Abstract
Background
The measurement of plasma renin activity using LC-MS/MS is an attractive alternative to radioimmunoassay techniques. A published method for renin activity by LC-MS/MS uses equipment that is not available in all LC-MS/MS laboratories. Here, we present an offline modification to allow users of LC-MS/MS to perform this analysis without specialist equipment.
Methods
Samples were prepared in duplicate and incubated for 6.5 h and 24 h. Solid phase extraction was performed offline using Waters Oasis MAX ion-exchange 96-well plate. A method comparison was performed between this assay and a previously published assay using on-line solid phase extraction.
Results
The offline method for plasma rennin activity gave similar results to the on-line method across the concentration range. Analytical performance was also found to be comparable and the use of a 24 h incubation for low samples was also deemed unnecessary.
Discussion
The method described is an acceptable alternative to a previously published assay which does not require the use of highly specialist equipment and will be applicable to the majority of LC-MS/MS users.
Introduction
The measurement of plasma renin activity (PRA) using LC-MS/MS as an alternative to radioimmunoassay techniques is often favourable due to decreased safety risks and a more rapid turnaround of samples. A previously published method demonstrates that the measurement of PRA can be achieved using LC-MS/MS; however, this on-line solid phase extraction (SPE) method uses equipment that is not available in all laboratories. 1 We present an offline SPE alternative to allow users of LC-MS/MS to perform this analysis without specialist equipment.
Materials and methods
During routine use the PRA results from the 6.5 h and the 24 h plates were similar, questioning whether the 24 h plate was required. To determine if the 24 h plate is required to improve assay sensitivity with the on-line sample preparation, results (
Plasma samples (
Inter-assay imprecision was assessed using Bio-Rad Hypertension quality control material (Bio-Rad, Hemel Hempsted, UK).
Results
When comparing PRA results below 0.3 nmol/L/h between the 6.5 h and 24 h incubation plates, the Passing–Bablok analysis showed: 24 h plate = 0.94 × 6.5 h plate − 0.03 nmol/L/h. The Bland–Altman bias plot is shown in Figure 1. This confirmed that the results were comparable in this low activity range, deeming the 24 h incubation unnecessary. Subsequent comparisons were performed using the 6.5 h plates only and results below the lower limit of quantitation were not included in the analysis.
Difference between low renin activities obtained after either a 6.5 h or 24 h incubation.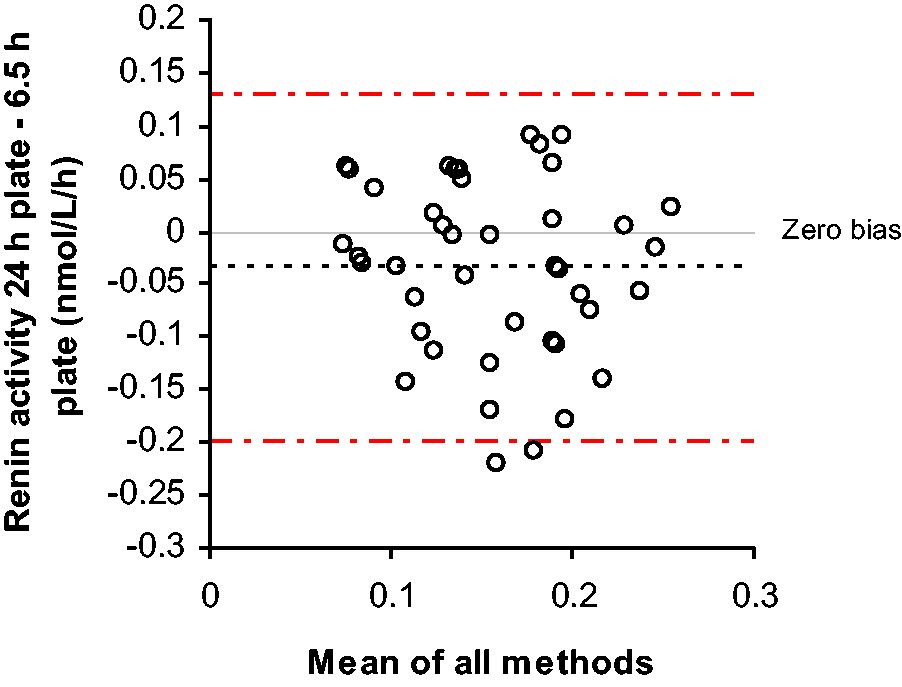
The results from the offline preparation described above were compared to the original method of Carter
The imprecision observed with the new sample preparation was noted to be superior to the on-line assay. The offline assay gave CVs of 7.5, 6.7 and 6.0% at plasma renin activities of 1.20, 2.98 and 9.86 nmol/L/h while the on-line assay gave CVs of 12.5, 6.6 and 7.4% at similar activities. This improved precision was also demonstrated by a superior lower limit of quantitation of 0.05 nmol/L/h.
Conclusions
We have described an alternative sample preparation for PRA analysis which does not require expensive on-line SPE equipment. The offline technique does require some amount of extra time for the preparation of the samples by using a positive pressure manifold for the SPE. However, the time required to prepare and prime the on-line SPE equipment has also been removed, thus almost neutralizing the time required for the offline extraction. The assay described here shows superior precision to the on-line method and also removes the need for a 24 h incubation, therefore reducing the turnaround times for this analysis. The lower limit of quantitation of the previous assay was 0.1 nmol/L/h using the 24 h incubation; this assay shows no decline in performance at these low concentrations without a 24 h incubation (Figure 1). The type of analysis described here can be applied to measure renin activity in most LC-MS/MS laboratories.
Footnotes
Acknowledgements
None declared.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Not applicable.
Guarantor
LJO.
Contributorship
The development of the assay described was performed by LJO and JA. Validation was performed by KM and SL. All authors have contributed to the manuscript.
