Abstract
Background
The estimated glomerular filtration rate is a rather important measurement for patients under intensive care, since they often receive several drugs, and impaired renal function may result in misleading dosing. The estimated glomerular filtration is derived from mathematical models using serum creatinine, a measurement that suffers interference of some drugs, such as metamizole. This study intended to evaluate the impact on patient stratification for dose adjustment of two antimicrobials (meropenem and vancomycin) caused by metamizole interference in creatinine measurement by dry chemistry.
Methods
A cross-sectional study was conducted with a group of 108 hospitalized patients under metamizole prescriptions at fixed intervals. Serum creatinine concentrations were determined by enzymatic dry chemistry and Jaffé assays, and the estimated glomerular filtration rate was calculated through the CKD-EPI equation. Patients were stratified in groups according to their estimated glomerular filtration rate for drug dosing of vancomycin and meropenem.
Results
Creatinine values were significantly lower in measurements performed by the dry chemistry method in comparison to Jaffé assay (P < 0.0001) when patients are under metamizole treatment. A significant bias (–40.3%) was observed between those two methods, leading to a significant difference (P < 0.0001) in patient classification according to renal function using the CKD-EPI equation for dosing adjustment.
Conclusions
During the validity of metamizole treatment, the stratification for drug dosing by the estimated glomerular filtration rate is not reliable if the creatinine measurement is done through dry chemistry. Clinical and laboratory staff must be aware of these limitations and cooperate to optimize pharmacotherapy.
Introduction
The estimated glomerular filtration rate (eGFR) is an indirect measurement of nephrons’ functionality, determined by mathematical models that use serum creatinine measurement combined with other parameters such as age, sex, race and weight.1–3 This parameter is recommended for renal patients’ evaluation by the Brazilian Society of Nephrology, since serum creatinine is a poorly sensitive marker and patients with regular values may have a decrease in renal function due to other variables involved in glomerular filtration. 4 In fact, for young adults and children, serum creatinine alone failed to identify acute kidney injury (AKI) in 67.2% of patients with decreased urinary output. 5
The correct evaluation of renal function has an important impact for the intensive care physician, since there is an estimated incidence of 20 and 50% of AKI in patients in intensive care units (ICU). 6 Additionally, AKI is associated with an increase on the hospitalization period, hospital mortality and all-cause mortality after discharge, 7 , 8 also representing an increase of $5.4 billion to $24 billion in costs with hospitalization in the United States. 9
Within ICU, patients receive an average of 14 different drugs, 10 and renal function damage may lead to overdose or underdose of these drugs, so the precise evaluation of renal function is of absolute importance for drug dosing. 2 The eGFR is generally used in clinical practice for said purpose. 2 , 3
However, current laboratory tests are not yet able to uniformly reproduce a creatinine value, which leads to inaccuracy in eGFR. 11 Aiming to reduce the heterogeneity of results, the National Kidney Disease Education Program (NKDEP) recommended that all assays adopt a traceable calibration according to isotope dilution mass spectrometry (IDMS). 12 , 13
In addition to IDMS traceable calibration, manufacturers tried to develop analytical methodologies that would result in more accurate values in creatinine measurements. The primal method used was Jaffé reaction, which is based on the principle that picric acid in an alkaline environment forms a complex with creatinine, the colour of which can be determined by spectrophotometry. 14 Regardless of its simplicity and low cost, this methodology is very likely to have to several interferences, such as haemolysis in the sample, 15 , 16 hyperbilirubinaemia,15–17 elevated serum proteins, 15 , 17 hyperglycaemia 15 , 17 and lipeamia, 15 which may lead to falsely increased or diminished results.
Recently, the enzymatic methodology has been introduced in the market, which allowed a more specific measurement of creatinine through enzymes of different metabolic pathways. 11 It is noteworthy that dry chemistry system was utilized in this study, where creatinine is hydrolysed to creatine and then converted to sarcosine and urea by creatine amidino hydrolase. Sarcosine is oxidized to glycine, formaldehyde and hydrogen peroxide. Finally, a leuko dye is oxidized by hydrogen peroxide under the catalytic effect of peroxidase, forming a colourful product that can be quantified by reflectance spectrophotometry. 18 It is important to highlight that these enzymatic assays are able to mitigate the interference caused by haemolysis, bilirubin and lipaemia. 16
Despite the meaningful advances, technological limitations are still present in the clinical laboratory routine, especially due to exogenous interfering substances, several drugs that can interfere with the series of enzyme-catalysed chemical reactions like metamizole. 19 , 20 Due to the risk of agranulocytosis, metamizole use has been banned in several countries such as the United States, Japan, Australia and recently India, 21 while it remains the first choice of non-opioid analgesic in many countries of the European Union and Latin America, including Mexico and Brazil, 21 and being the most prescribed analgesic drug in Germany. 22
Perhaps for being an unused drug in the countries where diagnostic kit corporations are located, studies seeking to solve the interference of metamizole in enzymatic tests by dry chemistry were not found so far; therefore, studies are fundamental to help the laboratory and clinician to recognize this interference and to estimate its impact on decisions based on creatinine measurement.
Due to the widespread use of metamizole in some countries, as an example in Brazil where said drug is part of the national list of essential medicines of the public health system, 23 this study aimed to evaluate the impact on patient stratification for drugs dose adjustment (meropenem and vancomycin) caused by metamizole interference in creatinine measurement by dry chemistry.
Methods
Patients
Creatinine results from outpatients’ 629 serum samples (control group) and 108 serum samples from inpatients of Hospital Universitário Regional dos Campos Gerais (HURCG) who had a test order for creatinine in routine were analysed, without any further specimen.
Individuals under 18 years old and women who were known to be pregnant were excluded this study. The outpatients group included people with minor health problems and without prescription of any intravenous medication. The inpatients group included only people who had prescription for intravenous metamizole at fixed intervals (1 g every 6 h), those who were prescribed dobutamine or norepinephrine were excluded.
In addition, this study excluded all haemolysed serum samples according to the index provided by Vitros 5.1 FS Analyser (haemolysis greater than 50) and/or with hyperbilirubinaemia (total bilirubin greater than 5 mg/dL).
Serum samples
Blood samples were collected in SST tubes (BD Vacuteiner® – Becton Dickinson, Franklin Lakes, EUA) and serum was obtained after centrifugation at 3200 r/min for 15 min. All samples were obtained from routine exams of the intensive care unit in the morning, according to the standard procedure for venous or arterial puncture at the hospital.
Creatinine measurement
Serum creatinine was determined by two methods: enzymatic dry chemistry (routinely method) and Jaffé method, both using the VITROS 5.1 FS analyser (Ortho-Clinical Diagnostics). The enzymatic dry chemistry method applied slides produced by Ortho-Clinical Diagnostics, Inc. (Rochester, USA), following manufacturer’s recommendations from calibration and quality control (two daily levels of the Liquid Performance Verifier control). For Jaffé method were used reagents and standards with IDMS calibration from Labtest Diagnóstica S.A (Minas Gerais, Brazil), with internal quality control provided by Programa Nacional de Controle de Qualidade (PNCQ). The creatinine measures by the Jaffé method were performed within 1 h after the routine method. A single measure was performed for each method. The coefficient of variation (CV) for reproducibility was 3.1% for enzymatic dry chemistry and 6.2% for Jaffé method when creatinine measurements were performed.
eGFR and patient stratification
eGFR was calculated by the CKD-EPI equation using creatinine results obtained through the methods previously mentioned for inpatients who were receiving metamizol. The applied equation is available at the Brazilian Society of Nephrology website at: https://sbn.org.br/utilidades/calculadoras/. Age and sex data were obtained from the electronic medical records.
The patients were stratified into different groups according to the eGFR adopted for vancomycin and meropenem dosing, as described in manufacturer’s package insert 24 , 25 (Table 1).
Recommended dosage according to manufacturer's package insert for meropenem and vancomycin dose adjustment according to renal function.

eGFR: estimated glomerular filtration rate.
a1 dose unit: 500 mg to 2 g meropenem according to clinical indication.
Statistics
As the results did not present a normal distribution (Kolmogorov-Smirnov test), the comparative analysis was performed by non-parametric linear regression Passing-Bablok and by the Mann-Whitney U test. The Bland-Altman approach was used to evaluate the concordance degree between methodologies. The clinically acceptable limits were defined by the quality analytical specifications for a desirable bias, considering for creatinine a bias of ± 3.4%. 26 The Student’s t-test was used to evaluate the stratification of different groups according to the eGFR adopted for two antimicrobials dose adjustment (vancomycin and meropenem). The significance level was set P < 0.05. The data were analysed using MedCalc® software version 18.10.2 (MedCalc Software, Mariakerke, Belgium).
Ethics
This study was conducted in accordance with the Declaration of Helsinki and approved by the Research Ethics Committee of the State University of Ponta Grossa (UEPG), no. 2.896.948.
Results
The average age of the 108 inpatients was 56.5 years old (18 to 90 years old), with 58.3% (63) being men. Creatinine values were significantly lower (P < 0.0001) in measurements performed by the enzymatic method using dry chemistry when compared with the Jaffé colorimetric kinetic assay (Table 2).
Comparative analysis of creatinine results obtained by different methodologies of 108 patients.

aMann-Whitney U test, P <0.0001.
In the concordance analysis between the two methods, the Passing-Bablok regression obtained a slope of 0.627, indicating a negative proportional error of 37.2% with a consistent bias of 0.0562, showing a significant linearity deviation (P = 0.03) (Figure 1(a)). The Bland-Altman graph shows a significant bias for the creatinine results between the two methods, with an average difference out of the acceptable clinical limit of ± 3.4% 26 (Figure 1(b)).
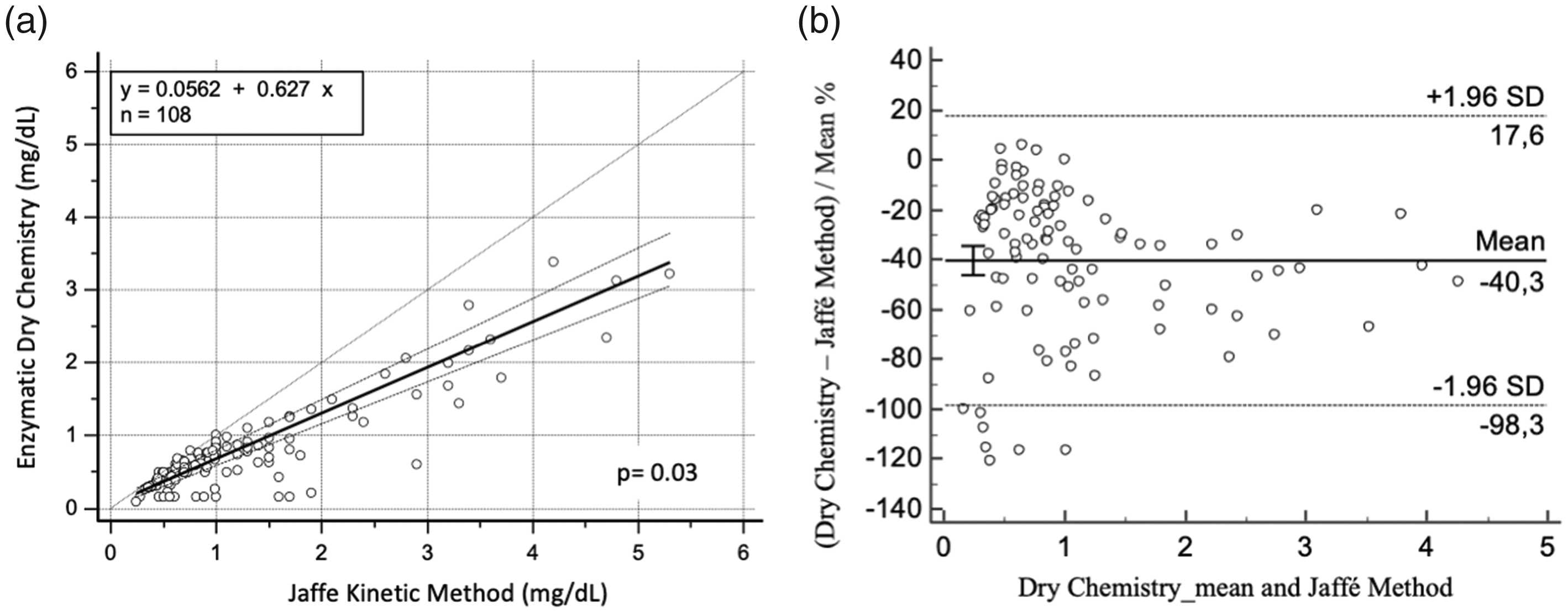
Concordance analysis between enzymatic dry chemistry and Jaffé method for creatinine measurement in patients with intravenous metamizole therapy: (a) Passing-Bablok regression line shows the correlation between the two methods (Jaffé kinetic method and Enzymatic dry chemistry) with significant linearity deviation, P = 0.03 and (b) Bland-Altman plot shows the significant dispersion of differences, indicating a poor concordance between the two measurement procedures, with a mean difference of –40.2% and a wide 95% CI (+17.6 to –98.3) in creatinine values measured by the enzymatic dry chemistry in comparison to the kinetic Jaffé procedure.
A parallel investigation with outpatients was conducted to validated results obtained. The average age of the 629 outpatients was 54 years old (18 to 98 years old), with 49.4% (311) being men. The concordance analysis applied is demonstrated in Figure 2.
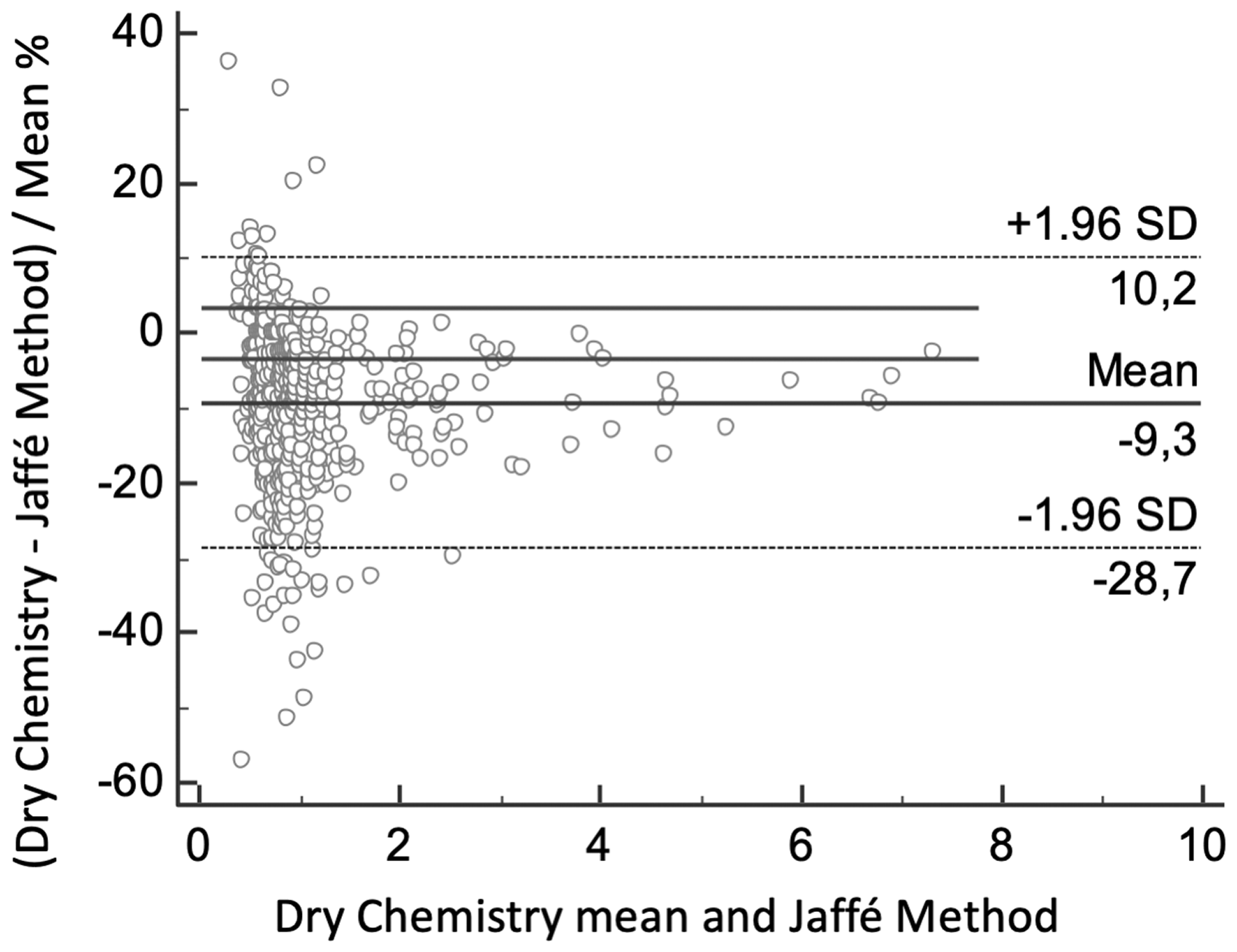
Concordance analysis between enzymatic dry chemistry and Jaffé method for creatinine measurement in outpatients with minor health problems and without prescription of any intravenous medication. Bland-Altman plot shows less significant bias for the creatinine results between the two methods, with a mean difference of –9.3% (+10.2 to –28.7) in creatinine values measured by the enzymatic dry chemistry in comparison to the kinetic Jaffé procedure.
Patients were stratified into different groups according to kidney function by the eGFR according to the manufacturer's package insert of vancomycin and meropenem, with the classification being displayed in Tables 3 and 4.
Classification used to meropenem dosing according to the eGFR calculated for the 108 patients with the creatinine results obtained by different methodologies.

eGFR: estimated glomerular filtration rate.
aStudent’s t-test, P < 0.0001.
Classification used to vancomycin dosing according to the eGFR calculated for the 108 patients with the creatinine results obtained by different methodologies.
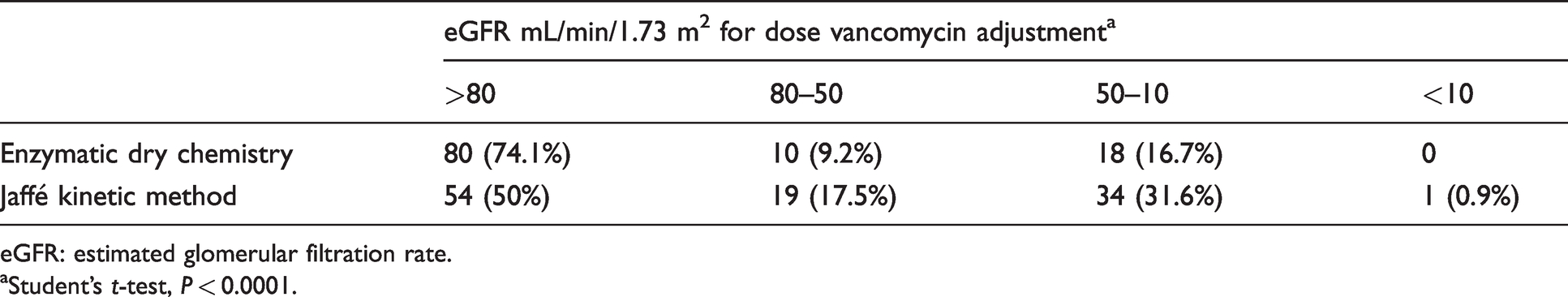
eGFR: estimated glomerular filtration rate.
aStudent’s t-test, P < 0.0001.
The negative bias from enzymatic dry chemistry leads to a significant difference (P < 0.0001) in the classification of individuals according to the renal function for the two antimicrobials.
Discussion
Serum creatinine is a fundamental component of equations to determine eGFR, and accurate measures are crucial for obtaining a reliable estimate rate of glomerular function, making it possible to diagnose and monitor renal disease, as well drug dosing. 1 The results obtained in this study suggests that patient stratification for drug dosage based on the eGFR should be performed with caution and taking into consideration the prescribed drugs when using enzymatic methodology by dry chemistry, since it can overestimate eGFR in patients receiving metamizole.
When comparing the results obtained by the two different methods, significantly lower values were observed in the measurements performed by the dry chemistry enzymatic assay, with a significative bias exceeding the acceptable clinical limit. These results suggest a negative interference by metamizole, corroborating other studies performed in vitro that have demonstrated that samples containing metamizole or its metabolites present with lower values for creatinine when determined enzymatically. 19 , 20
There are reports of the metamizole interference in the determination of other analysis by the same principle, being purposed an interaction in the peroxidase reaction. 19 Gascon-Roche and collaborators described a significant positive interference in the determination of free triiodothyronine (T3), cortisol, estradiol and a negative interference in the carcinoembryonic antigen, human chorionic gonadotropin and α-fetoprotein, when determined by an immunoenzymatic assay based in the luminescence that uses peroxidase as a marker. 27
In addition to metamizole, other substances can also affect creatinine values. 28 Since peroxide is necessary for enzymatic reactions, antioxidant agents can lead to a negative bias. 29 Catecholamine interference is also known, leading to significant decrease in some platforms, mainly with the use of dobutamine and norepinephrine. 30 , 31 In order to prevent this interference from impacting in the results, all patients who had catecholamine prescription during any time of hospitalization were excluded.
Due to negative bias in creatinine measurement by dry chemistry, a reduction in the calculated value to eGFR was induced in relation to calculation using creatinine measurement by the Jaffé methodology. The dependent methodology variation on the results led to a significant difference in the classification of individuals according to renal function for the two antimicrobials analysed, according to the recommended classification on the manufacturer's package insert, 24 , 25 so that individuals would be relocated to a group that would receive a higher dose of medications. This difference may prove to be clinically relevant, due to mistaken patient stratification for drug dosing, especially for those with AKI who have lower creatinine values. The eGFR is described by a hyperbolic function in relation to serum creatinine concentration, suffering less interference at higher concentrations, 32 as we showed in Bland-Altman plot (Figure 1) since the differences in the creatinine results between the two methods were larger with mean values lower than 1.3 mg/dL.
One may think that, due to the falsely elevated eGFR, a dose adjustment for a higher concentration of vancomycin could result in an aggravation of renal function and aggravation of an AKI, since the drug itself is nephrotoxic. 25 , 33 However, this study did not evaluate the clinical outcome, and studies are needed in this context to rule out the null hypothesis, that is, that the variation in stratification would not impact the outcome of patients.
Given the problem that, especially in ICUs, patients receive many medications and are under clinical conditions that can affect creatinine measurement by different methodologies, possible alternatives are found in the analysis of eGFR by cystatin C. As it is a biomarker produced and secreted at a constant rate and almost completely reabsorbed, its serum concentration depends almost exclusively of the glomerular function. 34 In addition, unlike creatinine, cystatin C results are not affected by diet, nutritional status, muscle mass, age and gender;34–36 however, it is known that high doses of corticosteroids 37 and thyroid disfunction 38 interfere in the results.
Despite the advantages of cystatin C when compared with creatinine, studies have shown that eGFR based just in this marker alone is no more accurate than the estimates made using creatinine. 39 In this sense, Inker et al. suggested a solution to improve the evaluation of renal function, combining the serum measurements of both. The study showed that the combination creatinine–cystatin C in the same equation is more accurate than any isolated marker to estimate GFR. 39
Another possibility to settle this problem, considering the high cost of cystatin C and its unavailability in laboratory practice, 40 would be through the development of protocols that indicate the possible interferences in the laboratory routine and directed the creatinine measurement to another methodology, such as of Jaffé. This kind of protocol was validated in some laboratories, with patients with a test order for creatinine under vasoactive drug treatment. 31 This would be a simple and cost-effective solution that would allow more reliable results. Other approaches may include the use of analgesics that do not cause bias in biochemical analyses, as well the serum measurement of drugs to be adjusted with therapeutic drug monitoring. 33
As far as we know, this work is the first one to show metamizole’s interference in the stratification for drug dosing in hospitalized patients using dry chemistry methodology and, at the same time, reproducing the patient's condition found by the clinician daily. Although it is not a controlled study, the pretest probability of the hypothesis was high, due to the homogeneity of results in the studies that analyse the correlation between metamizole and the creatinine measurement by dry chemistry, being those evidence corroborated by this study. The selection of the studied group excluded the main interfering variables that could cause confusion in the results, such as haemolysis, hyperbilirubinaemia and the use of catecholamines, and also used two methodologies with traceable calibration by IDMS to evaluate the effect of metamizole on the creatinine measurement.
However, this work also has limitations. There is a significant bias between the methodologies (Figure 2) despite metamizole, although it is a significant lesser bias when compared with metamizole, but further investigations should aim to calibrate the true metamizole bias. Although the main interferences described in the literature have been excluded, other substances may have impact in the results, since hospitalized patients have comorbidities and use a variety of medications. Another significant limitation is that it was a single centre observational study, so institutional and individual characteristics such as ICU routines, like time gap between blood specimen and drug administration, could result in confounding factors. Finally, the dose-dependent relation and drug administration by other routes, like oral metamizole, were not evaluated and also could interfere in the results.
Conclusion
The conclusion found as the end of the study was approached is that patient stratification for drug dosing by the eGFR is not reliable when patients are under intravenous metamizole treatment and creatinine is measured by dry chemistry enzymatic assay. Clinical and laboratory staff should cooperate for the development of protocols that mitigates this interference, as the inclusion of information about the drugs that patients receive in their test ordering, so patients could benefit from an optimal pharmacotherapy.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors would like to express their gratitude to Universidade Estadual de Ponta Grossa and the Hospital Universitário Regional dos Campos Gerais for the support.
Ethical approval
This study was conducted in accordance with the Declaration of Helsinki and approved by the Research Ethics Committee of the State University of Ponta Grossa (UEPG), no. 2.896.948.
Guarantor
MFM.
Contributorship
Conceptualization: MFM; Acquisition of data: LB, GR and LMD; Analysis: LB, GR, MFM; Investigation: LB, GR, CLB, DCK, MFM; Methodology: GR and MFM; Project administration: LB and MFM; Writing-original draft: LB and GR; Supervision: LMD, CLB, DCK, MFM; Writing-review and editing: LB, GR and MFM.
