Abstract
Background
Apolipoprotein E-containing high-density lipoprotein shows antiatherogenic properties in vitro. There is a need for a homogeneous assay to determine the concentration of apolipoprotein E-containing high-density lipoprotein for in vivo studies.
Methods
In the proposed homogeneous assay, lipoproteins other than apolipoprotein E-containing high-density lipoprotein were eliminated in the first step. Apolipoprotein E-containing high-density lipoprotein-cholesterol was measured in the second step. The control study used a 13% polyethylene glycol precipitation assay (control assay).
Results
The homogeneous assay showed good performance in validation studies. In subjects with normal liver function (n = 78), a significant correlation was found between the control assay and the homogeneous assay (r = 0.824). Serum apolipoprotein E-containing high-density lipoprotein cholesterol concentrations, determined by the control assay and the homogeneous assay, respectively, were 0.05 (0.04–0.10) (median [25th–75th percentile]) mmol/L and 0.10 (0.06–0.13) mmol/L for healthy individuals (n = 12), and 0.03 (0.01–0.13) mmol/L and 0.02 (0.01–0.02) mmol/L for patients with cholestasis (n = 6). The results indicate that the homogeneous assay recovers cholesterol contained in physiological apolipoprotein E-containing high-density lipoprotein, but not in pathological apolipoprotein E-containing high-density lipoprotein from cholestatic patients.
Conclusions
The proposed two-step homogeneous assay enables selective measurement of physiological apolipoprotein E-containing high-density lipoprotein cholesterol in common autoanalysers. This assay might uncover a role for apolipoprotein E-containing high-density lipoprotein in physiological conditions.
Introduction
Serum high-density lipoprotein cholesterol (HDL-C) concentration is an established negative risk factor for cardiovascular disease.1–3 However, several epidemiological studies have reported that high plasma HDL-C concentrations were not associated with a reduction of cardiovascular and non-cardiovascular mortality.4–7 Thus, it suggests that total HDL-C concentration may not be enough to assess the risk of the event.
HDL contains approximately 100 protein species, including apolipoprotein A-I, A-II, Cs and E. 8 As shown in apolipoprotein E (apoE)-knockout mice, apoE is associated with antiatherogenicity. 9 Hence, it is meaningful to separately consider apoE-containing HDL and apoE-deficient HDL as risk factors for atherosclerosis. 10 ApoE-containing HDL accounts for up to 10% of HDL in terms of cholesterol in adult human plasma.11–15 ApoE-containing HDL is a major lipoprotein species in the serum of human foetus, suggesting an important role for this lipoprotein in yet poorly understood lipid transport in the foetus. 16
ApoE-containing HDL-C increases in patients with cholestasis17–20 and those with cholesteryl ester transfer protein (CETP) deficiency.10,14,15,21,22 Additionally, synthetic CETP inhibitors are known to increase apoE-containing HDL.23–25 ApoE-containing HDL can be divided into two subspecies: one is small in size and cholesterol poor (apoE-containing HDL3) and the other is large and cholesterol rich (apoE-containing HDL2). 11 In relevance to atherosclerosis, apoE-containing HDL2 seems to be more important than the other, since it can mediate cholesterol efflux from macrophages more efficiently than apoE-deficient HDL2 10 and inhibit the agonist-induced platelet aggregation.26,27
ApoE-containing HDL and low-density lipoprotein (LDL) are difficult to separate from each other by ultracentrifugation and high-performance liquid chromatography (HPLC), since they overlap in density and particle size. 21 Moreover, apoE-containing HDL can be precipitated at least partly by polyanion precipitation, together with apoB-containing very low-density lipoprotein (VLDL) and LDL. 13 Furthermore, apoE-containing HDL-C represents only a small portion (<10%) of total HDL-C in healthy adults. 13 For these reasons, quantitative evaluation of apoE-containing HDL-C has been technically difficult.
A precipitation method using 13% (w/v) polyethylene glycol (PEG) and sodium phosphotungstate-dextran sulphate-MgCl2 (PT-DS-Mg++) for differential measurement of apoE-deficient and apoE-containing HDL-C has been reported by Chiba et al.13–15,20 This assay was used to show an increase in apoE-containing HDL-C in the patients with cholestatic liver disease13,20,28–30 and CETP deficiency.14,15,22 However, a more convenient homogeneous assay would have been highly useful for both routine measurements and large-scale clinical and epidemiological studies.
We have previously reported a three-step homogeneous assay for simultaneous measurements of apoE-deficient HDL-C, apoE-containing HDL-C and total HDL-C. 31 In this assay, a surfactant D201 – a polyoxyethylene benzylphenyl ether derivative – plays a critical role. D201 reacts with apoE-deficient HDL-C at lower concentrations, and it can also react with total HDL-C at higher concentrations. On the basis of this unique nature of D201, we established the three-step homogeneous reagent and demonstrated the relationship between the apoE-deficient HDL-C, apoE-containing HDL-C and total HDL-C. 31 Using this assay, we successfully detected a significant increase in apoE-containing HDL-C in the serum samples from the subjects with hyperalphalipoproteinaemia with total HDL-C concentrations higher than 2.59 mmol/L (100 mg/dL). 31 In spite of its advantages, this three-step reagent cannot be used widely since common autoanalysers do not always accommodate three-step reagents. Therefore, a two-step homogeneous reagent for apoE-containing HDL-C would be highly useful for clinical laboratories.
Here, we report a novel two-step homogeneous assay for the selective measurement of apoE-containing HDL-C. This assay might serve as a useful tool in clinical and epidemiological studies to elucidate the currently unclear role of the apoE-containing HDL in the development and prevention of atherosclerosis.
Material and methods
Samples
All blood samples were obtained after an overnight fast from healthy volunteers (n = 12; five men and seven women; age 58 ± 16 years) and the outpatients of the Department of Diabetes and Endocrinology, Sapporo City General Hospital. Serum samples were separated by centrifugation at 1500 × g for 10 min and used immediately.
In order to study the effect of cholestasis on the measurements, serum samples were obtained from the patients with cholestasis (n = 6; four men and two women; age 63 ± 11 years; 3 mg/dL ≤ total bilirubin <10 mg/dL), and were compared with those from the healthy volunteers. In order to study the correlation between the proposed homogeneous assay and the control precipitation assay, serum samples were obtained from the patients without cholestasis (n = 78; 30 men and 48 women; age 60±17 years old; total bilirubin ≤1.0 mg/dL). In order to study the correlation between the proposed homogeneous assay and the three-step homogeneous assay, 31 serum samples were obtained from an additional batch of patients without cholestasis (n = 17; 5 men and 12 women; age 60±15 years old).
Control assay
In the control study, the serum concentration of apoE-containing HDL-C was measured by the control precipitation method as previously reported. 13 Briefly, two precipitation reagents were prepared: a 13% (w/v) PEG reagent (PEG 6000, Wako Pure Chemicals Industries Ltd) and a PT-DS-Mg++ reagent containing 1.8 g/L dextran sulphate, 3.0 g/L sodium phosphotungstate and 0.1 mol/L MgCl2 (HDL-C Daiichi, Daiichi Pure Chemicals Co. Ltd, Tokyo, Japan). ApoE-deficient and total HDL-C were given by total cholesterol (TC) in the supernatant of the PT-DS-Mg++ precipitation and the 13% PEG precipitation, respectively. ApoE-containing HDL-C was estimated by subtracting apoE-deficient HDL-C from total HDL-C.
Protocol of the proposed homogeneous assay
The reaction scheme is shown in Figure 1(a). In the first step, non-HDL lipoproteins and apoE-deficient HDL were decomposed by the first reagent (Reagent 1), which contains a low concentration of D201 to decompose the former and the latter lipoprotein species, respectively. 31 Solubilized cholesterol was decomposed through a cholesterol esterase/cholesterol oxidase/catalase reaction without colour development. In the second step, apoE-containing HDL-C interacted with the second reagent (Reagent 2), containing a high concentration of D201, resulting in colour development. Briefly, a high concentration of D201 reacts with apoE-containing HDL selectively among the remaining lipoproteins, releasing cholesterol specifically from apoE-containing HDL. Solubilized cholesterol from apoE-containing HDL is decomposed through a cholesterol esterase/cholesterol oxidase reaction, resulting in the production of hydrogen peroxide. The reaction mixture including the resulted hydrogen peroxide turned purple-red with coupler in the presence of peroxidase. The absorbance was measured at 600 nm.
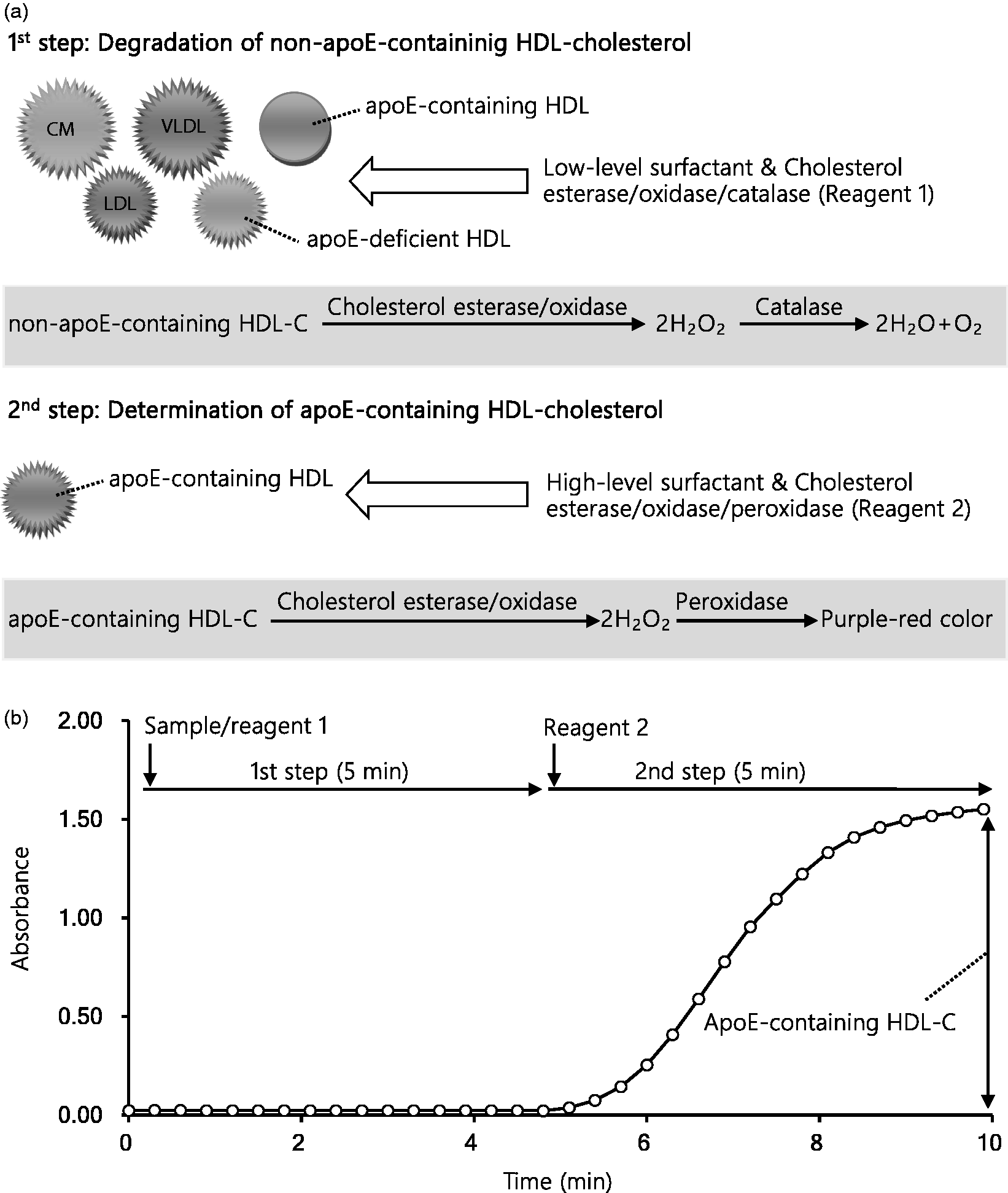
The principle of the proposed two-step homogeneous apoE-containing HDL-C assay. (a) Scheme of the assay. In the first step, lipoproteins other than apoE-containing HDL, such as CM, VLDL, LDL and apoE-deficient HDL, are decomposed by the surfactant D201 at a lower concentration. Cholesterol oxidase generates hydrogen peroxide, which is decomposed to water and oxygen by catalase. No colour development occurs in this step. In the second step, apoE-containing HDL was solubilized with the surfactant D201 at an increased concentration. Cholesterol oxidase generates hydrogen peroxide, which in turn is used as a substrate for peroxidase leading to colour development. (b) The reaction time course of the assay. Using a Hitachi 7170, 180 μL of Reagent 1 was mixed with 7.0 μL of the serum sample and reacted at 37°C for 5.0 min. Next, the sample mixture was reacted with 60 μL of Reagent 2 at 37°C for 5.0 min. Absorbance was measured at the main wavelength of 600 nm and the secondary wavelength of 700 nm, with the change in absorbance observed after 5 to 10 min of the reaction for apoE-containing HDL-C, and multiplied by factors calculated on the basis of calibration to estimate the cholesterol concentration.
All reactions were performed using a Hitachi 7170 Chemistry analyzer (Hitachi Ltd., Tokyo, Japan). The time course is shown in Figure 1(b). Serum (7.0 μL) was mixed with 180 μL of Reagent 1 and reacted at 37°C for 5 min. Then the sample mixture was reacted with 60 μL of Reagent 2 at 37°C for 5 min. Absorbance was measured at the main wavelength of 600 nm and the secondary wavelength of 700 nm, with the change in absorbance observed after 5–10 min of the reaction for apoE-containing HDL-C, and multiplied by factors calculated on the basis of calibration to estimate the cholesterol concentration.
Optimization of the surfactant concentration
For the determination of the optimal concentration of the surfactant (D201), we tested several concentrations of D201: 0.05–0.75% (final concentration) for Reagent 2. A serum sample with a high apoE-containing HDL-C concentration was used for this purpose.
Linearity
Linearity was examined using a commercially obtained purified human HDL (Bioresource Technology Inc., FL, USA). ApoE-containing HDL-C concentration in the HDL solution was determined by 13% PEG/PT-DS-Mg++ precipitation assay. Dilution was done with saline. All assays were performed in triplicate. The lower limit of detection was determined as the ΔOD of which mean −2.6 SD does not overlap with the mean +2.6 SD of the zero standard.
Sample stability
Serum samples obtained from healthy subjects were isolated within 1 h after blood sampling and pooled. The pooled serum was stored refrigerated (4°C), at room temperature (25°C) or at –80°C to evaluate sample stability of apoE-containing HDL-C concentration quantified by our novel homogeneous assay.
Influence of coexistent substances
Tests for interfering substances were performed with the Interference Check A Plus (Sysmex Corporation, Kyoto, Japan), to examine the effect of conjugated bilirubin, unconjugated bilirubin, haemoglobin and chyle. According to the manufacturer’s instructions, pooled serum was mixed with the suspected interfering substance at 11 different concentrations ranging from 0% to 100%. The apoE-containing HDL-C in each sample was measured using the proposed homogeneous assay.
Influence of LDL-C
Lipoprotein fractions were isolated from serum by sequential ultracentrifugation. 32 Briefly, ultracentrifugation was performed using a near-vertical tube rotor (MLN-80; Beckman Coulter, Fullerton, CA) on a model Optima MAX (Beckman Coulter). First, 2.0 mL of serum was adjusted with 6.0 mL of KBr solution to d = 1.019 kg/L and then centrifuged at 78,000 × g for 20 h at 4°C. The top fraction (2.5 mL) containing d < 1.019 lipoproteins, specifically chylomicron, VLDL and IDL, was isolated by aspiration. The bottom fraction was then adjusted with 2.5 mL of KBr solution to d = 1.063 kg/L and centrifuged again at 121,000 × g for 18 h at 4°C. After centrifugation, the top fraction (2.5 mL) containing LDL was recovered. The LDL fraction was dialysed against PBS overnight at 4°C. The cholesterol concentration of LDL fraction was 2.38 mmol/L (92 mg/dL).
Fifty microlitres of either the LDL sample or saline were spiked into 450 μL of serum obtained from the same subject. Five sets of the same sample were prepared. Then, the samples were subjected to total cholesterol and apoE-containing HDL-C homogeneous assays.
Correlation studies
Correlation between the control precipitation assay and the proposed homogeneous assay was studied using the serum samples from patients without cholestasis. Correlation between the proposed assay and the three-reagent homogeneous assay was studied using the serum samples from patients without cholestasis from different batches.
Comparison between study groups
Concentrations of apoE-containing HDL-C were compared between the control precipitation assay and the proposed homogeneous assay. Serum samples with the concentrations of apoE-containing HDL-C higher than 0.4 mmol/L or 15.5 mg/dL were excluded from the study owing to insufficient accuracy of this assay.
Statistical analysis
Wilcoxon rank-sum test was applied for the comparison of apoE-containing HDL-C concentrations between the proposed homogeneous assay and the control precipitation method. All statistical analyses were performed with EZR (Nankodo Co., Ltd, Tokyo, Japan). Significance level was set at P < 0.05.
Results
Optimized concentration of surfactant
The concentrations of HDL-C measured by the 13% PEG and PT-DS-Mg++ precipitation assay were 5.06 mmol/L (196 mg/dL) and 3.67 mmol/L (142 mg/dL), respectively (Figure 2). The results obtained by the homogeneous method with the D201 concentrations higher than 0.20% were close to those obtained by the 13% PEG precipitation assay. On the other hand, the results obtained by the homogeneous method with the D201 concentrations of 0.05–0.10% were similar to those obtained by the PT-DS-Mg++ precipitation assay.
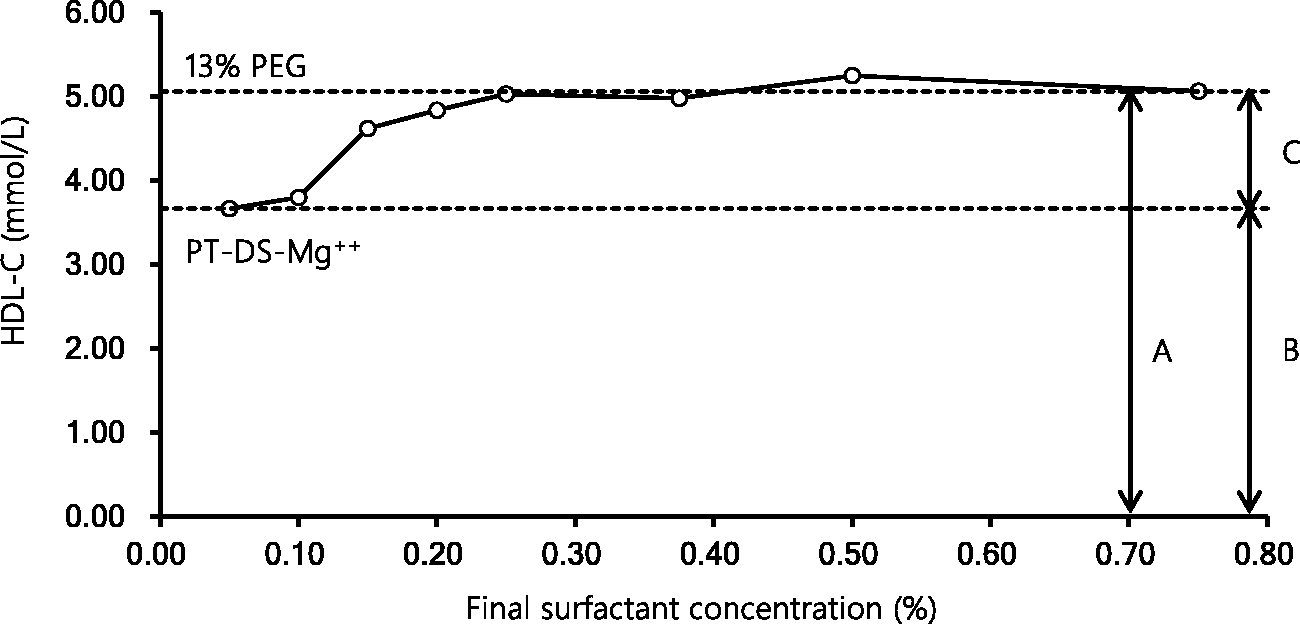
Reactivity of the surfactant D201 with HDL. To determine the optimal concentration of the surfactant D201 in the two-step homogeneous apoE-containing HDL-C assay, we prepared eight kinds of reagents with various concentrations of the surfactant (final concentration, 0.05–0.75%) as R2. A serum sample with high concentration of apoE-containing HDL-C was used in this experiment. Each arrow represents the concentrations of total HDL-C (a), apoE-deficient HDL-C (b) and apoE-containing HDL-C (c). The top line (broken) corresponds to the measurement by 13% PEG precipitation assay, indicating total HDL-C. The bottom line (broken) corresponds to the measurement by the PT-DS-Mg++ precipitation assay, indicating apoE-deficient HDL-C.
Reproducibility
Reproducibility was studied using three serum samples with different apoE-containing HDL-C concentrations: low (0.06 mmol/L or 2.4 mg/dL), medium (0.21 mmol/L or 8.0 mg/dL) and high (0.47 mmol/L or 18.2 mg/dL). The within-day CVs (n = 20) of the proposed homogeneous assays were 1.5% (low), 2.0% (medium) and 2.6% (high). The between-day CVs of the proposed homogeneous assay in six runs every day for five days were 6.2% (low), 5.0% (medium) and 3.8% (high).
Linearity
The linearity of the proposed homogenous assay was confirmed in the concentration range between 0.01 mmol/L (0.3 mg/dL) and 0.59 mmol/L (23.0 mg/dL) apoE-containing HDL-C (Figure 3). Thus, lower detection limit was recognized as 0.01 mmol/L (0.3 mg/dL).
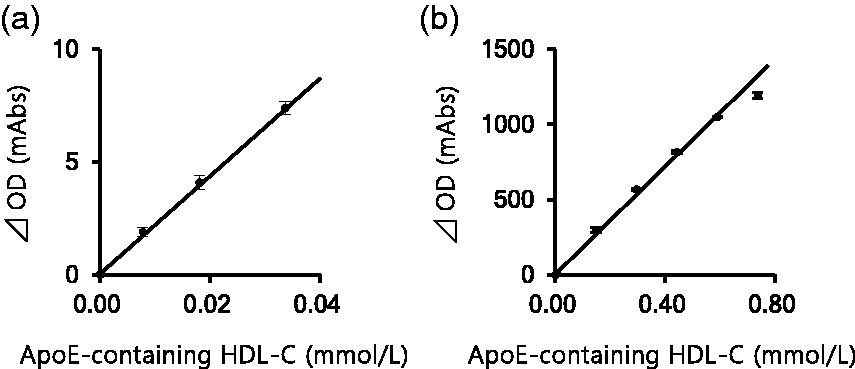
Linearity of the proposed two-step homogeneous apoE-containing HDL-C assay. (a) Low-level linearity was tested by using pooled serum samples from the patients with hypo-HDL-cholesterolaemia. (b) High-level linearity was tested by using a concentrated HDL fraction derived from serum samples with high HDL-C concentrations. Dilution was done with saline.
Sample stability
Serum samples were stable for five days under refrigeration or for 9 h at room temperature and were not affected by four freeze–thaw cycles (Figure 4(a)) or storage at –80°C for a month (data not shown).
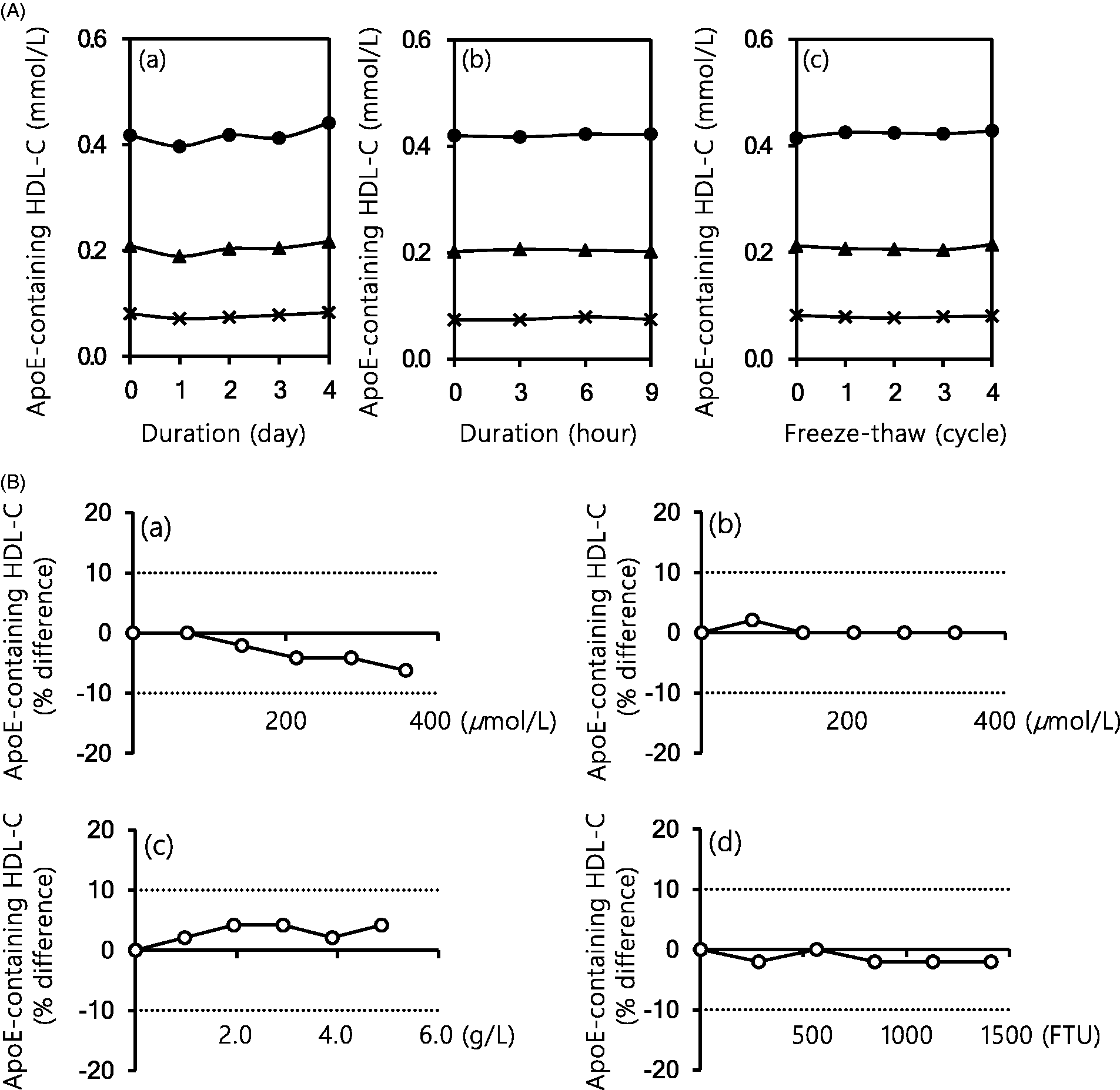
Sample stability and influence of coexistent substances on the proposed two-step homogeneous apoE-containing HDL-C assay. (A) Sample stability for the proposed assay: (a) refrigerated (4°C); (b) room temperature (25°C) and (c) −80°C, freeze–thaw cycles. The serum samples with low, medium or high apoE-containing HDL-C concentrations were stored at 4°C, 25°C and −80°C each. (B) Influence of the coexistent substances on the proposed assay: (a) conjugated bilirubin; (b) unconjugated bilirubin; (c) haemoglobin and (d) chyle.
Influence of coexistent substance
The proposed two-step homogeneous assay was not influenced more than ±10% by conjugated bilirubin (<357 μmol/L, 21 mg/dL), unconjugated bilirubin (<333 μmol/L, 20 mg/dL), haemoglobin (<5.0 g/L) and chyle (<1410 Formazin Turbidity Unit) (Figure 4(b)).
Influence of LDL
We spiked isolated LDL or saline (control) into normal serum samples (n = 5). TC measurements after spiking LDL were 4.94 ± 0.03 mmol/L (191 ± 1.2 mg/dL), while that for the control was 4.81 0.02 mmol/L (186 ± 0.7 mg/dL). The apoE-containing HDL-C determined by the proposed homogeneous assay was 0.135 ± 0.001 mmol/L (5.22 ± 0.04 mg/dL) after spiking LDL, whereas that for the control was 0.136 ± 0.001 mmol/L (5.26 ± 0.05 mg/dL), showing no statistically significant difference.
Correlation
In the samples from patients without cholestasis (n = 78), the regression equation between the control precipitation assay (x) and the proposed homogeneous assay (y) was: y = 0.944x + 0.07 (r = 0.824, P < 0.01) (Figure 5(a)). In the samples from patients without cholestasis (n = 17), the regression equations and correlation coefficients between the three-reagent homogeneous assay and the proposed homogeneous assay were y = 0.71x + 0.04 (r = 0.987, p < 0.01) (Figure 5(b)).
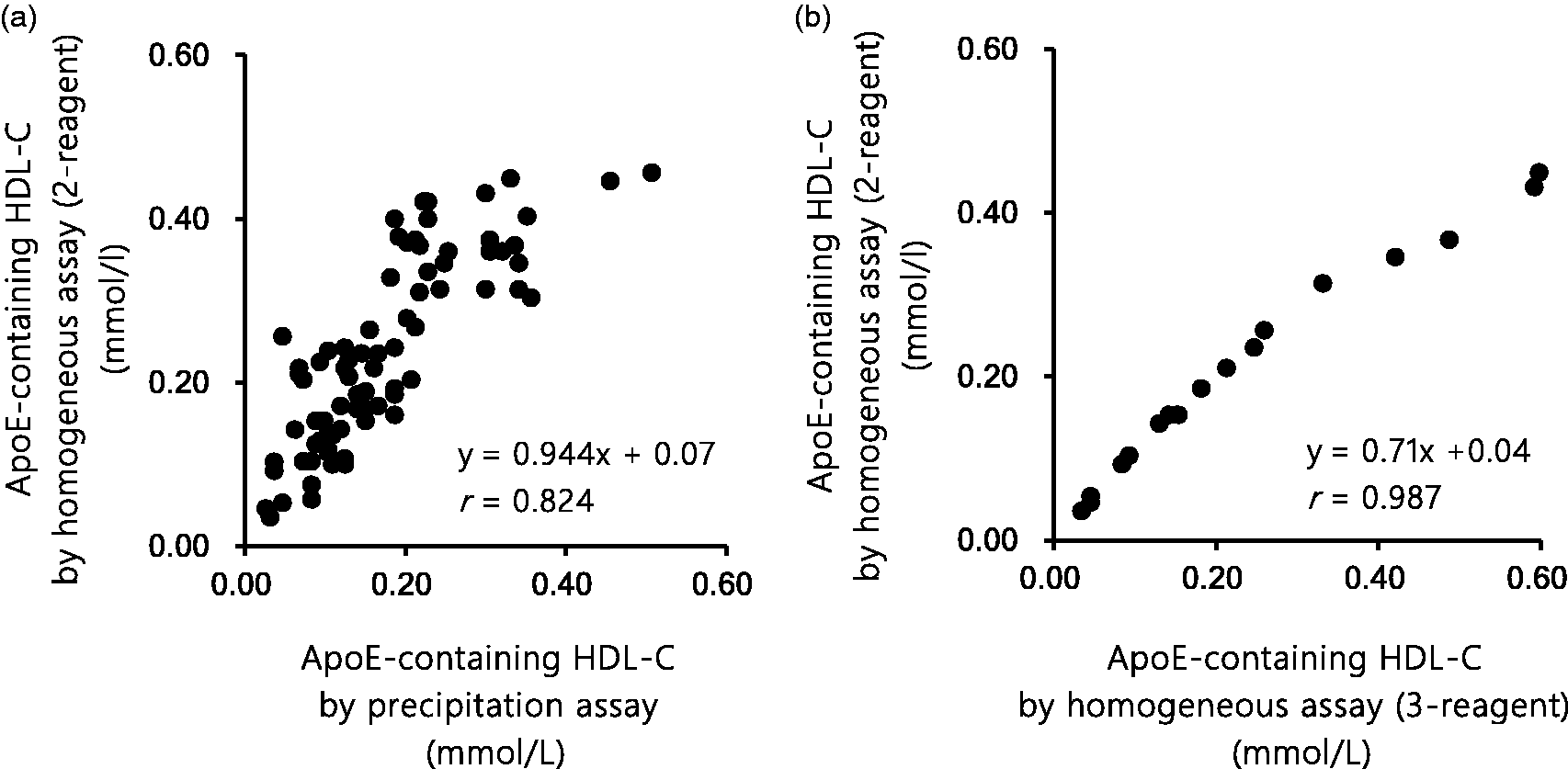
Correlation of the proposed two-step homogeneous apoE-containing HDL-C assay with (a) the control precipitation apoE-containing HDL-C assay or (b) the three-step homogeneous apoE-containing HDL-C assay.
Distribution of serum apoE-containing HDL-C concentration
The serum apoE-containing HDL-C concentration showed a non-parametric distribution by the Shapiro–Wilk test in each study group. Therefore, the Wilcoxon rank-sum test was performed for comparison of its distribution among the study groups. For the healthy individuals (n = 12), the median (25th–75th percentile) of serum apoE-containing HDL-C concentration was 0.05 (0.04–0.10) mmol/L (or 2.1 [1.4–4.0] mg/dL) for the control precipitation assay, while it was 0.10 (0.06–0.13) mmol/L (or 3.8 [2.2–4.9] mg/dL) for the proposed homogeneous assay, showing no statistically significant difference. For the patients with cholestasis (n = 6), on the other hand, it was 0.03 (0.01–0.13) mmol/L (or 1.2 [0.3–5.0] mg/dL) for the control precipitation assay, while it was 0.02 (0.01–0.02) mmol/L (or 0.7 [0.4–0.9] mg/dL) for the proposed homogeneous assay (Figure 6). Thus, the proposed homogeneous assay showed apparently lower results than the control precipitation assay for the cholestatic samples (without statistically significant difference).
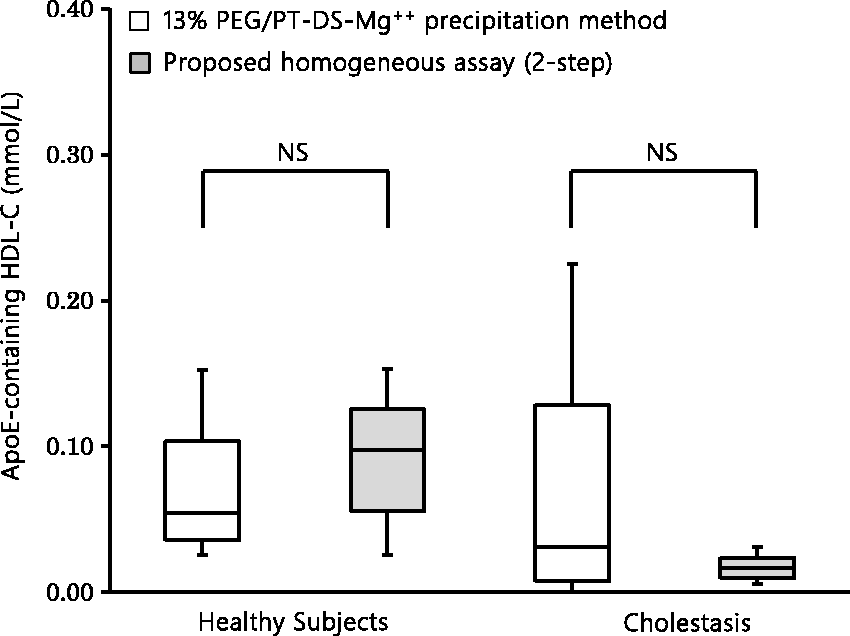
Comparison between the proposed two-step homogeneous apoE-containing HDL-C assay and the control precipitation apoE-containing HDL-C assay in (left) healthy subjects (n = 12, five men and seven women, age 58 ± 16 years, mean ± SD), and (right) patients with cholestasis (n = 6, four men and two women, age 63 ± 11 years).
Discussion
Here, we reported a novel two-reagent homogeneous assay for apoE-containing HDL-C that can run in common autoanalysers and complete the measurement in 10 min (Figure 1). The key detergent, D201, of this assay was first used in our previous study where the three-step homogeneous assay for apoE-containing HDL-C was developed. 31 Reaction of D201 with HDL subpopulations differs depending on its concentrations. 31 On the basis of this knowledge, we applied D201 to the development of the two-step homogeneous assay for apoE-containing HDL-C.
In the first step of the proposed homogeneous assay, non-HDL lipoproteins and apoE-deficient HDL were decomposed by Reagent 1 containing a low concentration (0.05–0.1%) of D201. Solubilized cholesterol was decomposed without colour development. Remaining cholesterol in apoE-containing HDL was solubilized by a higher concentration of D201 (≥0.2%) and subjected to colour development (Figure 2). The basic performance of this assay was excellent in linearity, storage and stability and robustness in the presence of LDL and other coexistent substances (see result section ‘Influence of LDL’ and Figures 3 and 4).
Common autoanalysers are equipped with an operating cycle system suited to popular two-step reagents but not to rare three-step reagents. Moreover, the three-step reagent is troublesome in reagent management: (1) time-consuming reagent exchange work is needed, (2) more attention to reagent deterioration is required and (3) some autoanalysers such as LABOSPECT 008 (Hitachi, Tokyo, Japan) have a unique reagent cartridge that can only use one-step or two-step reagents. Hence, the proposed two-step homogeneous assay is better in the usability in clinical laboratories than our previously reported three-step homogeneous assay. 31
Good correlation between the proposed homogeneous assay and the control precipitation assay supports selectivity of the proposed assay for apoE-containing HDL-C (Figure 5(a)). The proposed two-step homogeneous assay showed a good correlation with the three-step homogeneous assay proposed earlier (r = 0.987). However, we observed slightly lower measurements in the former assay than in the latter at apoE-containing HDL-C concentrations higher than 0.40 mmol/L (15.5 mg/dL) (Figure 5(b)). This discrepancy between the homogeneous assays might be explained by different times used for the elimination of apoE-deficient HDL: 5.0 min for the two-step assay (the first reaction to eliminate all lipoproteins including apoE-deficient HDL other than apoE-containing HDL) and 2.5 min for the three-step assay (the second reaction to eliminate apoE-deficient HDL) (Figure 1). It should be noted that this discrepancy occurs at higher concentrations of apoE-containing HDL-C (>0.4 mmol/L or >15.5 mg/dL) (Figure 5(b)). Such high concentrations of apoE-containing HDL-C are rarely found in physiological conditions, and therefore the proposed two-step homogeneous assay may work well in most clinical cases. The three-step homogeneous assay, however, would be a better choice for serum samples with apoE-containing HDL-C concentration higher than 0.4 mmol/L (or 15.5 mg/dL). This concentration of apoE-containing HDL-C corresponds to 3.07 mmol/L (119 mg/dL) of total HDL-C, according to the equation of correlation in our previous study: y = 0.4715x–1.0475, where y is apoE-containing HDL-C and x is total HDL-C. 31 Such high total HDL-C concentrations are rarely observed in clinical laboratories, except for the cases of CETP deficiency.
As shown in Figure 6, the proposed homogeneous assay and the control precipitation assay showed similar results for the healthy subjects. On the other hand, in the subjects with cholestasis, the proposed homogeneous assay showed lower results than the control precipitation assay, although the difference was not statistically significant. Previous studies reported that cholestatic serum samples showed altered HDL compositions and consequently varied reactivities to HDL-C reagents.33,34 Only the 13% PEG precipitation can recover cholesterol from cholestatic HDL, generating the discrepancy with the proposed homogeneous assay. Low response of the proposed homogeneous assay to cholestatic apoE-containing HDL is a merit of this assay, since cholestatic apoE-containing HDL is known to be poor in the ability to mediate cholesterol efflux from macrophages. 34
In a recent report by Qi et al., 35 apoE-containing HDL-C, measured by homogeneous assay that we developed, was found to be a potential marker for the antiatherosclerotic function of HDL. They suggested that the apoE-containing HDL-C/total HDL-C ratio might work as a measurable marker for early detection of asymptomatic individuals with elevated risk for coronary heart disease, which could mean a clinical significance of measurement of apoE-containing HDL-C. This possibility requires further investigation in large epidemiological or clinical studies, where our proposed two-step homogeneous assay should serve as a useful and convenient tool.
Conclusions
Our newly developed two-step homogeneous assay for apoE-containing HDL-cholesterol could be useful to elucidate the unclear role and regulatory mechanism of apoE-containing HDL-C.
Footnotes
Acknowledgements
The results of this study utilized the Advanced Lipid Analysis Laboratory of the Health Innovation & Technology Center of the Department of Health Sciences of Hokkaido University. We are very grateful to have been given the opportunity to make use of such a facility and experimental environment.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: YI is an employee of Denka Seiken Co., Ltd. The technology used in this study has been patented within Japan (5813284) and the United States (9145578).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Ministry of Education, Culture, Sports, Science, and Technology (MEXT) Regional Innovation Strategy Support Program Sapporo Health Innovation ‘Smart-H’. The principal investigator, HC, was also in receipt of joint research funding from Denka Seiken Co., Ltd.
Ethical approval
This study was approved by the Ethics Committees of the Faculty of Health Sciences, Hokkaido University (08–78) and the Sapporo City General Hospital (H21–035-108).
Guarantor
HC.
Contributorship
YT, HC and S-PH researched literature and conceived the study. YT and TS were involved in lipid measurements and data analysis. YT, NW, AN and MF were involved in providing the samples and patient recruitment. YT and YI were involved in the development of HDL-cholesterol subfractions using homogeneous assay. YT wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
