Netherlands. To our knowledge, no studies have been performed to assess the contribution of each determinant simultaneously in one single group of small infants and toddlers. We investigated the prevalence and determinants of vitamin D deficiency in the general Dutch population for children aged 6–48 months (the kiDs study).
Study design
Study population
The kiDs study was conducted in the central part of the Netherlands among infants and toddlers 6–48 months old who visited one of the Public Health Services in the region Utrecht (52° N). Our main study was to include 600–800 children (6–48 months) with half of them of non-Western origin. Here we present a pilot study of 150 infants. A random sample of 145 children in late summer and 392 children in late winter was invited from the registries of the Department of Youth Health Care of the Public Health Services. Exclusion criteria were: malabsorption due to celiac disease, cystic fibrosis or inflammatory bowel disease, chronic liver or kidney disease, use of medication that affects vitamin D metabolism, contraindications for blood drawing, lowered immune status, e.g. HIV/AIDS or tuberculosis, osteogenesis imperfecta or other rare genetic bone disease. This study was conducted according to the Declaration of Helsinki (2013) 9 and approved by the local Medical Ethics Committee. Written informed consent was obtained from both parents of all participants. For determining the prevalence of deficiency, our study was powered at 150–400 inclusions.
Measurements
The study consisted of an online questionnaire, a physical examination by a trained physician and capillary blood sampling using a fingerprick and bloodspot sample collection.
Because of the reported season effect8,10 in comparable countries, vitamin D status was measured in late summer (September/October) and in late winter (February/March). Regarding possible effects of raising awareness by the study information given, the blood sample was generally taken within three weeks after the invitation.
Vitamin D assessment
To avoid phlebotomy, we used dried blood spot analyses by LC-MS/MS performed by DBS Laboratories Geleen, the Netherlands. The measurement procedure is based on Eyles’ method 11 employing UHPLC using an Agilent 6460C triple-quadrupole MS/MS. Bloodspot punches are extracted with an alkaline surfactant solution, allowing release of 25OHD3 from D-binding protein, followed by hexane extraction and derivatization prior to injection. 11 Our method was calibrated with a panel of serum calibrators provided by LabQuality (Helsinki, Finland) as proposed by the Vitamin D Standardization Programme. 12 Inter-assay imprecision was calculated at three levels: coefficients of variation were 9.8% at 48.3 nmol/L, 8.7% at 76.4 and 7.6% at 139.2 nmol/L. Mean bias to an LC-MS/MS method13 serving as reference was 3.0 nmol/L.
Physical examination
A specially trained physician performed the fingerprick and an extensive physical examination. Information on height and weight was obtained from the patient information system of the Public Health Services. The height, weight, weight for height and BMI were transformed to age-specific Z scores for Dutch children. 14
Online questionnaire
Using the SurveyMonkey® software, parents filled out a digital questionnaire regarding demographic characteristics (age, gender, ethnic background, parents’ educational level), exposure to sunlight, skin type (modified Fitzpatrick Skin Type Scale), and child's diet and intake of dietary supplements. We used an extensive food frequency questionnaire that we designed for assessing the vitamin D and calcium intake from diet and supplementation, since a validated questionnaire suitable for Dutch babies and toddlers does not exist. Where needed, the questionnaires were completed by telephone interview. Vitamin D content of diet was calculated as an average intake per day.
As the original Fitzpatrick skin types 15 include items related to sun exposure, and many parents reported no sun exposure (yet), we used a modified version of the Fitzpatrick score, meaning that for those children who had not (yet) been in the sun, the score was solely based on the colour of hair, eyes and skin of the child.
Definition of deficiency
No international consensus on the definition of vitamin D deficiency in children exists yet. The ESPGHAN Committee of Nutrition, 4 the American Academy of Pediatrics, 16 the Institute of Medicine 17 and the German Nutritional Society 18 define <50 nmol/L deficient. Therefore we used the definitions: <50 nmol/L deficient and <25 nmol/L severely deficient.
Statistical analyses
Statistical analyses were performed using SPSS (version 22.0, IBM, Chicago). Participants’ characteristics were presented by season with means plus standard deviation (SD) for continuous variables, and with percentages for categorical variables. Overall prevalence of deficiency was calculated as weighted average of the season-specific prevalence of deficiency. Prevalence by age group was also weighted by size of the subpopulations in each season. Linear regression analysis was used to estimate the relative effects of vitamin D intake through diet and supplementation on vitamin D level.
Logistic regression was used to estimate odds ratios (ORs) and 95% confidence intervals (CI) of association with vitamin D deficiency of potential determinants like season, skin type, formula milk and supplementation.
To evaluate the association of vitamin D deficiency with growth and weight, we calculated Z-scores by age and sex for height, weight, height for weight and BMI, using the national growth data. We then categorized children into low (at least one SD below the mean), average and high (at least one SD above the mean). These categories were used in a logistic regression model to estimate ORs and 95% CI for vitamin D deficiency. Associations calculated were either crude (model 1), adjusted for age and sex (model 2) or additionally adjusted for the other determinants (model 3). Missing data on vitamin D status were reason for exclusion, participants with missing data on all questions (no questionnaire filled out) were only included in the crude prevalence rate. Missing data on dietary intake variables were assumed to indicate non-exposure and were set to 0. Missing data on all other variables were not addressed.
Results
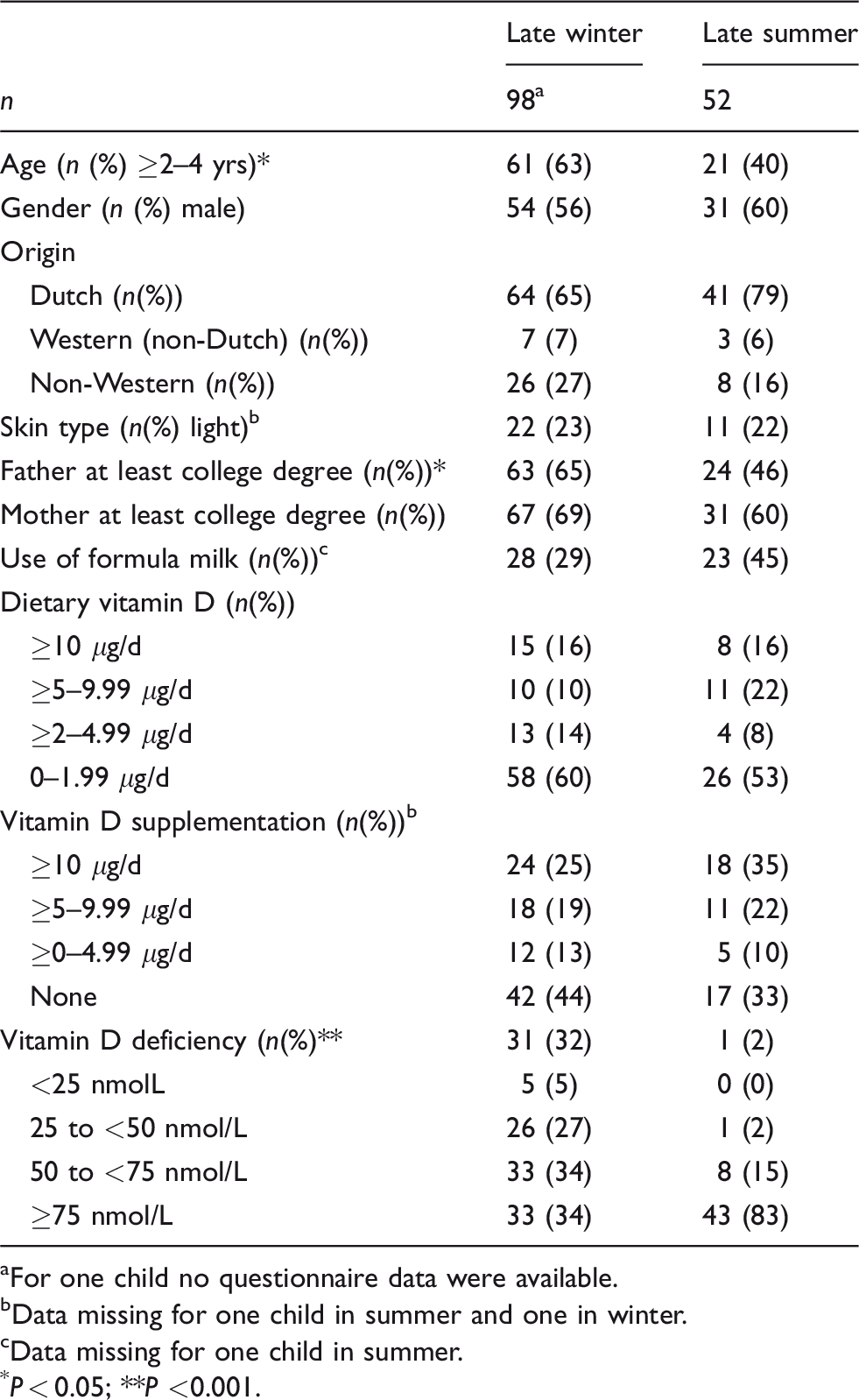
A total of 150 children were included in this pilot study: 52 children in late summer and 98 in late winter. The response rates were 35% in summer and 25% in winter. For 20% of the non-respondents, reasons for refusal were available. Most frequent reasons for refusal were ‘no time’ for measurements (around 50%), or rejection of the finger prick needed (around 25%). Non-participants for whom we had data were similar to participants with regard to age and parental education, non-participants were more frequently of Dutch origin.
The characteristics of the participants are presented in Table 1. Age ranged from 6 to 48 months both in summer and winter and averaged 28 months. More children of non-Western origin participated in winter (27%) than in summer (16%). No differences were seen in the distribution of skin types between winter and summer. In late summer, 2% of the children were deficient, while in late winter, 32% of the children were deficient and 5% severely deficient.
Population characteristics by season.
For one child no questionnaire data were available.
Data missing for one child in summer and one in winter.
Data missing for one child in summer.
P < 0.05; **P <0.001.
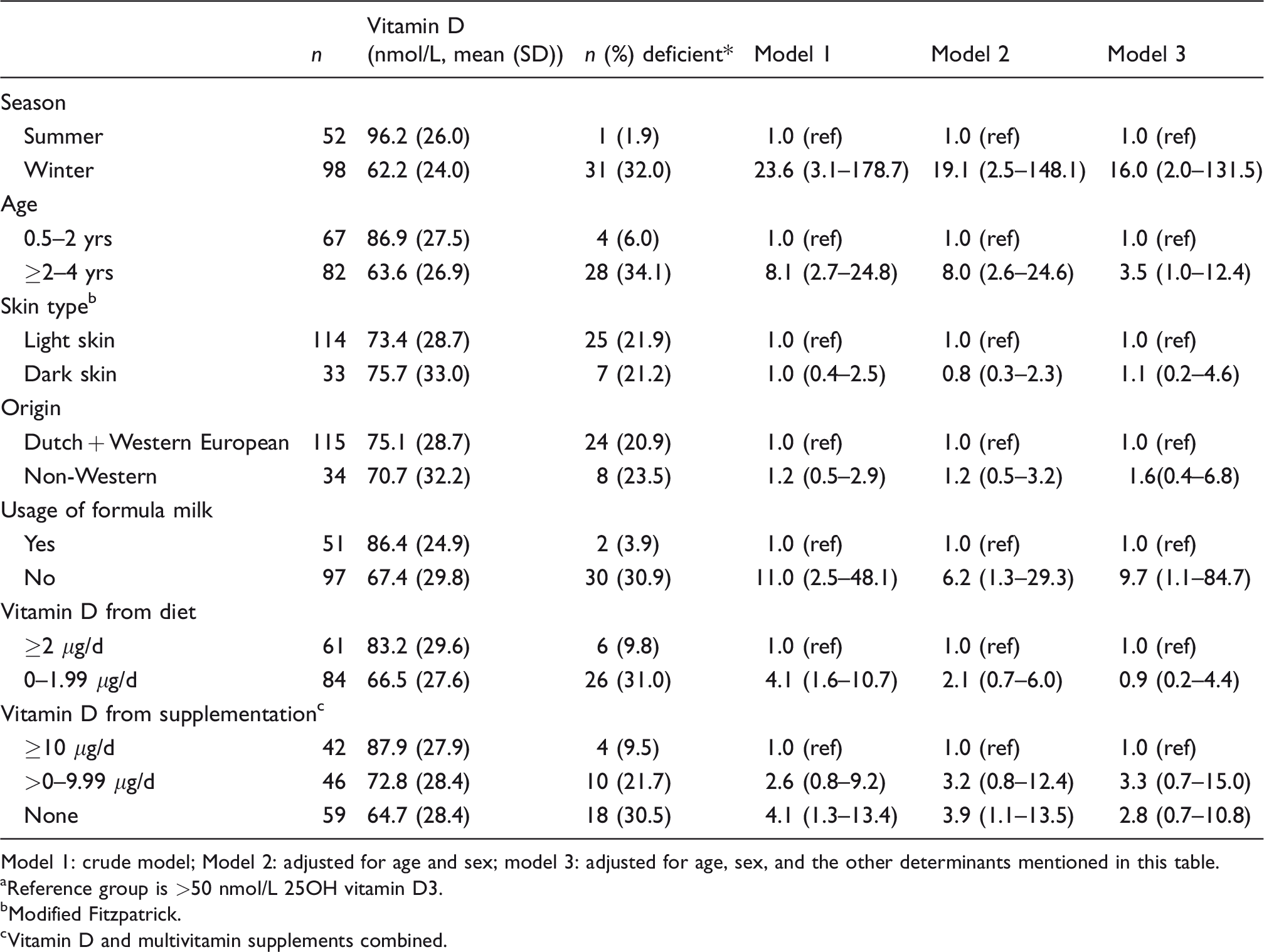
Table 2 shows the vitamin D status and ORs (95% CI) of (severe) vitamin D deficiency by season, age, skin type, origin, usage of formula milk and vitamin D from diet and supplementation. Mean vitamin D levels were 96.2 nmol/L in late summer and 62.2 nmol/L in late winter.
Vitamin D status and ORs (95% CI) of vitamin D deficiency a by season, skin colour, usage of formula milk and supplementation.
Model 1: crude model; Model 2: adjusted for age and sex; model 3: adjusted for age, sex, and the other determinants mentioned in this table.
Reference group is >50 nmol/L 25OH vitamin D3.
Modified Fitzpatrick.
Vitamin D and multivitamin supplements combined.
In winter, children had a 16 times higher odds of vitamin D deficiency than in summer and children aged 2–4 years had a 3.5 times higher odds than younger children. Not using formula milk resulted in a 10 times higher odds and not adhering to supplementation guidelines (67%) in a 3 times higher odds, irrespective of the dose.
In winter, vitamin D status was directly related to vitamin D intake from diet and supplementation. After adjustment for age, sun exposure and season, each μg of vitamin D from the diet contributed to 0.14 nmol/L higher 25OH vitamin D3 level (P = 0.04) and each μg of vitamin D from supplementation contributed to 0.51 nmol/L higher 25OH vitamin D3 level (P = 0.003). We found no association between a dark skin or non-Western European origin with the vitamin D status of these young children. It might be that our sample size was too small to detect a possible existing association.
We studied the association of weight and height with vitamin D status. Children with high Z-scores for both were more likely to be deficient than those with low or average scores, after correction for age and season (high weight OR = 1.6, 95% CI: 1.1–2.5; high weight for height OR = 1.5. 95% CI: 1.0–2.3). The difference in Z-scores between vitamin D deficient and sufficient children was 0.27 (SE 0.13, P < 0.05) for weight and 0.46 (SE 0.26, P = 0.07) for weight for height, also corrected for age and season. Height was not associated with vitamin D deficiency, nor with absolute vitamin D level.
Discussion
Determinants of vitamin D deficiency for children aged 6–48 months were winter, age ≥2 years, no formula feeding, no vitamin D supplementation and overweight. The prevalence of vitamin D deficiency found for late summer (2%) and late winter (32%) in Dutch children <48 months is comparable to the annual average of 22.8% deficiency (<50 nmol/L) in West European children aged 12–36 months. 7 This season effect was reported previously by others4,8,10,19 using non-standardized immunoassays. Vitamin D status was directly related to vitamin D intake from diet and supplementation, also after adjustment for relevant factors such as season.
The lower vitamin D status for the 24–48 months aged children compared with <24 months children is partly explained by a lower intake of vitamin D from fortified formula feeding and a lower adherence to supplementation. When only adjusting for gender, older children had an eight times higher odds of deficiency, and when additionally adjusting for adherence to the guidelines and usage of formula milk the older children had a 4.1 times higher odds (95%CI:1.2–13.7). It should be noticed that there were more young children in summer and more older children in winter (Table 1) and season is the strongest determinant of deficiency. After adjustment of all potential determinants – including season – the older children had a 3.5 times higher odds (95%CI: 1.0–12.4) compared with younger children. Our study confirms a German study of declining vitamin D status after about two years of age. 6 This determinant is not addressed so far in governmental or medical guidelines. Only 33% of the children in our study were supplemented with the Dutch recommended 10 μg vitamin D daily and 36% used no form of supplementation at all (Table 2). Previous studies are inconsistent concerning the association between vitamin D status and weight status under four years. Our study and others show an inverse association between vitamin D status and weight for height in this age group.20,21
Improving the vitamin D status of infants aged 6–48 months demands a careful consideration of all determinants. The major determinants described are sun exposure, intake of fortified food and vitamin supplements,8,10 similar as what we found in our study. However, sun exposure may be limited by discouragement in public campaigns. Intake of non-fortified cow’s milk was reported to be associated with a higher prevalence of vitamin D deficiency. 7 Increasing the vitamin D intake by using vitamin supplements and fortified food have been demonstrated and advocated to be beneficial.4,16,19,22–25 Sun exposure is an item under fierce debate, given the long-term effects of early sun exposure. Still, a sensible exposure could be of benefit.
Strengths and limitations
Although a pilot study, its major strength is the extensive data collection focussed on determinants of vitamin D status with emphasis on dietary intake of vitamin D in children from the general population aged between 6 and 48 months. Our sample size of 150 children was large enough to show significant associations of several determinants with vitamin D status. However, it did not allow us to do stratified analyses by season or other subgroup analyses. The response rates were 35% in summer and 25% in winter. Despite our aim to sample a group representative for the general population of infants and toddlers, the response of parents with a higher level of education was higher. Since a positive association has been reported between higher socio-economic status and micronutrient intake including vitamin D, 26 the true prevalence of vitamin D deficiency might be even higher than shown in our pilot study. Finally, due to the cross-sectional nature of our study, we cannot conclude causality.
Conclusion
One-third of Dutch children in the age up to four years seem to be vitamin D deficient during winter. This might be explained by the low adherence (33%) to the advised daily supplementation of 10 μg. Determinants of vitamin D deficiency in infants and toddlers were winter, age over two years, no formula feeding, no or low vitamin D supplementation and overweight. Strategies for raising the vitamin D status for this group could be (improving adherence to) supplementation recommendation, fortifying food like cow’s milk in countries where this is not done at present, and advising sensible sun exposure.
Footnotes
Acknowledgements
We regret the death of one co-author, Henk Groeneveld, staff member of the Public Health Services region Utrecht. We thank all parents, children and the Department of Youth Health Care of the Public Health Services region Utrecht (director: Henk Kruisselbrink), Jack van der Heyden, BSc (DBS Laboratories Geleen), Wieteke Spinetto-Rubbens, MSc, Joanne Zwart, MSc, Elma Moons, Janneke Smit-van de Veen, Désirée Hennig, Ramon Mensink, Jaap Veerkamp and Wil Hogeman for all their support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The kiDs study was sponsored by Nutricia, the Netherlands. Nutricia had no influence in any way on the data collection, analysis, interpretation and presentation of the results of the kiDs study.
Ethical approval
Ethical approval was provided by the VCMO, with reference number NL48007.100.14.
Guarantor
JW.
Contributorship
MH-B wrote the protocol, coordinated the study, analysed data and co-wrote the manuscript. JW conceived the idea for the study together with PH, coordinated the vitamin D analysis, helped interpret the data and co-wrote the manuscript. HG helped set up the study, commented on the manuscript (HG died in 2018). EdL helped set up the study, helped interpret the data, commented on the manuscript. RS did the examinations in the summer, helped interpret the data. CP did the examinations in the winter, helped interpret the data. PP-dJ helped set up the study, analysed the data, commented the manuscript. PH conceived the idea for the study together with JW, did the measurements of the deficient children, helped interpret the data and helped writing the manuscript.
