Abstract
Background
Classical and 11-oxygenated androgens both contribute to the androgen pool. Regular monitoring of the androgen status is required in disorders of steroidogenesis, and multiplexing of androgens improves the diagnostic ability of an assay. Due to the cheap non-invasive collection, saliva is advantageous when multiple samples are required. Liquid chromatography-tandem mass spectrometry (LC-MS/MS) offers sensitive, simultaneous quantification of steroids with short run times. Here, we have developed an LC-MS/MS assay for the simultaneous measurement of 17-hydroxyprogesterone, androstenedione, testosterone, 11β-hydroxyandrostenedione and 11-ketotestosterone in saliva.
Methods
Samples (300 μL unstimulated whole saliva) were prepared by supported liquid extraction with dichloromethane and were reconstituted in 40% methanol. After online solid phase extraction with C18 cartridges, liquid chromatography was performed on a C8 column using a water/methanol gradient containing 0.1% formic acid and 2 mmol/L ammonium acetate. A Waters TQ-S mass spectrometer was used for quantification.
Results
Total run time was 6.4 min. For all analytes, recovery was between 89% and 109%, ion suppression between 86% and 105%. Intra- and inter-assay comparisons showed a coefficient of variation <10% and the bias between measured and nominal concentration varied between –8% and 10%. Interference with a large set of natural and synthetic steroids was excluded. The assay was applied for the measurement of the androgen profile in healthy men (n = 17) and women (n = 10) which confirmed the sensitivity of the assay to be appropriate.
Conclusion
We present a novel LC-MS/MS assay for the comprehensive profiling of classical and 11-oxygenated androgens with potential for routine clinical application.
Keywords
Introduction
The determination of androgen status in female and male patients with disorders of steroidogenesis, in particular in congenital adrenal hyperplasia (CAH), is essential to assess the quality of their treatment and to stratify their androgen-related cardio-metabolic risk. However, adrenal and gonadal androgen biosyntheses are complex, and there is extensive peripheral activation of androgen precursors. 1 Multiplexing of analytes is therefore required to improve the diagnostic ability of an androgen assay.2–4 The simultaneous measurement of the classical androgen testosterone and its precursor androstenedione in serum or saliva has previously been established as a sensitive marker for metabolic risk in polycystic ovary syndrome (PCOS), a syndrome of androgen excess in women.2,5–7
Additionally, recent research on androgen biosynthesis pathways has identified the class of 11-oxygenated androgens as a significant driver of androgenic activity in addition to the classical androgens like testosterone. 8 The 11-oxygenated androgen pathway is initiated by adrenal conversion of androstenedione to 11β-hydroxyandrostenedione (11OHA4), a major adrenal androgen precursor. 9 11OHA4 can be further converted to 11-ketotestosterone (11KT), which activates the androgen receptor with the same potency and efficacy as testosterone.10,11 In both common (PCOS) and rare (CAH) androgen excess conditions, 11-oxygenated androgens make the major contribution to the circulating androgen pool.12–15 It is therefore essential to include these analytes in the routine assessment of androgen status. Treatment monitoring in CAH requires regular and frequent sample collections and analysis. The collection of saliva for steroid analysis represents an excellent option for this, as it can be conducted by the patients on their own and at home, without the need for clinic attendance or support by clinical personnel. Additionally, a significant proportion of androgens in circulation is bound to sex-hormone binding globulin and therefore considered bio-inactive according to the free hormone hypothesis. 16 Concentrations in saliva, however, represent the free, unbound fraction of the hormone and more likely reflect bioactive concentrations. 17 Liquid chromatography-tandem mass spectrometry (LC-MS/MS) is currently the tool of choice for steroid analysis due to its outstanding selectivity and short run times.18,19 Additionally, LC-MS/MS has the sensitivity to accurately quantify low analyte concentrations, as are expected in saliva, and allows for multiplexing of analytes of interest.
We, therefore, developed and validated an LC-MS/MS assay for the simultaneous measurement of the classical androgens testosterone and androstenedione and the 11-oxygenated androgens 11OHA4 and 11KT. Additionally, the assay includes 17-hydroxyprogesterone (17OHP), which is the accepted reference standard for the diagnosis and monitoring of treatment efficiency in CAH.20,21 The structures of the analytes are shown in Figure 1. This specific combination of analytes in the presented assay can comprehensively support the assessment of androgen status in both female and male patients with disorders of androgen excess and deficiency.
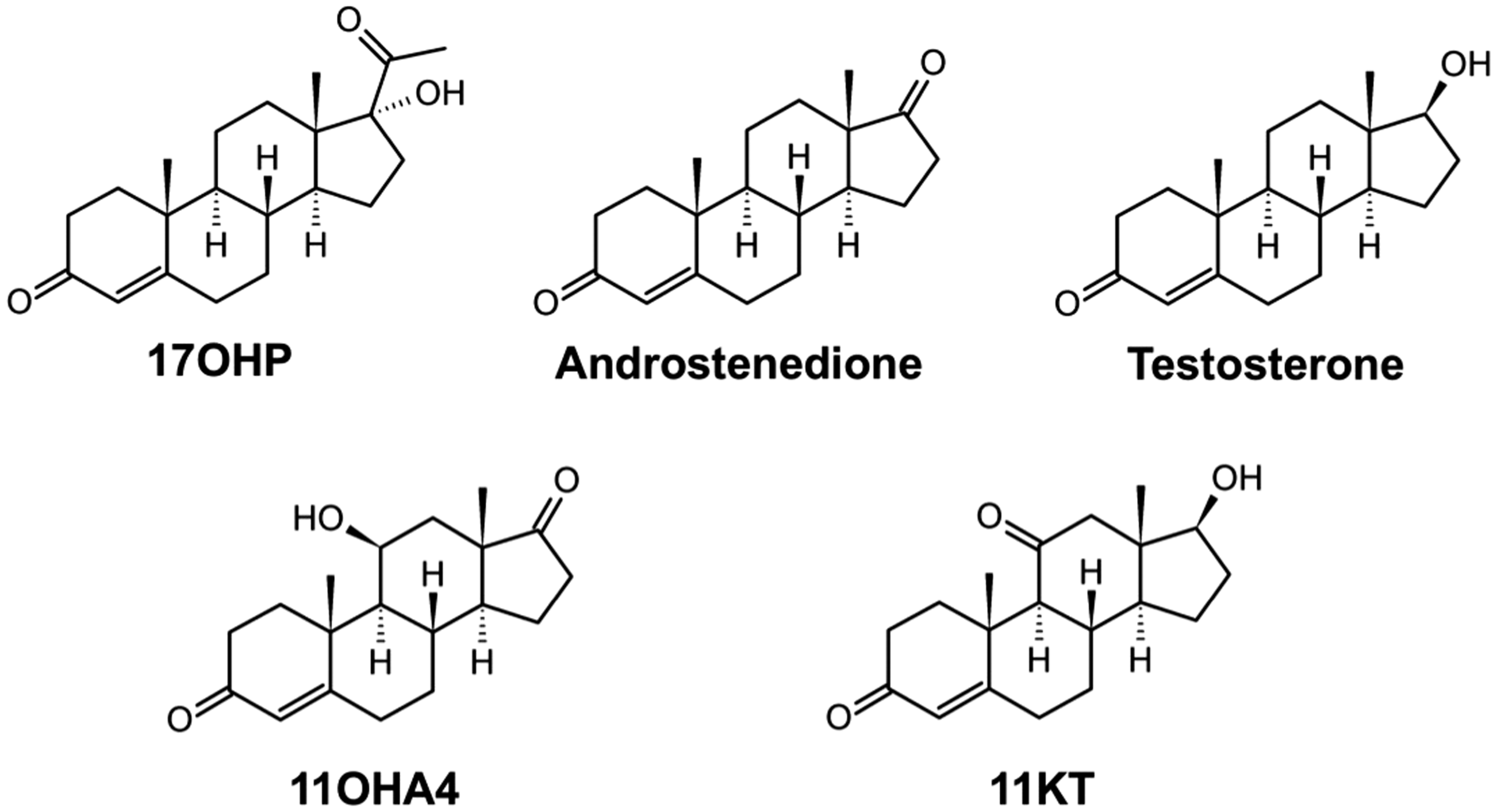
Structures of 17-hydroxyprogesterone (17OHP), androstenedione, testosterone, 11β-hydroxyandrostenedione (11OHA4) and 11-ketotestosterone (11KT).
Material, subjects and methods
Collection of saliva samples
Unstimulated whole saliva was collected by the passive drool technique as previously described. 22 The passive drool technique was chosen because commercially available collection devices, like Salivettes®, can affect analyte recovery and results of the measurement due to unspecific adsorption of non-polar steroids such as testosterone.23,24 To avoid blood or other contaminations of the sample, participants were asked to avoid brushing teeth 2 h before the sample collection, avoid eating 1 h before the sample collection and to rinse their mouth with water 10–15 min prior to sample collection. Immediately after, collection samples were frozen at –20°C. Samples were thawed, centrifuged at 2500 g for 5–10 min and the clear supernatant was aliquoted and stored at –80°C for analysis.
The use of human saliva samples has been ethically approved by the Science, Technology, Engineering and Mathematics Ethical Review Committee of the University of Birmingham (ERN_17-0494). All participants gave written informed consent prior to the sample collection. Samples were anonymized, and no identifying information was available at the laboratory. Healthy volunteers provided morning samples collected between awakening and 10 a.m. Male participants (n = 17) were between 20 and 35 years of age. Female participants (n = 10, age range 26–39) had a regular menstrual cycle, did not take oral contraceptives and collected the sample on day 21 of the menstrual cycle. The start of the menstrual bleed was defined as day 1.
Preparation of stock solutions, standards and QCs
17OHP, androstenedione and testosterone were purchased as solutions in methanol (Cerilliant, Sigma Aldrich, Poole, UK). 11OHA4 and 11KT were purchased as powder (Sigma Aldrich, Poole, UK). Stock solutions were prepared by dissolving or diluting the respective steroid in ultra-pure methanol (Fluka, Poole, UK) and were stored at –20°C. Two separately prepared stock solutions were used for the preparation of standards and quality controls (QCs) by dilution in a surrogate saliva matrix consisting of phosphate-buffered saline (pH 7.4) supplemented with 0.1% (w/v) bovine serum albumin (PBS/BSA). The standards and QCs contained a mixture of all analytes at the desired concentrations. Nine standards including a zero sample without analytes were used. Concentrations are summarized in Table 1. The internal standard mixture in methanol contained [13C3]-(2–4)–17-alpha-hydroxyprogesterone ([13C3]-17OHP; Cerilliant, Round Rock, USA), D7-(2,4,6,16)–4-androstene-3,17-dione (D7- androstenedione; CDN isotopes, Pointe-Claire, Canada), [13C3]-(2–4)-testosterone (Isosciences, King of Prussia, USA), D7-2,2,4,6,6,16,16–4-androsten-11β-ol-3,17-dione (D7-11OHA4; CDN Isotopes, Pointe-Claire, Canada), D3-16,16,17–11-ketotestosterone (D3-11KT; Isosciences, Philadelphia, USA).
Standard and QC concentrations, mass transitions m/z, cone voltages and collision energies for target analytes and internal standards.
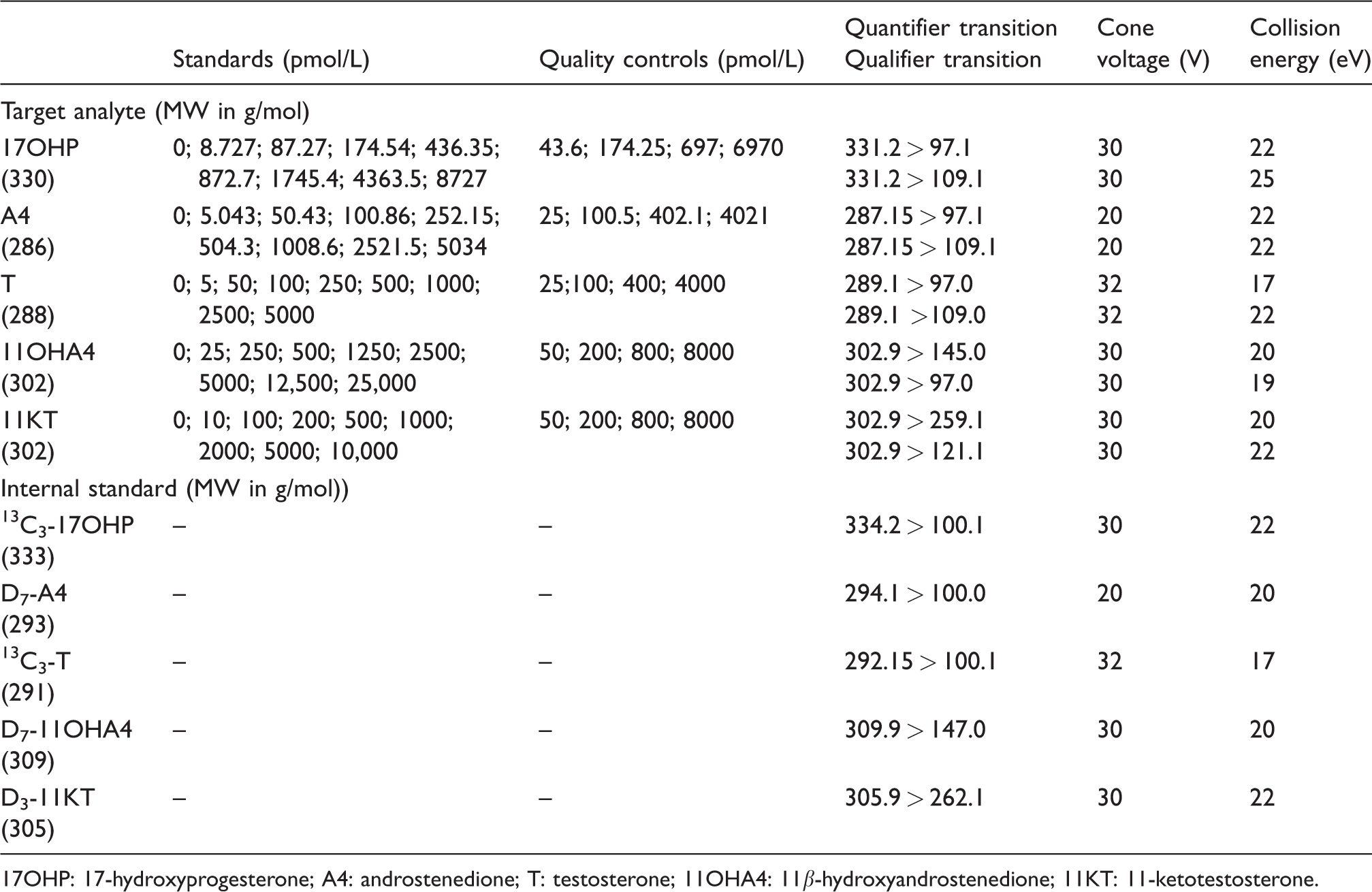
17OHP: 17-hydroxyprogesterone; A4: androstenedione; T: testosterone; 11OHA4: 11β-hydroxyandrostenedione; 11KT: 11-ketotestosterone.
Sample preparation by supported liquid extraction
Samples were extracted by supported liquid extraction (SLE) in a 96-well format, and a set of standards and QCs was added to each plate. Saliva sample (300 μL), standard or QC were pipetted into the wells of a 2-mL square well 96-well plate (Porvair, Leatherhead, UK). Twenty microlitres of the internal standard (IS) mixture (Table 1) in methanol was added using an Eppendorf multipette. The plate was mixed for 1 min. Samples were transferred onto the wells of a SLE plate (SLE+, Biotage, Uppsala, Sweden), and a vacuum was applied to draw the entire sample into the matrix of the plate. The plate was incubated at room temperature for 20 min. For elution, a fresh 96-well plate was placed under the SLE plate and 1 mL of dichloromethane (Chromasolv Plus, Sigma Aldrich, Poole, UK) was added onto each well of the SLE plate. The solvent was allowed to pass through into the collection plate until no residual solvent was visible on the wells. Subsequently, positive pressure was applied for 30 s to transfer the remaining solvent into the collection plate. The dichloromethane was evaporated using a Vacmaster 96 (Biotage, Uppsala, Sweden), and samples were reconstituted in 100 μL 40% (v/v) ultra-pure methanol in ultra-pure Milli-Q water (Merck-Millipore, Hertfordshire, UK).
Online solid phase extraction and liquid chromatography
Online solid phase extraction and chromatography were carried out using a Waters Online SPE Manager coupled to an Acquity ultrahigh pressure liquid chromatography system (Waters, Manchester, UK). A Waters XBridge C18 cartridge was conditioned with 0.5 of mL methanol and equilibrated with 0.5 mL of water. Fifty microliters of sample were injected onto the cartridge using 0.5 mL water. The cartridge was washed with 0.25 mL of 30% (v/v) methanol in water, and the analytes were subsequently eluted onto the analytical Kinetex® C8 column (100 × 3 mm, 2.6 µm; Phenomenex, Torrance, USA) using 55% of mobile phase A (2 mmol/L ammonium acetate in 0.1% (v/v) formic acid in distilled Milli-Q water) and 45% of mobile phase B (2 mmol/L ammonium acetate in 0.1% formic acid in ultra-pure methanol). The flow rate was 0.45 mL/min, and the column temperature was 45°C. For chromatographic separation, a linear gradient from 45 to 85% mobile phase B over 4 min was applied, followed by 1 min at 100% B and 1 min of re-equilibration at starting conditions. The eluate was directly injected into the mass spectrometer.
Mass spectrometry
Quantification was performed on a Waters XEVO TQ-S mass spectrometer operated in positive ion mode. The source offset was maintained at 30 V with a source temperature of 150°C. The capillary voltage was 0.75 kV, and the desolvation temperature and gas flow were 550°C and 1200 L/h. Multiple reaction monitoring mode was used with a dwell time of 0.016 s. All mass transitions are summarized in Table 1 together with respective cone voltages and collision energies. MassLynx NT 4.1 software was used for the system control and the MassLynx TargetLynx programme for data processing. Peak height ratios of analyte and IS, 1/x weighting and linear least square regression were used to produce the standard curves.
Validation
The assay was validated according to accepted guidelines.25,26
Matrix effect
Several saliva samples (n = 5 for 17OHP, androstenedione and testosterone; n = 6 for 11OHA4 and 11KT) or distilled water were extracted by SLE as described above. Dried extracts were reconstituted in 100 μL of 40% methanol and spiked with 10 μL of a steroids stock in methanol resulting in the final concentrations indicated in Table 2. As a background control to determine the signal from the non-spiked sample, a corresponding volume of water was added to the reconstituted extract of saliva samples or water. The matrix effect was calculated from the peak height determined in extracts of saliva compared to peak height determined in the extract of water after deducting the respective background signal: 100–[(heightH2O–heightsaliva)/heightH2O]×100. Matrix effects between 80% and 120% were considered acceptable.
Recovery and matrix effects.
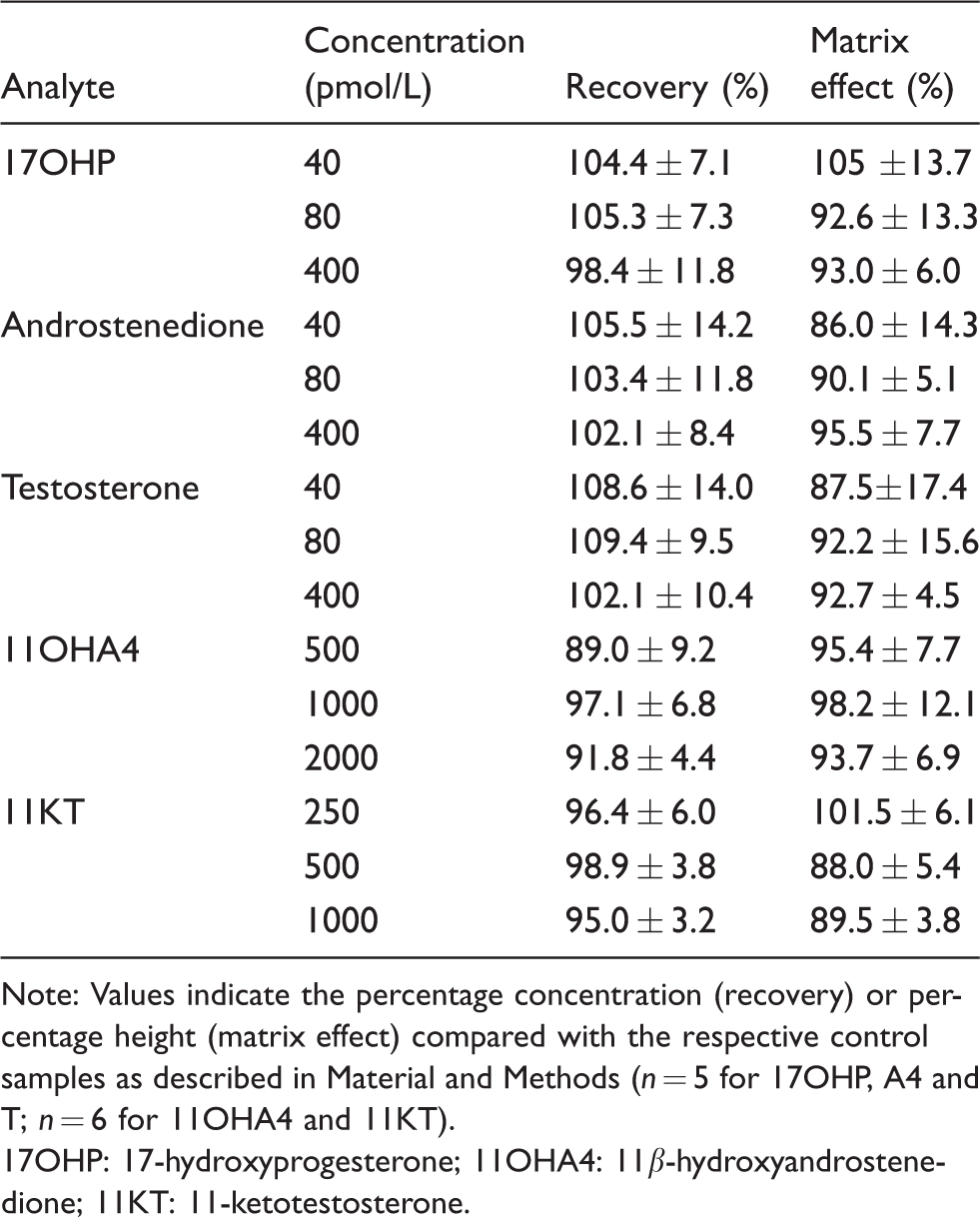
Note: Values indicate the percentage concentration (recovery) or percentage height (matrix effect) compared with the respective control samples as described in Material and Methods (n = 5 for 17OHP, A4 and T; n = 6 for 11OHA4 and 11KT).
17OHP: 17-hydroxyprogesterone; 11OHA4: 11β-hydroxyandrostenedione; 11KT: 11-ketotestosterone.
Recovery
Recovery includes extraction efficiency and matrix effects. Different saliva samples (n = 5 for 17OHP, androstenedione and testosterone; n = 6 for 11OHA4 and 11KT) or 50% (v/v) methanol were spiked with different concentrations of the analytes. As a background control for analyte concentrations before spiking, an equivalent volume of water was added to the samples. The samples were extracted as described above. Recovery was calculated from the concentrations measured in saliva compared with the concentrations measured in 50% (v/v) methanol deducting the respective background control. A recovery between 80% and 120% was considered acceptable.
Specificity
Solutions of common endogenous and synthetic steroids were prepared at supraphysiological concentrations in 50% (v/v) methanol in distilled water and analysed, after direct injection, for interference in all channels (Table 1) at the relevant retention times. The following steroids were tested: 21-deoxycortisol, aldosterone, corticosterone, cortisol, cortisone, DHEA, dexamethasone, pregnenolone, DHEAS, budesonide, fludrocortisone, fludrocortisone acetate, fluocinolone, triamcinolone, triamcinolone acetonide, prednisolone, prednisone, 21-hydroxyprogesterone, epitestosterone, beclomethasone, 11-deoxycortisol, dihydrotestosterone, cyproterone acetate, levonorgestrel, desogestrel, norethisterone and ethinylestradiol.
Dilution linearity
Different saliva samples (n = 6 for 17OHP, androstenedione and testosterone; n = 4 for 11OHA4 and 11KT) were spiked with the top concentration of the standard range for the respective analyte (see Table 1) and serially diluted (factor 2) with PBS/BSA. Measured concentrations were plotted against calculated theoretical concentrations and curves were judged linear if the correlation coefficient of linear regression (R2) was >0.9900. The bias between calculated theoretical and measured concentrations was calculated and a bias ≤10% was considered acceptable.
Precision and bias
Saliva samples with three different concentrations and PBS/BSA-based samples spiked with different concentrations of the analytes were analysed in 10 different runs to assess inter-assay imprecision as well as 10 times within the same run for intra-assay imprecision. The saliva samples were pooled female saliva, pooled male saliva and pooled mixed male and female saliva to allow to cover the range of concentrations and get the volume required for replicate analysis. The percentage coefficient of variation (CV) was calculated from the ratio of standard deviation and mean. The bias (spiked PBS/BSA-based samples only) was calculated as percentage deviation between the mean observed and nominal concentrations. CV and bias were considered acceptable if ≤10%.
Lower limit of quantification
The lower limit of quantification (LLOQ) was defined as the lowest concentration for which 10 replicates of PBS/BSA-based samples gave a bias and CV of less than 20%.
Post-extraction stability
Saliva samples (n = 17 for 17OHP, n = 18 for androstenedione, n = 16 for T, n = 10 for 11OHA4 and 11KT) were extracted in duplicate and reconstituted for analysis. The first plate was measured immediately after extraction, while the second plate was stored in a refrigerator for and overnight period before being analysed (10°C for 22 h for 17OHP, androstenedione and testosterone or at 4°C for 18h for 11OHA4 and 11KT). Percentage bias between the results of the two measurements was calculated.
Carry-over
A PBS/BSA-based sample containing 86,667 pmol/L 17OHP, 100,000 pmol/L androstenedione, 99300 pmol/L testosterone, 250,000 pmol/L 11OHA4 and 100,000 pmol/L 11KT was analysed followed by sample containing only PBS/BSA. Percentage carry-over of the analytes into the following sample was calculated from the peak height determined for the two samples.
Results
Validation of the analytical performance
All analytes eluted as discrete, identifiable peaks with co-elution of their respective deuterated or 13C-labelled IS. Figure 2 shows the chromatograms of a saliva sample. T and 17OHP co-eluted, but due to the difference in m/z of the parent ions (m/z of 17OHP 331.2 and 13C3-17OHP 334.2 and m/z testosterone 289.1 and 13C3-testosterone 292.15), no interference was observed. The total run time of the assay was 6.4 min injection-to-injection. For each batch, individual standard curves for each analyte were generated by plotting the ratio of analyte to IS peak height against the nominal concentrations. Standard curves were linear (R2 >0.99) over the entire range given in Table 1 and correlated with the respective nominal concentrations with reproducibility between batches. The evaluation of matrix effects (n ≥ 5 different saliva samples) showed percentage signals with mean values between 88 and 105% for three different concentrations per analyte and indicated no significant ion suppression or enhancement by the saliva matrix (Table 2). For recovery (n ≥ 5 different saliva samples), combining extraction efficiency and matrix effects, mean values were between 89 and 109.4% for three different concentrations of all analytes (Table 2). Interference was excluded for a large set of natural and synthetic steroids (see section Specificity for a complete list) for all analyte and IS channels by directly injecting supraphysiological concentrations. Serial dilutions of saliva after the addition of the analytes at concentrations of the respective highest standard (Table 1) showed linearity with R2 values >0.99, when measured concentrations where plotted against the theoretical concentrations. The bias between the measured and theoretical concentrations was 1.0 ± 5.9% for 17OHP, –9.1 ± 10.5% for androstenedione, –2.2 ± 6.9% for testosterone, 8.8 ± 6.3% for 11OHA4 and 7.3 ± 4.6% for 11KT.
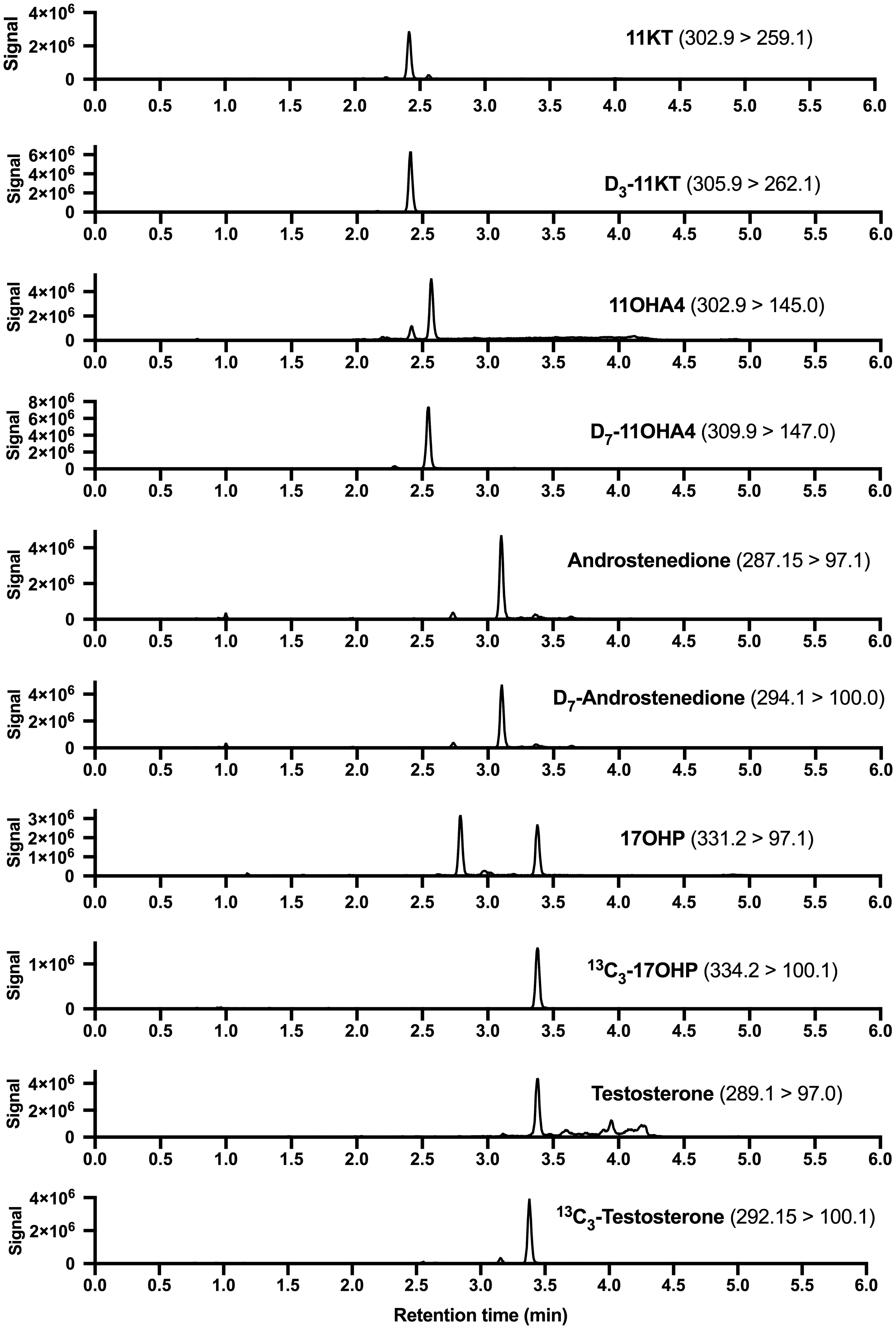
Chromatograms of a saliva sample for all target analytes and their internal standards. Quantifier transitions (m/z) are indicated.
The evaluation of the precision of the assay within and between batches is summarized in Table 3. For spiked PBS/BSA-based samples, the intra-assay CV was <8.1% and the intra-assay bias ranged between –6.6 and 4.2% for all analytes. The respective inter-assay CV was <10.0%, and the inter-assay bias was between –3.1 and 10.2%. For selected saliva samples with three different concentrations of each analyte, the intra-assay CV was <6.8% and the inter-assay CV was <9.3%. The LLOQs were 12.5 pmol/L for 17OHP (1.875 fmol on column), 6.25 pmol/L for androstenedione (0.9375 fmol on column), 5 pmol/L for testosterone (0.75 fmol on column), 50 pmol/L for 11OHA4 (7.5 fmol on column) and 5 pmol/L for 11KT (0.75 fmol on column). Post-extraction stability was tested for overnight storage. The bias between the direct and post-storage analysis was between –8.4 and 8.8% for 17OHP, –14.9 and 4.9% for androstenedione, –30.6 and 19.9% for testosterone, –8.8 and 4.8% for 11OHA4 and –8.3 and −0.5% for 11KT. Carry-over was 0.15% for 17OHP, 0.62% for androstenedione, 0.65% for T, 0.075% for 11OHA4 and 0.036% for 11KT.
Intra- and inter-assay precision and bias.
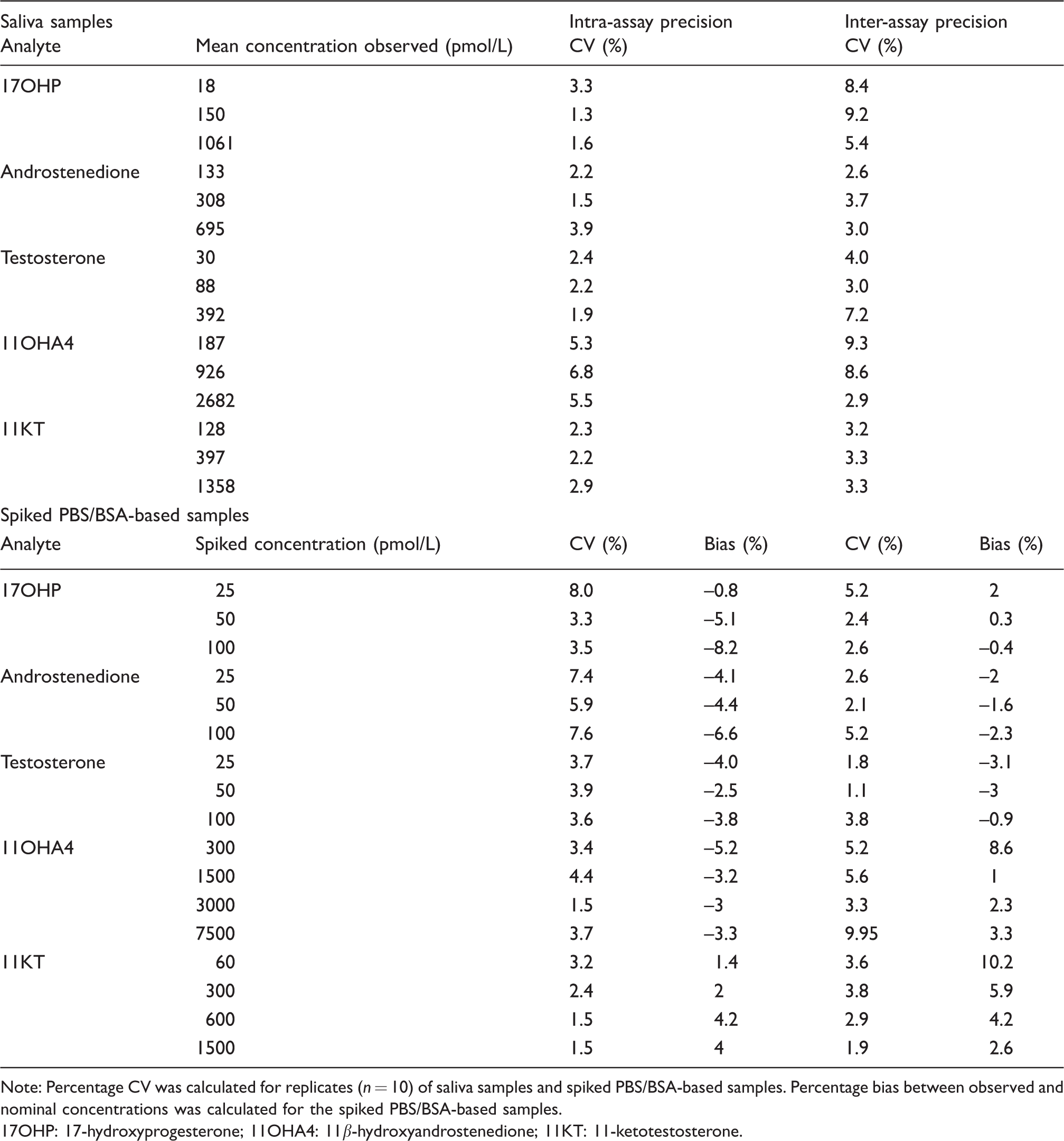
Note: Percentage CV was calculated for replicates (n = 10) of saliva samples and spiked PBS/BSA-based samples. Percentage bias between observed and nominal concentrations was calculated for the spiked PBS/BSA-based samples.
17OHP: 17-hydroxyprogesterone; 11OHA4: 11β-hydroxyandrostenedione; 11KT: 11-ketotestosterone.
Salivary androgens in healthy volunteers
We applied the assay to saliva samples collected by healthy male (n = 17) and female volunteers (n = 10) between awakening and 10 a.m. Female participants had a regular menstrual cycle and provided samples collected on day 21 of the cycle. The results are summarized in Figure 3. Concentrations were above the LLOQ for all samples and analytes. For male participants, median concentrations (range) were 56 (18–133) pmol/L for 17OHP, 147 (86–310) pmol/L for androstenedione, 187 (130–320) pmol/L for testosterone, 450 (127–2114) pmol/L for 11OHA4 and 276 (129–548) pmol/L for 11KT. For female participants, median (range) values were 117 (27–201) pmol/L for 17OHP, 242 (107–367) pmol/L for androstenedione, 18 (7–149) pmol/L for testosterone, 448 (201–2039) pmol/L for 11OHA4 and 233 (139–535) pmol/L for 11KT. As expected, women had higher 17OHP concentrations than men (P = 0.0009), but lower testosterone concentrations (P < 0.0001). No differences between men and women were observed for analytes that are primarily of adrenal origin: androstenedione, 11OHA4 and 11KT. 11OHA4 showed the widest inter-individual variation followed by 11KT.
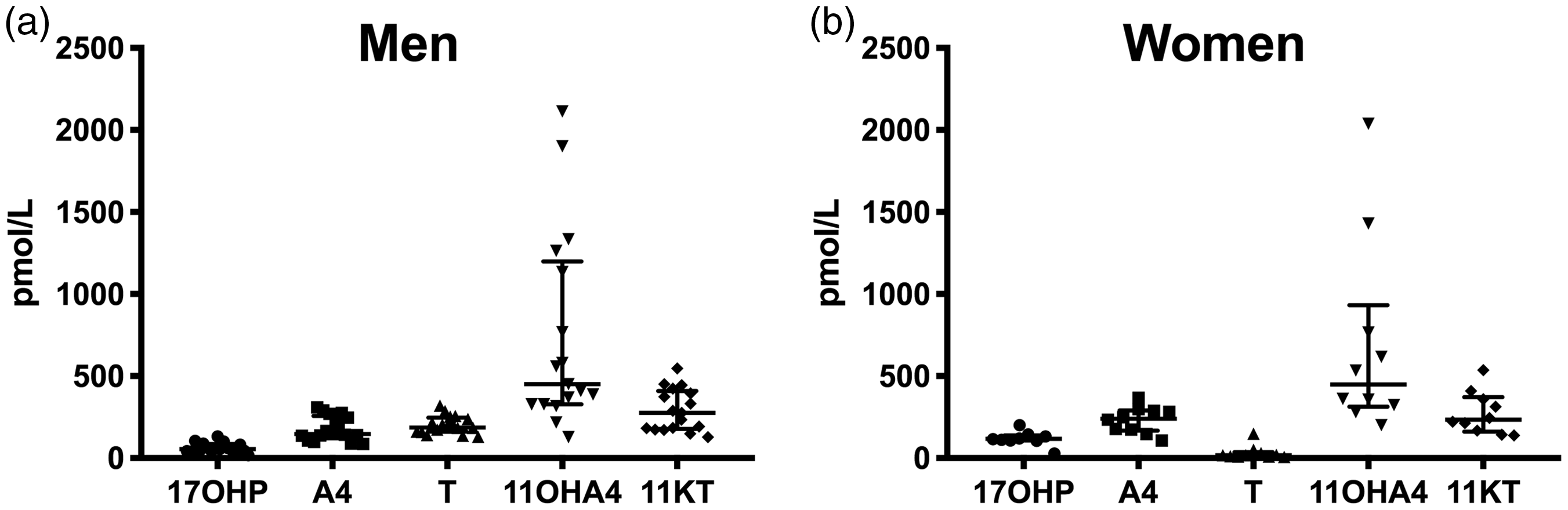
Morning salivary androgen concentrations in healthy men (A, n = 17) and women (B, n = 10). Samples were collected between awakening and 10 a.m. Participating women had a regular menstrual cycle and collected the sample on day 21 of the cycle. Individual data points, median and interquartile range are depicted.
Discussion
We present a novel LC-MS/MS assay for the simultaneous measurement of highly clinically relevant steroids derived from the classic androgen pathway, androstenedione and testosterone, and the 11-oxygenated androgen pathway, 11OHA4 and 11KT, as well as the precursor steroid 17OHP in saliva. This specific combination of analytes allows for the comprehensive single assay assessment of androgen status including monitoring of biochemical control in CAH, which is required over the entire course of life for treatment titration and risk stratification. The use of saliva facilitates the collection of multiple samples over long periods of time due the simple, self-administered collection technique. While previously published assays for salivary androgens were limited to the measurement of combinations of testosterone, androstenedione and 17OHP,7,27–29 our new assay enables a complete assessment of the androgen profile, by including 11-oxygenated androgens, which have recently been shown to significantly contribute to the circulating androgen burden in PCOS and CAH.12,13 The LLOQs for 17OHP, androstenedione and testosterone of our assay are comparable27,28 or superior 29 to previously published assays. The short run time of the assay (6.4 min) compares favourably to other assays (9.5 to 10 min)27–29 and allows for the implementation of the assay into routine practise of clinical laboratories. We performed a strict validation of the developed assay that guarantees its accurate, precise and reproducible performance according to accepted criteria.25,26 The assay was demonstrated to be highly selective excluding interference with a large set of natural and synthetic steroids. There are no significant effects of the saliva matrix and the assay has excellent recovery, intra- and inter-assay precision and linearity. The high bias between the results for testosterone before and after storage of the extracted samples indicates limited postextraction stability of testosterone. If the accurate quantification of testosterone is required, the samples thus need to be analysed without postextraction storage. However, all other analytes are stable for an overnight period.
We have successfully applied the assay for preliminary measurements of the salivary androgen profile in healthy male and female volunteers. This is the first time 11-oxygenated androgens have been measured in saliva. Measured concentrations were above the LLOQ for all analytes, demonstrating appropriate sensitivity of the assay to quantify the low picomolar concentrations in saliva. For androstenedione and testosterone, the presented concentrations compare well to data previously published by our laboratory5,7 and others.27,28 Male 17OHP concentrations are also in the same range as previously published, but female 17OHP concentrations were higher in our samples compared with the data published by Mezzullo et al., which can be explained by age differences of the participants between the two studies and the collection of our samples exclusively on day 21 of the menstrual cycle. 28 We observed a comparably high degree of inter-individual variability for salivary 11OHA4. 11OHA4 is the second most abundant steroid produced by the adrenal gland after DHEAS and a sensitive marker for adrenal steroid output,9,14 which is maximal in the early morning with a subsequent decline. The samples in this study have been collected between awakening and 10 a.m. Differences in the timing of sample collection within this window are likely to be magnified due to the higher morning production rate of 11OHA4 compared with other steroids generated by down-stream metabolism in peripheral tissue or, at least in part, primary gonadal production; this is a likely explanation for the observed higher variation in salivary 11OHA4. It should be noted that, due to the limited number of participants and the limitation of female samples to a specific day of the menstrual cycle, the presented data does not yet allow the derivation of reference ranges.
In conclusion, we have developed and validated a multisteroid salivary LC-MS/MS assay for comprehensive assessment of both classic and 11-oxygenated androgen pathways with high potential for routine clinical use for the assessment of androgen excess conditions and treatment monitoring in CAH.
Footnotes
Acknowledgments
The authors would like to thank all participants for the collection of saliva samples.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The Science, Technology, Engineering and Mathematics Ethical Review Committee of the University of Birmingham approved this study (ERN_17-0494).
Guarantor
BGK.
Contributorship
BGK, JEA and LS have contributed to the method development. JEA and LS have conducted the validation of the assay and the measurement of saliva samples. All authors have contributed to the writing and editing of the article. All authors have accepted responsibility for the entire content of this submitted article and approved submission.
