Abstract
Background
The aim of this study was to measure the parameters of oxidative stress in the blood of patients with post-traumatic stress disorder.
Methods
The study included 80 male war veterans who participated actively in the Homeland war in Croatia. Volunteers were divided into two groups: 50 veterans diagnosed with post-traumatic stress disorder and 30 without diagnosis. The self-assessment Hospital Anxiety and Depression Scale and the Beck Depression Inventory were used to detect the severity of depression and anxiety in the post-traumatic stress disorder patients. Catalytic concentrations of superoxide dismutase and glutathione peroxidase in erythrocytes and the concentration of malondialdehyde in serum were measured spectrophotometrically.
Results
Although the catalytic concentrations of erythrocyte superoxide dismutase and erythrocyte glutathione peroxidase were within the reference range for both groups, the values obtained for the post-traumatic stress disorder group were significantly lower (P < 0.001). For serum malondialdehyde concentrations, no statistically significant differences between the groups were found.
Conclusions
Lower catalytic concentrations of erythrocyte superoxide dismutase and erythrocyte glutathione peroxidase in patients with post-traumatic stress disorder may indicate a weaker response to oxidative stress due to impaired enzyme activity and/or decreased synthesis. Conversely, no significant changes in serum malondialdehyde concentrations suggest a compensated balance and adaptive response to (oxidative) stress.
Introduction
Free radicals, atoms, molecules or ions with an unpaired number of electrons are physiological products of metabolism which directly participate in the protection against infection, or act as coordinators of inflammatory response. 1 In a healthy organism, the production and various interactions of free radicals are tightly controlled by enzymatic defence mechanisms such as the superoxide dismutase (SOD), glutathione peroxidase (GPx) and catalase (CAT), or via the action of non-enzymatic antioxidants such as ascorbic acid, Vitamin E and glutathione. 2 However, excessive production of free radicals and/or impaired protection mechanisms lead to oxidative stress and result in radical-induced damage of lipids, proteins and DNA. 3 Tissue damage caused by oxidative stress includes a diversity of biological phenomena such as aging, carcinogenesis, atherosclerosis and neurodegeneration. 4 The severity of oxidative stress can be measured via the concentration of reactive oxygen/nitrogen species (ROS/RNS) or the concentration of antioxidants. Furthermore, biomolecules derived through the action of free radicals can be used as markers of oxidative stress: damaged DNA/RNA (i.e. 8-hydroxy(deo)guanosine), the oxidation/nitration of proteins (i.e. protein carbonyl content) and the products of lipid peroxidation (i.e. 4-hydroxy-2-nonenal, HNE; malondialdehyde, MDA).
Lipid peroxidation, defined as the oxidative deterioration of lipids containing any number of carbon–carbon double bonds, is considered to participate significantly in the creation of oxidative stress, since polyunsaturated fatty acids (PUFAs) frequently serve as a target for free radicals resulting in their multiplication through a cascade of reactions. 1 The primary products of lipid peroxidation, which include conjugated dienes and hydroperoxides, are relatively unstable and decompose into secondary products such as alcohols, aldehydes and ketones. Although numerous state-of-the-art methods have been developed, the concentration of overall lipid peroxidation is still most frequently assessed as the concentration of serum malondialdehyde (sMDA) which is determined by a reaction with thiobarbituric acid (TBARS Assay).
Lipids comprise about 60% of the brain’s dry weight with a high ratio of PUFAs, and the brain utilizes more than 20% of total O2 consumed by the body, which is why the brain is highly susceptible to lipid peroxidation that might result in serious damages and loss of function.5–7 Oxidative stress may alter neurotransmission patterns, neuronal function and brain activity in general, and can also disrupt the membrane integrity.8,9 Both the importance of fatty acid composition in brain cell membranes and the involvement of PUFAs in the development of various psychiatric disorders have been intensively studied.10–12 A significant number of these results point to a relationship between the lack of essential PUFAs, particularly eicosapentaenoic (EPA, C20:5n-3) and docosahexaenoic (DHA, C22:6n-3), and a variety of psychiatric symptoms and disorders such as stress, anxiety, cognitive impairment, mood disorders and schizophrenia. In our previous study, 13 a significant negative correlation between plasma EPA concentration and the severity of clinical symptoms in patients with post-traumatic stress disorder (PTSD) was found.
According to recently released DSM V criteria, 14 PTSD is classified as a trauma- and stressor-related disorder caused by a traumatic experience such as exposure to actual or threatened death, serious injury or sexual violation. It is characterized by four major symptom clusters: re-experiencing, arousal, avoidance and persistent negative alterations in cognitions and mood. The evolvement of PTSD results in impairments in the individual’s social interactions, work capability or everyday functioning, although it is unclear why certain individuals develop PTSD as a reaction to traumatic incidents while others do not.
Since oxidative stress is a general phenomenon and the markers of oxidative damage in the central nervous system correlate well with those in peripheral materials, the majority of analyses were performed on easily accessible materials such as blood, saliva and cerebrospinal fluid. 5 A substantial number of authors found increased amounts of markers of oxidative stress in plasma/serum, and/or deviations in the activity of defence enzymes in patients suffering from psychiatric disorders.3,7,15–19 A recent review by Hovatta et al. 20 analysed available data on the relationship between oxidative stress and anxiety, depression and co-morbid disorders, and the possible mechanisms of impaired defence against oxidative stress. Stress-induced alterations in the concentration of neurotransmitters such as excitatory amino acids, serotonin and gamma-amino-butyric acid can modulate the immune response and vice versa, and cytokines, mediators produced by immune cells, maintain homeostasis by communicating with other biological systems such as the hypothalamic-pituitary-adrenal (HPA) axis.21,22
Recently, Kuloglu et al.23,24 recognized a link between oxidative stress and some anxiety disorders (obsessive-compulsive and panic disorder), demonstrating that oxidative metabolism can influence anxiety regulation. Bouayed et al. 25 in their review summarized data which demonstrate a link between oxidative stress and anxiety, but an unambiguous cause-effect relationship has yet to be determined. Some of the reviewed studies suggest oxidative stress causes anxiety-related behaviours but none clarify the underlying, specific mechanisms in detail.
The risk factors for developing PTSD appear to be related to a combination of genetic predisposition and environmental factors.26,27 It was first recognized among Vietnam War veterans but was also subsequently diagnosed in people expressing similar symptoms in response to various traumas, whether in war or civil life. 28 During the 1990s, the Homeland War struck Croatia. This unfortunate experience resulted in a vast number of individuals suffering from PTSD. Since PTSD has some clinical symptoms similar to those found in depression, bipolar disorder, schizophrenia and other psychiatric disorders, the present study investigates whether similar changes in oxidative/antioxidative balance could be confirmed. Certain lifestyle and nutritional habits, which are possible cofactors in the processes of oxidative stress, were also observed in the study participants.
Subjects and methods
Subjects
The study included 80 male Croatian Homeland War veterans. Fifty veterans were outpatients from University Psychiatric Hospital Vrapče who were diagnosed with PTSD according to the Diagnostic and Statistical Manual of Mental Disorders (DSM-IV), 29 and the International Statistical Classification of Diseases and Related Health Problems (ICD-10) 30 diagnostic criteria. The control group included 30 participants who had also served in the Homeland War, but did not develop PTSD. The diagnosis evaluation was performed by experienced psychiatrists in order to confirm the PTSD diagnoses according to the DSM-IV and ICD-10. Although an updated edition of the manual (DSM-V) 14 was released in the meantime, where PTSD is detached from anxiety disorders and included in a new section of trauma- and stressor-related disorders, all our patients met the criteria for PTSD diagnosis according to both DSM IV and V. The self-assessment Hospital Anxiety and Depression Scale (HADS) 31 and the Beck Depression Inventory (BDI-II) 32 were used to detect the severity of depression and anxiety in the PTSD patients. The 21 BDI-II items, as well as 14 items from the HADS, are each scored on a four-point scale from 0 = ‘no symptoms’ to 3 = ‘extreme symptoms’. The sum of the first seven items from the HADS is the severity index for anxiety, and the sum of the last seven is the index for depression. The severity of PTSD was evaluated by a clinician-rated instrument, i.e. the Clinical Global Impression – Severity scale (CGI-S). 33 The severity of the patient’s PTSD is assessed by CGI-S in a seven point range: from 1 (‘reference, not at all ill’) to 7 (‘extremely ill’). All the participants were free of all medication for at least two weeks prior to entering the study, with the exception of veterans with PTSD who were treated with an antidepressant (sertraline, 50 mg/day) for at least two months. Ten of the PTSD patients were also on therapy with anxiolytics (4 – diazepam; 3 – alprazolam; 3 – lorazepam). Exclusion criteria were: alcohol and substance abuse or dependence, food supplementation (i.e. vitamins E and/or C), presence of a severe neurologic disorder, presence of an infectious disease or presence of a severe physical disorder. The study protocol was approved by the Ethics Committees of the University Psychiatric Hospital Vrapče and the School of Medicine University of Zagreb, Croatia. Prior to entering the study all participants signed an informed consent, and afterwards completed a general health questionnaire. 34
Blood sample collection
Vein blood samples were collected after overnight fasting into 5 mL vacutainer tubes (Becton Dickinson, Franklin Lakes, NJ, USA) containing ethylenediaminetetraacetic acid. The samples were then centrifuged at 3000 rpm for 10 min at 15℃ to separate the plasma from blood cells. After washing procedures, erythrocytes isolated from blood samples were haemolysed by the freezing and thawing sequence.
Measurement of antioxidant enzymes activity
Haemolysates of erythrocytes were used to measure catalytic concentrations of erythrocyte glutathione peroxidase (eGPx) (EC 1.11.1.9) and erythrocyte superoxide dismutase (eSOD) (EC 1.15.1.1), as well as haemoglobin (Hb) concentration. The eGPx activity in erythrocyte haemolysates was measured using a commercial RANSEL kit and eSOD activity was measured with RANSOD reagent (both from Randox Laboratories Ltd., Dublin, Ireland) on SABA 18 auto-analyzer (AMS, Rome, Italy). The eGPx activity, expressed in nanomoles of nicotinamide adenine dinucleotide phosphate oxidized per minute, was measured by a coupled modification of the assay procedure of Paglia and Valentine. 35 The assay for eSOD activity was based on the method described by Woolliams et al., 36 using xanthine and xanthine oxidase to produce superoxide radicals which then react with nitroblue tetrazolium to form a red formazan dye. The inhibitory effects of eSOD on the initial rate of this reaction were measured, and one unit of SOD activity is that which causes a 50% inhibition of the rate of reduction of the substrate under the conditions of the assay. In haemolysates, a mass concentration of haemoglobin was also measured (Herbos Dijagnostika, Sisak, Croatia) and eGPx and eSOD catalytic concentrations were expressed in terms of U/g Hb.
Determining sMDA concentrations
Serum MDA concentrations were measured using the modified TBARS method described by Anguloa et al. 37 and Botsoglou et al. 38 The results were calculated and expressed as concentration of MDA (µmol/L) using the extinction coefficient of 156,000 Lmol−1 cm−1. 39
Laboratory methods
Serum triacylglycerols (TG), total cholesterol (T-CH) and high-density lipoprotein cholesterol (HDL-CH) were measured by enzymatic colorimetric methods (Roche Diagnostics Corporation, Indianapolis, IN, US). Low-density lipoprotein cholesterol (LDL-CH) was calculated as described by Friedewald et al. 40
Statistical analysis
The obtained data were evaluated with STATISTICA 10 software (StatSoft, Inc., Tulsa, Oklahoma, USA). The data distribution was tested with Kolmogorov–Smirnov and Shapiro–Wilk’s W test. Descriptive statistics were performed, and the results are presented as mean ± standard deviation (mean ± SD). The results were processed by Student’s t-test, Mann–Whitney and Chi-square test, and the correlation of study parameters was tested by Spearman rank order correlations. The observed differences were considered statistically significant at the level of P < 0.05.
Results
Demographic data
Sociodemographic characteristics of the study participants.
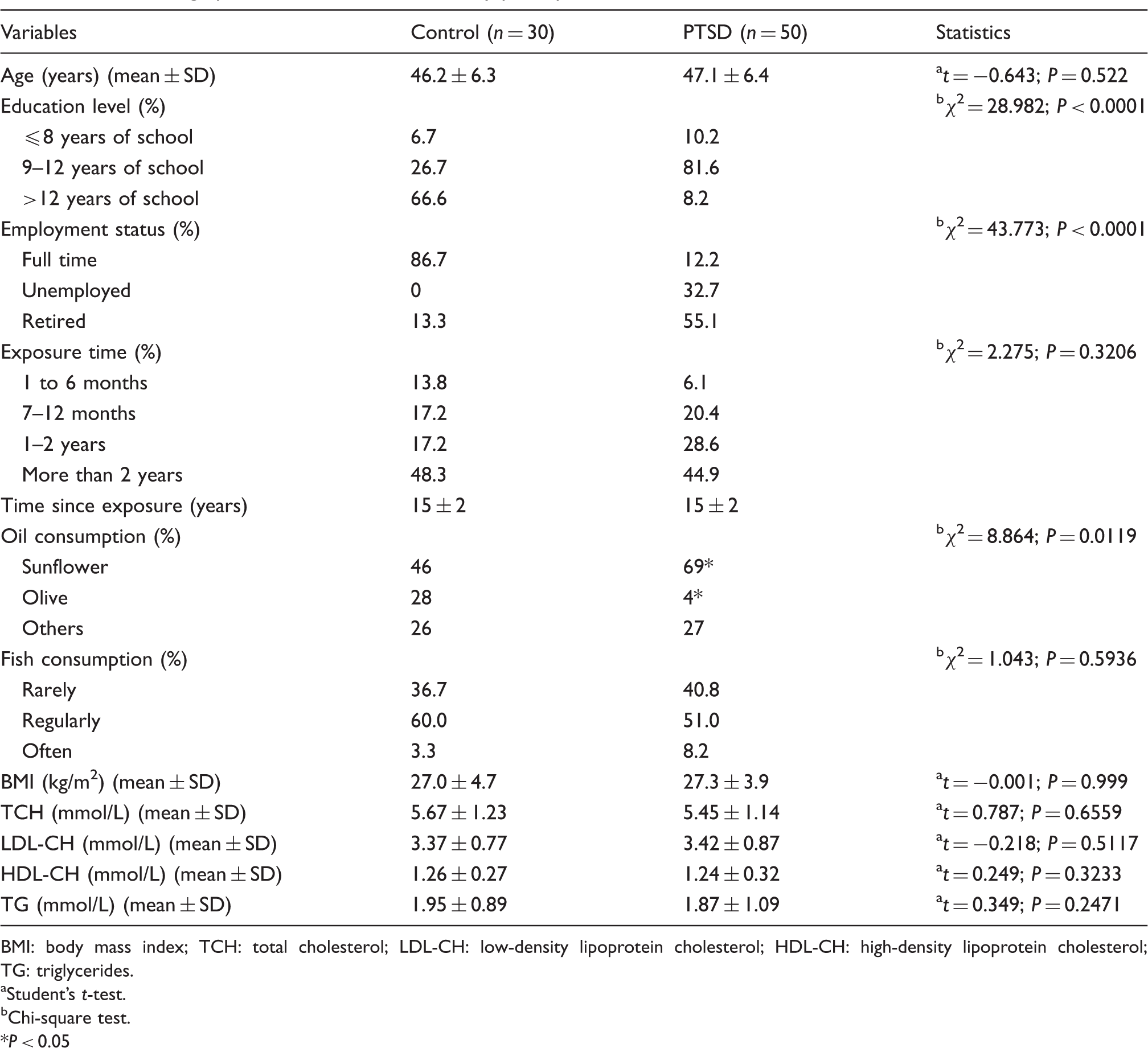
BMI: body mass index; TCH: total cholesterol; LDL-CH: low-density lipoprotein cholesterol; HDL-CH: high-density lipoprotein cholesterol; TG: triglycerides.
Student’s t-test.
Chi-square test. *P < 0.05
Lifestyle and nutritional habits
Data on nutritional habits included information about the body mass index (BMI) as well as the consumption of fish and various oils (Table 1). The mean BMI value was similar for both groups. Regarding the oil type, sunflower oil was preferred in both groups with significantly higher consumption frequencies recorded in patients with PTSD (P = 0.0203). However, the control group consumed significantly more olive oil compared to the PTSD group (P = 0.0025). For fish consumption, no significant differences were found between the groups. Analysis of laboratory data revealed no significant differences in lipid parameters between the two groups.
Severity of depression and anxiety in PTSD participants
The comparison between the severity of PTSD, depression and anxiety in the PTSD group is presented in Figure 1. The total sum of points from BDI-II is originally assembled in four subscores in the following order: minimal depression (0–11 points); mild depression (12–19 points); moderate depression (20–26 points); and severe depression (≥27 points). However, since HADS gives three subscores: (1) no symptoms (0–7 points); (2) moderate symptoms (8–10 points); and (3) severe symptoms (11–21 points), for the purpose of this study the first two subscores from BDI-II have been summarized into one combined subscore, to ensure data comparability. The PTSD severity in participants, evaluated by CGI-S, ranged between the following four subscores: mildly ill, moderately ill, markedly ill and severely ill. For the purpose of this study, the last two subscores (markedly ill and severely ill) have been summarized into one combined subscore. Statistical tests confirmed a strong positive correlation between the anxiety (HADS-A) and depression severity index (HADS-D) obtained by HADS alone (r = 0.741, P < 0.05). Positive correlations were also observed between the results obtained by the three different scales: BDI-II versus HADS-A (r = 0.835, P < 0.05), BDI-II versus HADS-D (r = 0.716, P < 0.05) and between CGI-S versus BDI-II (r = 0.53, P < 0.05).
Comparison between the scores in PTSD patients obtained by Beck Depression Inventory (BDI-II), Hospital Anxiety and Depression Scale for anxiety (HADS-A) and depression (HADS-D), and Clinical Global Impression-Severity scale (CGI-S). Values are expressed as percentage of total group.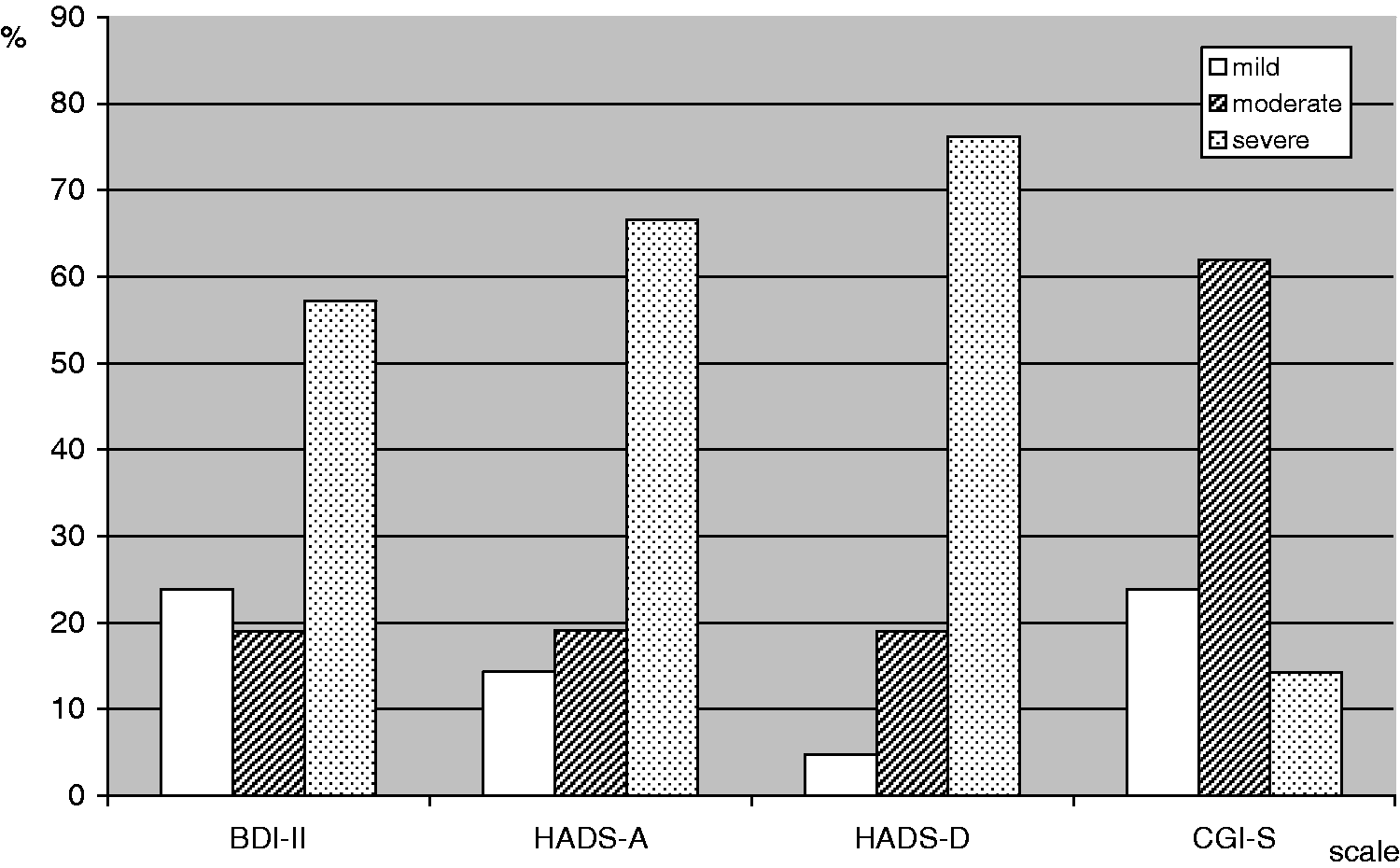
Antioxidant enzymes’ activity and MDA concentrations in the PTSD and control group
Data for the eSOD and eGPx catalytic concentrations and sMDA concentration of the control and the PTSD group are shown in Figure 2. Values for catalytic concentrations of antioxidative enzymes are expressed as U/mg Hb in order to avoid possible errors resulting from erythrocyte washing and dilution. Mass concentrations of Hb were within reference values and did not differ significantly between the two groups. A significant difference was noted between PTSD and the control group in eSOD (t = 4.1307; P = 0.000090) and eGPx (t = 3.53797; P = 0.000683) activities, with lower values exhibited in the PTSD group. No significant differences were found in sMDA concentrations. The data analysis was further focused on relations between the component traits of (anti)oxidative status and serum lipids concentration. A statistically significant correlation was only found for eSOD activity versus serum TCH concentration (r = −0.3322, P < 0.0500) for patients with PTSD. This relationship was not confirmed neither for the control group (r = 0.1384; P > 0.0500), nor for the total group (r = 0.0161; P > 0.0500). Other tested correlations were also not significant (all P > 0.005, data not shown).
Erythrocyte superoxide dismutase (eSOD) and glutathione peroxidase (eGPx) catalytic concentrations and serum lipid peroxide (sMDA) concentration in patients with PTSD and controls included in the study. Values are expressed as mean ± SD. *P < 0.05.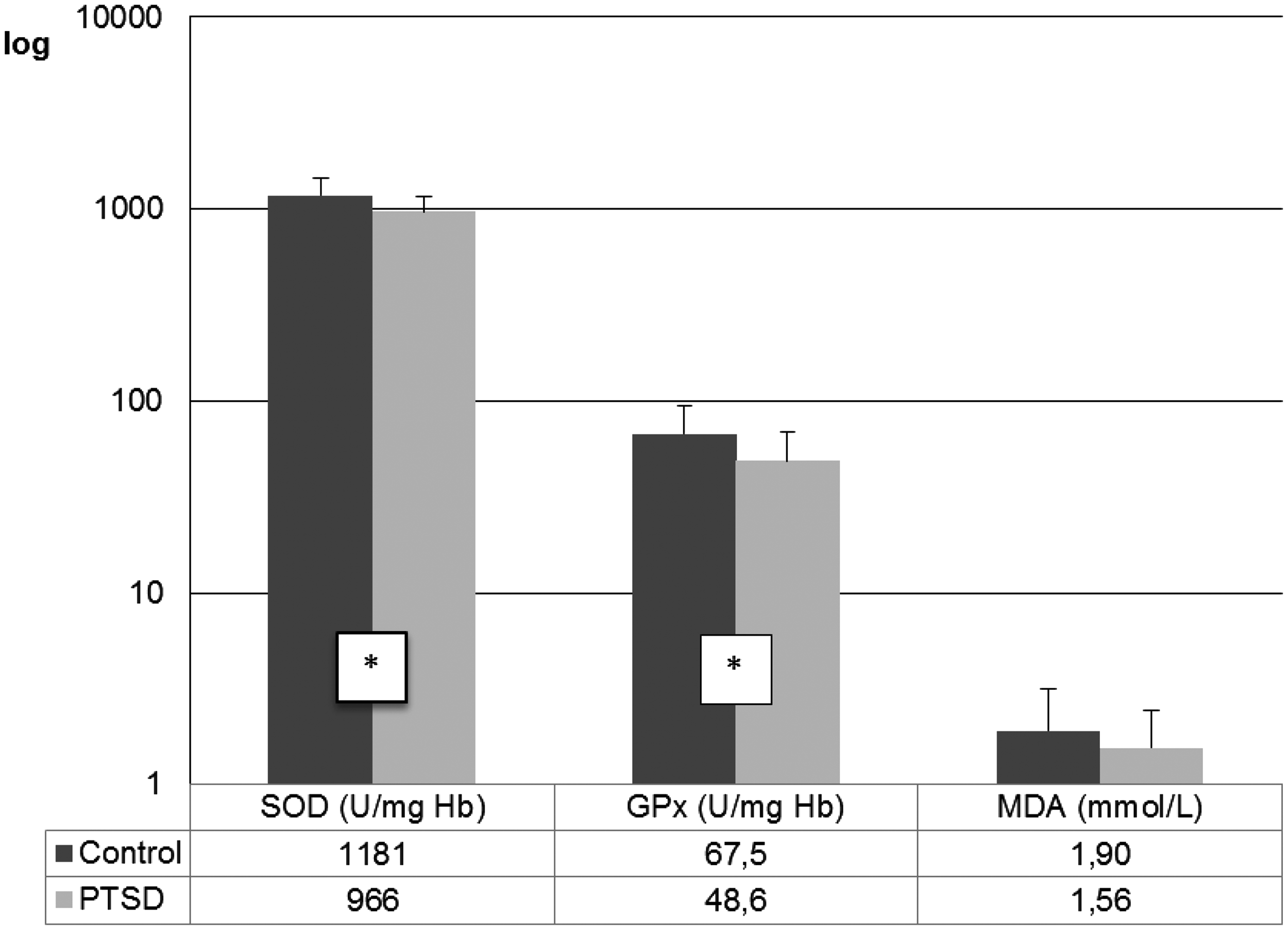
Discussion
Very few studies have been conducted on patients afflicted with a combat-related PTSD, and to the best of our knowledge, none of them analysed the relationship between parameters of oxidative stress and the severity of clinical symptoms.
Fifty patients who met the DSM-IV criteria for PTSD but did not have any previously documented somatic illnesses were recruited among outpatients in the University Psychiatric Hospital Vrapče, Zagreb, Croatia. Traumatic experiences during the Homeland War acted as a trigger for PTSD development for all of them. The control group was matched with 30 healthy volunteers who also served in the Homeland War, fortunately without serious consequences on their mental health. All participants were males of similar age, and the time spent in the war did not differ significantly between groups. Thus, two homogeneous groups were obtained according to gender, age, stressor and duration of illness.
The adverse effects and the intensity of the damage caused by free radicals strongly depend on the protective action of enzymatic (i.e. SOD, GPx and CAT) as well as non-enzymatic antioxidants (i.e. vitamins A, C and E, glutathione and selenium). In our study, the catalytic concentrations of eSOD and eGPx were within the reference range for both groups, but statistically significant differences for eSOD and eGPx activity between the two groups were found, with lower values obtained for the PTSD group. Similar results for SOD and GPx were obtained by Dadheech et al. 41 in blood of patients with schizophrenia, while Akyol et al. 42 reported decreased SOD with unchanged GPx catalytic concentrations.
Superoxide dismutase and GPx are related enzymes since the latter removes hydrogen peroxide, produced by SOD during superoxide radical dismutation. A positive correlation between SOD and GPx was demonstrated when tested in all participants as a whole (r = 0.56; P < 0.05). This positive correlation between SOD and GPx was again confirmed when tested within each study group separately (r = 0.50; P < 0.05 for PTSD group, and r = 0.43; P < 0.05 for control group).
These results could be explained through the data described by Zieker et al. 43 who referred different gene expression pattern in patients with PTSD, with SOD being downregulated. In addition, genes for some interleukins and thioredoxin, another protein involved in antioxidative defence, were also downregulated. In contrast to the above mentioned results, Tezcan et al. 44 did not find any difference in SOD and GPx activities between patients with PTSD and healthy controls. In literature, there are reports on lower SOD activity being related to aging processes and some other disorders.45,46 Attention is drawn to the negative correlation between eSOD activity and cholesterol concentration, specifically in the PTSD group. Although similar relationships were observed in human and animal studies with nutritional interventions with different types and amounts of fats,47,48 available data are ambiguous and the final explanation of related mechanisms is yet to be found.
Oxidative stress is demonstrated by increased concentrations of plasma peroxides. 49 Dadheech et al. 41 and Akyol et al. 42 have found increased oxidative stress in patients with schizophrenia as indicated by higher MDA concentrations. However, in our study, concentration of sMDA did not differ significantly between the two groups. Likewise, Tezcan et al. 44 did not find any increase in MDA concentration in the serum of PTSD patients.
The principal targets of lipid peroxidation are unsaturated fatty acids originating mainly from food. Therefore, some nutritional habits of the participants were compared, i.e. oil type and frequency of fish consumption. Generally, no significant differences were found between the two groups, with the exception of oil type consumption. Although the control group consumed more olive oil, no correlations with serum lipid parameters, antioxidant enzyme activities and sMDA concentrations were found. Additionally, age, time spent in the war, BMI and fish consumption did not correlate significantly with parameters of oxidative stress (P > 0.05 for all correlations) (data not shown).
Since anxiety and depression are the most common co-morbid symptoms in PTSD, self-report questionnaires were given to the affected participants. More than 50% of them reported severe and about 20% reported moderate symptoms of depression in BDI-II as well as in HADS. Separate scores for depression and anxiety from HADS revealed anxiety symptoms to be more pronounced. Most of the participants with PTSD were moderately ill when evaluated by the clinician-rated scale. Observed discrepancies between clinician-rated and self-administered severity of illness were reported in a number of studies. 50
It is worth noting that the education level was significantly higher in the control compared to the PTSD group, and higher education may indicate better cognitive abilities. Similarly, Horner and Hamner 51 in their review of performance on standard neuropsychological tests among individuals with PTSD suggested that certain individuals who are at risk for developing PTSD are also characterized by subtle cognitive deficits. Whether or not, and how, the level of education and cognitive abilities can be related to the likelihood that certain trauma will result in PTSD development, still remains to be investigated. Furthermore, because of their disease, the majority of PTSD patients lost their jobs, and/or were forced to retire. Lower incomes undoubtedly affected their social status as well as lifestyle and nutritional habits which resulted in lower nutritional quality, as shown by oil type consumption, although the higher BMI suggests that the quantity of food intake was not lowered. As reported by Braš et al., 52 PTSD symptoms were associated with a lower quality of life in the affected population. Since it had not been possible to analyse the quality of nutrition at the moment of the disease onset no conclusions on this topic can be attempted, but bearing in mind the impact of certain nutrients like vitamins, selenium, iron and others on (anti)oxidative status, these relationships certainly deserve further attention.
Finally, some drawbacks of this study must be pointed out. The main one is the fact that the PTSD patients were on antidepressant therapy with sertraline, which might cause, through the alleviation of depressive symptoms, a reduction of plasma markers of oxidative stress. 53 However, the patients could have risked further exacerbation of clinical symptoms had this antidepressant been removed from their therapy. In order to avoid the dose effect, all the patients were on the same dose (50 mg/day). In addition, changes in the regulation of the HPA axis could be followed. Gotovac et al. 54 have shown in their longitudinal study on a similar group of PTSD patients initially increased cortisol concentrations and natural killer cell cytotoxicity. However, cortisol concentrations decreased with the passing of time after a traumatic experience. It is worth noting that the changes in cortisol concentration reported in the above mentioned study were still within the physiological range, as were the reported significantly lower activities of sSOD and sGPx, although still within reference values. Furthermore, only a few nutritional questions were included. Further, more detailed studies about the influence of nutritional and environmental factors on parameters of oxidative stress should bring new insights into mechanisms of the disease onset and development.
In conclusion, the presence of increased oxidative stress indicators in patients suffering from PTSD could not be confirmed. However, our data suggest that lower catalytic concentrations of eSOD and eGPx in patients with PTSD may indicate a weakened response to oxidative stress due to impaired enzyme activity and/or decreased enzyme synthesis. Although no changes in MDA concentrations were found at the time of this study, presumably oxidative stress was present soon after the traumatic experience. Our results suggest a compensated balance between lipid peroxidation and enzymatic activity, though the lower level of oxidative stress due to therapy with antidepressants (sertraline) must also be taken into account. Finally, oxidative stress might be involved in the onset of PTSD, but is not essentially responsible for the continuance of the illness. Further studies are necessary to reveal the underlying mechanisms in PTSD development and possible treatments.
Footnotes
Acknowledgements
The authors thank all the volunteers who participated in the study. The authors also wish to thank Mr. Goran Kapustić and Mr. Franjo Ljubić for their skilful assistance, and Mrs. Laurette Rako and Ms. Dijana Gugić for manuscript editing and language advice.
Declaration of conflicting interests
None declared.
Funding
This research was funded by the grant from the Croatian Academy of Sciences and Arts.
Ethical approval
The study protocol was approved by the Ethics Committees of the University Psychiatric Hospital Vrapče and the School of Medicine, University of Zagreb, Croatia.
Guarantor
ID.
Contributorship
LBŠ, DK and ID researched literature and conceived the study. LBŠ, BBLj, MMP and MĆ performed laboratory analyses. LBŠ, DK, ID and NM were involved in protocol development, gaining ethical approval, patient recruitment and interpretation data analysis. JA and ID undertook statistical analysis. LBŠ wrote the first draft of the manuscript. PFG was involved in patient treatment and recruitment. All authors reviewed and edited the manuscript and approved the final version of the manuscript. LBŠ and DK have contributed equally to this work.
