Abstract
Background
Vitamin E is routinely supplemented to preterm babies, including those with neonatal sepsis. Our aim was to examine the effects of neonatal sepsis and vitamin E on antioxidative system (AOS) in the blood.
Methods
A prospective, randomized, open label study involved 65 preterm neonates (control/sepsis – 34/31), which were divided into two subgroups – non-supplemented and supplemented with vitamin E (25 IU/day for 60 days). The activities of superoxide dismutase, catalase (CAT), glutathione peroxidase (GPx), and glutathione reductase (GR) were determined in erythrocytes at days 0, 30, and 60, following sepsis diagnosis.
Results
There was no difference in the activity of AOS between controls and neonates with ongoing sepsis. At 60 days, septic neonates showed higher CAT activity compared to controls (P = 0.027), and lower GPx activity compared to 0 days (P = 0.022). The later was mitigated by vitamin E, which on the other hand provoked lower GPx activity at 30 days, compared to untreated septic neonates (P = 0.014). In addition, vitamin E suppressed GR activity in septic neonates (P = 0.025 and P = 0.017 at 30 and 60 days). Finally, vitamin E supplementation in control neonates provoked a significant increase of GPx activity (P = 0.015 at 60 days).
Conclusions
The absence of altered redox settings in the blood of neonates during sepsis episode, and vitamin E-provoked decrease in the activity of some components of AOS, suggest that the supplementation of vitamin E in these patients might not be rational.
Introduction
Current recommendation is that preterm infants should be routinely supplied with vitamin E.1,2 Preterm infants have less vitamin E reserves, and early supplementation of vitamin E is aimed at correcting the deficiency and at preventing adverse effects of inadequate antioxidative status. 2 In line with this, the supplementation of vitamin E decreases the risk of some prematurity-related conditions (e.g. intracranial hemorrhage, severe retinopathy, and blindness). However, it also significantly increases the risk of infection and sepsis. 1 This problem is exacerbated by a very high incidence of sepsis in preterm neonates in general (e.g. the risk for preterm neonates with birth-weight between 1000 and 1500 g in developed countries is 10%, which is around tenfold higher compared to term newborns). 3 High susceptibility to sepsis could be attributed to the immaturity of neonatal immune system, which shows low capacity to produce reactive oxygen species in response to the presence of pathogens. Immune system uses reactive oxygen species to damage and kill pathogens, so the application of extracellular antioxidants, such as vitamin E, might further suppress poor bactericidal capacity of blood of preterm neonates.4,5
Pathological events in sepsis are interconnected into a self-sustaining and self-promoting pathological process named the ‘sepsis redox cycle’, which may result in the total loss of cellular function. 6 The pathogenesis of neonatal sepsis has been related to specific redox changes taking place in the intracellular compartment of different cells and tissues (the process has been marked as ‘neonatal sepsis redox cycle’). 5 It is important to note that preterm septic neonates are particularly susceptible to a wide spectrum of different morbidities, such as respiratory distress syndrome, bronchopulmonary dysplasia, periventricular leukomalacia, severe intraventricular hemorrhage, cerebral palsy, and vision and hearing impairments.7,8 For example, the incidence of all morbidities taken together in one retrospective cohort study on low birth weight neonates that survived septic shock has exceeded 70%. 9 The development of all of these morbidities has been related to oxidative stress,10–13 and to a high susceptibility of developing and proliferating cells in specific organs to oxidative injury and apoptosis. 5 Pertinent to this, it appears reasonable to apply antioxidants in order to treat neonatal sepsis and to prevent the development of morbidities. However, the main site of antioxidative activity has to be taken into account. Coupled with ascorbate, vitamin E generally acts as extracellular antioxidant in the blood, 14 so the rationale for its application in neonatal sepsis is not clear. Cherian and co-workers have reported that there are no significant differences in the activity of antioxidative system (AOS) enzyme – superoxide dismutase (SOD), the levels of lipid peroxidation and glutathione (GSH) in erythrocytes, and plasma ascorbate concentration, in septic babies compared to age-matched controls. 15 One study has shown only a transient increase of total hydroperoxide level in the blood of septic newborn piglets, while biological antioxidant potential was even increased at some time points during the 6-h monitoring period. 16 Another group observed, in two studies, merely a slight increase in the level of lipid peroxidation products in the serum of septic neonates compared to controls.17,18 They have also found increased serum activities of AOS enzymes – SOD, catalase (CAT), and GSH peroxidase (GPx) in septic neonates. However, these results appear to be misleading, since the authors did not take into account that the levels of AOS enzymes in human serum are drastically lower (one to four orders of magnitude) in comparison to erythrocytes.19–22 Therefore, the reported differences could represent a consequence of lysis of a small number of erythrocytes, which might be easily provoked by pathogens. 23 It is worth mentioning that the proper way to determine redox settings in the blood is to measure AOS activity in erythrocytes.24,25
The aim of our study was to determine the effects of sepsis and vitamin E supplementation on redox settings in the erythrocytes of preterm neonates, during and following the sepsis episode.
Materials and methods
Study design
A prospective, randomized, open label study was conducted at the Department of Clinical Pharmacology, School of Medicine, University of Belgrade in collaboration with the Institute for Neonatology, Belgrade, from October 2008 to September 2009. Protocol was approved by the hospital ethics committee. Inform consents were obtained from all parents.
Neonates with sepsis were included in study by the following criteria: (i) birth weight < 2500 g; (ii) gestational age ≥ 32 and <38 weeks; (iii) the absence of malformations, inborn errors of metabolism, and life threatening conditions (other than sepsis); (iv) the absence of multiple episodes of sepsis; and (v) no hospitalization in another unit for more than 24 h. The diagnosis of sepsis included both, clinical signs of sepsis recognized and explicitly stated by the intensive-care physician and biological signs of sepsis, positive hemoculture and/or at least two of the following blood parameters: positive C-Reactive Protein >5 mg/L; WBC count >25 × 1012/L; WBC count <4 × 1012/L; immature to total neutrophil (I/T) ratio >0.16 in the first day of life, and >0.12 at any other time. Controls (preterm neonates without sepsis) were matched for gestation and birth weight. Both control and sepsis group were divided into two groups: one group received 25 IU/day of orally supplemented dietary vitamin E (Natural vitamin E-Oil, Natural Wealth, NBTY Inc., NY, USA) during the period of 60 days (+vit E group), while the other group did not receive vitamin E (−vit E group), in addition to the recommended pharmacotherapy treatment (Figure 1).
Study design. n is the number of neonates in each (sub)group.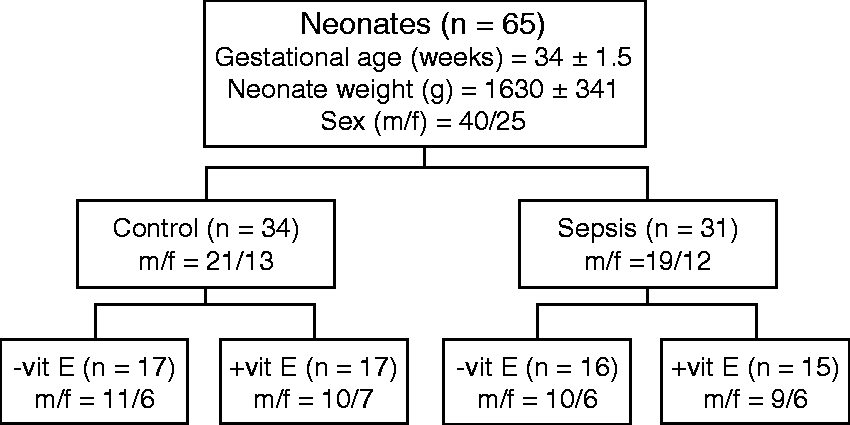
Biochemical analysis
Venous blood samples (1 mL) were obtained in heparinized tubes from each participant at the initial admittance into the unit, and 30 and 60 days after the first collection. Samples were immediately centrifuged at 2000 g/10 min/4℃. Supernatants and buffy coats were carefully removed. Separated erythrocytes were washed three times with 0.9% NaCl at 4℃, snap-frozen in liquid nitrogen, and stored at −80℃.
Erythrocytes (0.3 mL) were lysed by adding 3 mL of ice-cold distilled water. Hemoglobin (Hb) concentration was measured by Drabkin method. This procedure is based on the oxidation of hemoglobin and its derivatives to methemoglobin in the presence of alkaline potassium ferricyanide. Methemoglobin reacts with potassium cyanide to form cyanmethemoglobin, which has maximum absorption at 540 nm. The absorbance at 540 nm is proportional to the total hemoglobin concentration. SOD activity was determined by the adrenaline method. 26 One unit of activity is defined as the amount of enzyme that decreases the rate of adrenalin auto-oxidation at pH 10.2 by 50%. Interference with Hb was eliminated by precipitation prior to the assay using ethanol/chloroform (1 : 1, v/v) that was followed by centrifugation at 5000 g/5 min/4℃. The activity of CAT was determined according to the method of Beutler. 27 One unit is defined as the amount of enzyme that reduces 1 µM of H2O2/min. The activities of GPx and GSH reductase (GR) were determined as described previously.28,29 All the chemicals used in this study were purchased from Sigma-Aldrich (St. Louis, MO, USA) and Merck (Darmstadt, Germany).
Statistical analysis
All performed laboratory measurements were blind to the investigators. Statistical differences between each group of data were determined by non-parametric two-tailed Mann–Whitney test using STATISTICA 6.0 (StatSoft Inc., Tulsa, OK, USA). Results were taken to be statistically different if P <0.05. The results are presented as mean values ±SD. P values are presented only if statistical difference was determined.
Results
The baseline characteristics of 65 preterm neonates, which completed the study, are presented in Figure 1. Among septic preterm neonates, positive hemoculture was determined in 10: five neonates were positive for Streptococcus β-haemolyticus, three for Escherichia coli, one for Staphyloccus aureus, and one for Klebsiella pneumonia. During sepsis (0 days), there was no difference in the activity of AOS compared to non-septic neonates. SOD activity did not show any significant changes in any of the patient groups (Figure 2(a)). Two months after sepsis episode, septic infants showed higher CAT (Figure 2(b)) activity compared to control infants. The supplementation of vitamin E did not prevent this change. Even more, it appears that vitamin E potentiated sepsis-related changes in CAT activity at 30 days.
The activity of superoxide dismutase and catalase in erythrocytes obtained from control (white columns) and sepsis group of neonates (gray columns), non-supplemented (no pattern) or supplemented with vitamin E (pattern); before the supplementation of vitamin E (0 days) and at 30 and 60 days of treatment. (a) Activity of superoxide dismutase and (b) activity of catalase. Statistically significant differences (P < 0.05) are presented.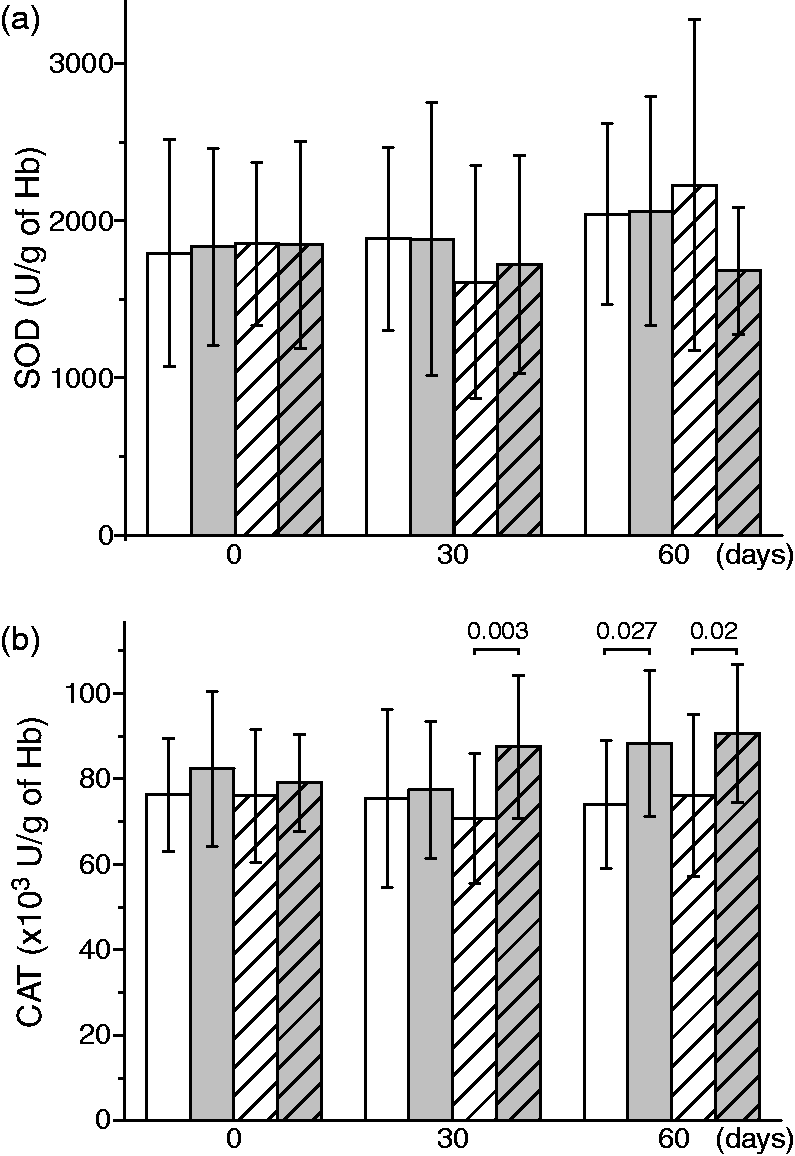
GPx activity in untreated controls remained unaltered over the period of two months, whereas in untreated septic neonates it showed a slight decrease at 60 days compared to 0 days (Figure 3(a)). The later was mitigated by vitamin E supplementation, which on the other hand provoked a drop of GPx activity in +vit E group at 30 days compared to −vit E group of septic neonates. Finally, the supplementation of vitamin E resulted in significantly increased GPx activity in control neonates over the period of 60 days. Figure 3(b) shows that both −vit E groups (control and sepsis) exhibited gradual increase of GR activity during the period of two months. However, the supplementation of vitamin E to septic neonates prevented this process, as implicated by the lower levels of GR activity in +vit E group at 30 and 60 days compared to untreated septic neonates.
The activity of GPx and reductase in erythrocytes obtained from control (white columns) and sepsis group of neonates (gray columns), non-supplemented (no pattern) or supplemented with vitamin E (pattern); before the supplementation of vitamin E (0 days) and at 30 and 60 days of treatment. (a) activity of GSH peroxidase and (b) activity of GSH reductase. Statistically significant differences (P < 0.05) are presented.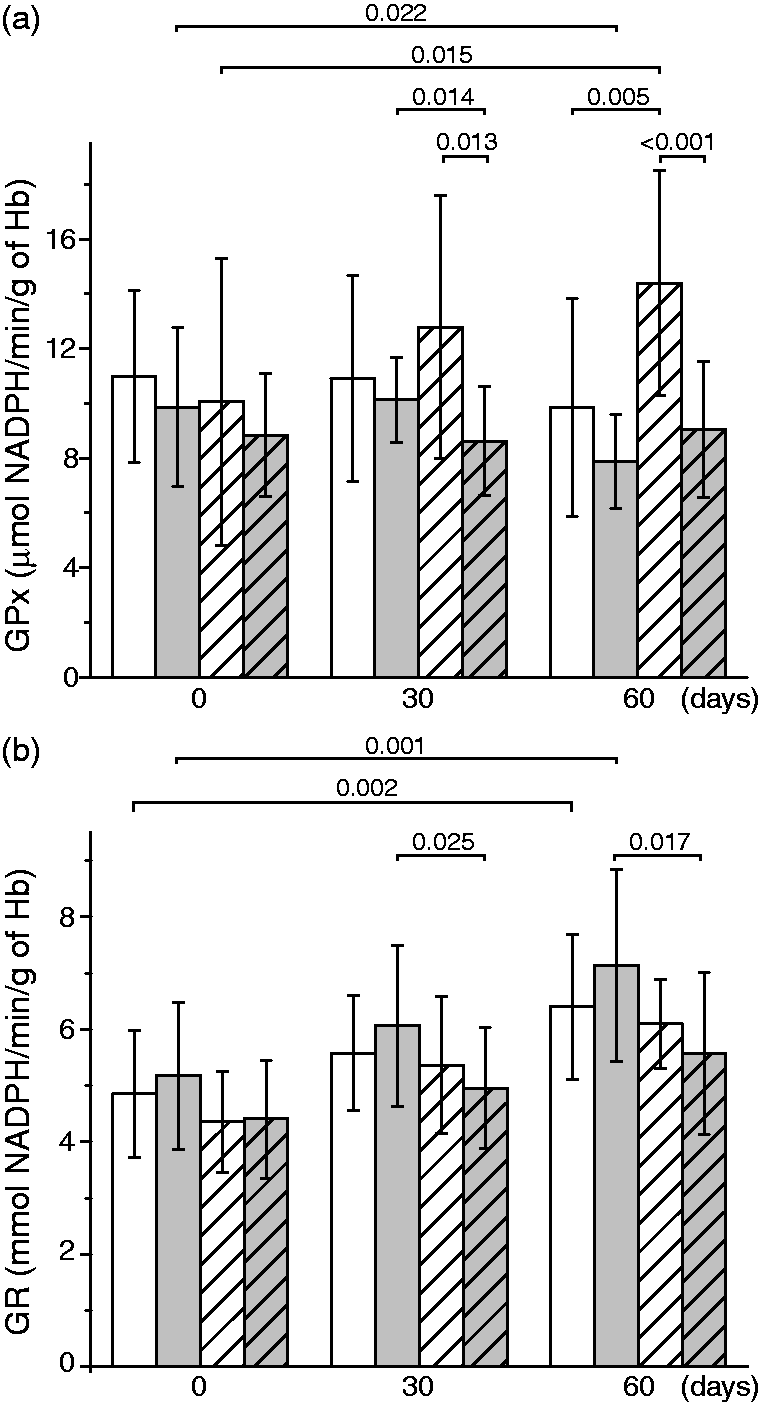
Discussion
The results presented here shed a new light on several important issues regarding the pathophysiology of neonatal sepsis and the antioxidative therapy of preterm neonates. We found that neonatal sepsis is not related to significant acute redox changes in the blood, which is line with some previous assertions. 15 This implies that the application of extracellular antioxidants in neonatal sepsis treatment might be futile. However, the activity of AOS enzymes in the period after sepsis episode was altered, showing a specific profile – a significantly increased CAT activity and decreased GPx activity (at 60 days) compared with neonates who have not experienced sepsis. The changes of redox settings in the blood following sepsis episode might have their share of responsibility for the development of neonatal sepsis-related morbidities. Both enzymes (CAT and GPx), the activities of which is opposingly altered in neonatal sepsis survivors, remove hydrogen peroxide (H2O2) in erythrocytes. CAT is the primary erythrocyte enzyme responsible for coping with exogenous (e.g. produced during inflammation), but also with endogenous (produced via oxyHb auto-oxidation) H2O2.30–32 However, GPx plays a specific role in the protection of erythrocyte membrane and Hb from oxidative damage. GPx removes H2O2 which is produced by auto-oxidation of membrane-bound oxyHb. This distinct compartment is inaccessible to CAT. 32 In addition, GPx is known to remove lipid hydroperoxides. Heme degradation is particularly promoted if the level of GSH (which is needed for GPx function and maintained by GR activity) is low. The supplementation of vitamin E appears to potentiate sepsis-related changes in CAT activity, but it mitigated the decrease of GPx activity. On the other hand, the positive effects on GPx are most likely annihilated by vitamin E-provoked decrease of GR activity, which results in lower GSH levels. The resulting suppression of coupled GPx/GR activity in erythrocytes might lead to facilitated aging process in erythrocytes, membrane oxidative damage, hemolysis, and heme degradation.32–35
It has been proposed that erythrocytes act as a sink for extracellular H2O2. 36 We showed that vitamin E up-regulates GPx activity and, consequently, the H2O2-removing capacity of blood of control neonates. It is noteworthy that the positive correlation between the serum level of α-tocopherol and GPx activity in erythrocytes in healthy neonates has been established previously. 37 The combination of direct radical-scavenging activity of vitamin E and the increased capacity of erythrocytes to remove H2O2 in neonates might weaken bactericidal potential of blood, since H2O2 is used by the innate immune system to kill pathogens. This may account for the relation between vitamin E supplementation and the increased risk of infection and sepsis in neonates.
Different mechanisms by which vitamin E might affect AOS in erythrocytes are plausible. Human erythrocytes are anucleate when mature, which means there is no de novo synthesis of (AOS) enzymes. Therefore, in mature erythrocytes vitamin E might act by affecting the complex network of redox regulation of AOS enzymes.24,25 However, vitamin E-provoked increase in GPx activity in controls showed a gradual increase over the two months. During this period of time, a significant portion of erythrocytes in the blood is replaced by the new cells, which are formed in the bone marrow. At this level, vitamin E could affect gene expression resulting in altered AOS profile. 38 Pertinent to our study, there are several reports on vitamin E-related increase of expression of GPx in different cell types.39–41 It is worth mentioning that vitamin E is involved in the regulation of expression of genes, other than those that are relevant for redox processes, such as cellular traffic. The role of vitamin E in cellular trafficking could best explain the neurological symptoms seen in vitamin E deficiency. 38
Alternatively to dietary supplementation, the level of vitamin E vitamin could be increased in breast milk by modifying mother’s diet. This approach could potentially eliminate any need for the application of supplements to vitamin E-deficient preterm neonates. The level of vitamin E in breast milk could be increased by higher fat and sufficient vitamin E intake.42,43 It is worth mentioning that it appears that vitamin E supplementation to exclusively breast-fed preterm neonates does not result in increased blood level of vitamin E. 44 Finally, prematurity-related vitamin E deficiency might be amortized by the supplementation of this vitamin during the course of pregnancy. 45 In summary, results in this study imply that on the account of neonatal sepsis the supplementation of 25 IU/day of vitamin E appears futile, whereas its application in non-septic preterm neonates might increase the risk of infection and sepsis via redox mechanisms. However, the limitations of the present study were small number of patients and one dosage of vitamin E. Altogether, pros and cons should be carefully weighted in each case before deciding whether vitamin E is to be supplemented or not.
Footnotes
Declaration of conflicting interests
None declared.
Funding
This work was supported by the Ministry of Education and Science of the Republic of Serbia, grant numbers OI 173014 and III 43004.
Ethical approval
Ethical committee of the Institute for Neonatology, Belgrade, Serbia (09/2008).
Guarantor
MB.
Contributorship
MB designed the study, performed antioxidant enzymes measurement and data analyses, and wrote the manuscript. BO followed up patients during the study. NBP followed up patients during the study. SS performed antioxidant enzymes measurement and reviewed/edited the manuscript. IS performed statistical analyses, made the figures, and wrote the manuscript.
