Abstract
Background
Plasma-free metanephrines (PFM) or urinary fractionated metanephrines (UFM) are the preferred biochemical tests for the diagnosis of phaeochromocytoma and paraganglioma (PPGL). Borderline increased results should be followed up to either exclude or confirm diagnosis.
Methods
We extracted all PFM and UFM results reported by our laboratory over a six-month period from the laboratory information system. We categorized patients with borderline increased results according to whether follow-up testing had been performed as suggested in the initial laboratory report. Questionnaires were then sent to all requesting doctors and medical notes reviewed where available.
Results
Two hundred and four patients with borderline increased PFM or UFM were identified. Sixty-five (38.5%) of 169 patients with borderline increased PFM had a repeat test out of which 36 were normal and 29 did not normalize. Of 35 patients with borderline increased UFM, 17 (48.6%) had subsequent PFM measurement, out of which 15 were normal. Questionnaires were returned to 106 (52%) patients. Of these, the most frequent indication for testing was hypertension (
Conclusion
Only 38% of patients with borderline increased PFM had a repeat PFM measurement. This was not significantly higher when compared with the 28% in a previous audit that we reported in 2010 (
Background
Phaeochromocytomas and paragangliomas (PPGLs) are catecholamine-producing tumours arising from the chromaffin cells of the adrenal medulla or extra-adrenal paraganglia, which usually manifest with moderate or severe hypertension. The recommended initial biochemical testing for PPGL is measurement of either plasma-free or urinary fractionated metanephrines which, if increased, demonstrate excess production. 1 Liquid chromatography with mass spectrometry or electrochemical detection is the suggested method for their measurement. 1 While very high concentrations of metanephrines (>3 times the upper reference limit for normetanephrine and > 4 times the upper reference limit for metanephrine) have a high predictive value for the diagnosis of PPGL, borderline increased values can also be the result of inappropriate sampling conditions, medications and physiological stress and should be clarified with repeat testing after exclusion of these factors. 2 It has been recommended that all positive results should be followed up, with the caveat that whether additional comprehensive testing procedures or a wait-and-retest approach is pursued remains a matter of clinical judgment based on the pretest probability of a tumour and the extent and pattern of increases in test results. 1 While at the clinical interface the biochemical results can be considered in the context of pretest probability, the reporting laboratory often does not have adequate information regarding the patient’s presentation and the pretest probability in order to report increased values in terms of the likelihood of the presence of PPGL in individual patients. Therefore, we have taken the pathway of reporting increased metanephrine values as ‘borderline increased – phaeochromocytoma not excluded’ or ‘clearly increased – phaeochromocytoma likely’. 3
In 2010, we established that only 28% of borderline increased PFM reported by our laboratory were followed up with a repeat test. 4 Based on this, we modified the interpretative comment for borderline increased plasma metanephrines that was reported to the requesting clinicians to encourage repeat testing (Table 1).
Interpretative comment for borderline elevated results.
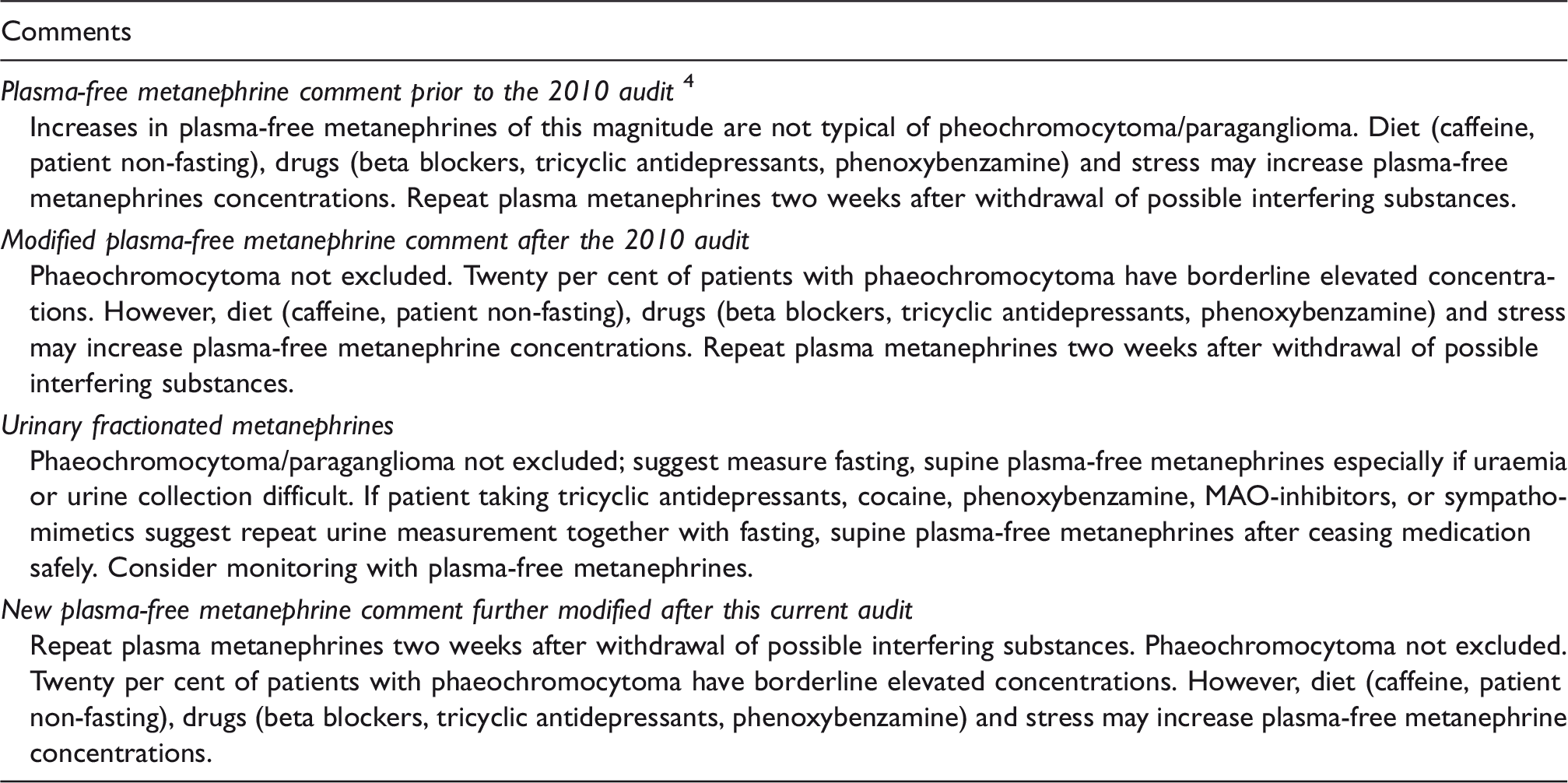
Note: Last box contains the newest comment for PFM, modified following this current audit.
This audit aimed to assess the follow-up of patients with borderline increased PFM following the change in the interpretative comment. In addition, the audit also looked at the follow-up and management of patients with increased UFM and clearly increased PFM to determine whether the interpretative comments are adequate in assisting the clinician to take appropriate steps in order to exclude or confirm phaeochromocytoma.
Methodology
Measurement of PFM and UFM
Both PFM and UFM were measured by isotope dilution-mass spectrometry following sample preparation using solid phase extraction cartridges and then liquid chromatography. 5 Plasma samples were preferably collected after fasting and 30 min of supine rest. Plasma-free normetanephrine and metanephrine concentrations were reported. Acidified 24-h urine collections were required for UFM. The samples were subject to acid hydrolysis prior to extraction such that the results were reported as 24-h excretions of total (conjugated and free) normetanephrine, metanephrine and 3-methoxy tyramine.
Data collection
All results for PFM and UFM performed at PathWest Laboratory, Fiona Stanley Hospital, between June 2015 and December 2015 were extracted from the laboratory information system in January 2017. Borderline PFM and UFM results were defined using values as shown in supplementary table.2,6 The laboratory results were accompanied by interpretative comments (Table 1). Clearly increased results are those above the borderline range and are considered diagnostic of phaeochromocytoma if medication-induced false increases have been excluded. Patients with borderline increased results were examined for a follow-up test in the period between June 2015 and December 2016. Questionnaires (see supplementary material) were then sent to the requesting doctors or doctors in charge, and medical notes reviewed where available. The proportion of cases with repeat testing in 2016 was compared with that in the previous audit in 2010 using the χ 2 test (MedCalc Statistical Software version 16.2.1; https://www.medcalc.org; 2016).
The audit was registered as a quality activity with the Safety, Quality & Risk Committee, South Metropolitan Health Service, Western Australia (Quality Activity No: 13553).
Results
Borderline PFM and UFM results
A total of 1635 results were reported for PFM, of which 218 (13.3%) were categorized as borderline increased. Of these, 169 were first-time results and were included in further analysis. Sixty-nine (40.8%) requests were from general practice, 57 (33.7%) from a hospital/specialist outpatient setting and the remaining 43 (25.5%) were hospital inpatients. The repeat testing rates were similar in the three settings. Sixty-five (38.5%) patients had a repeat test, of which 36 were normal and 29 remained increased (Figure 1).
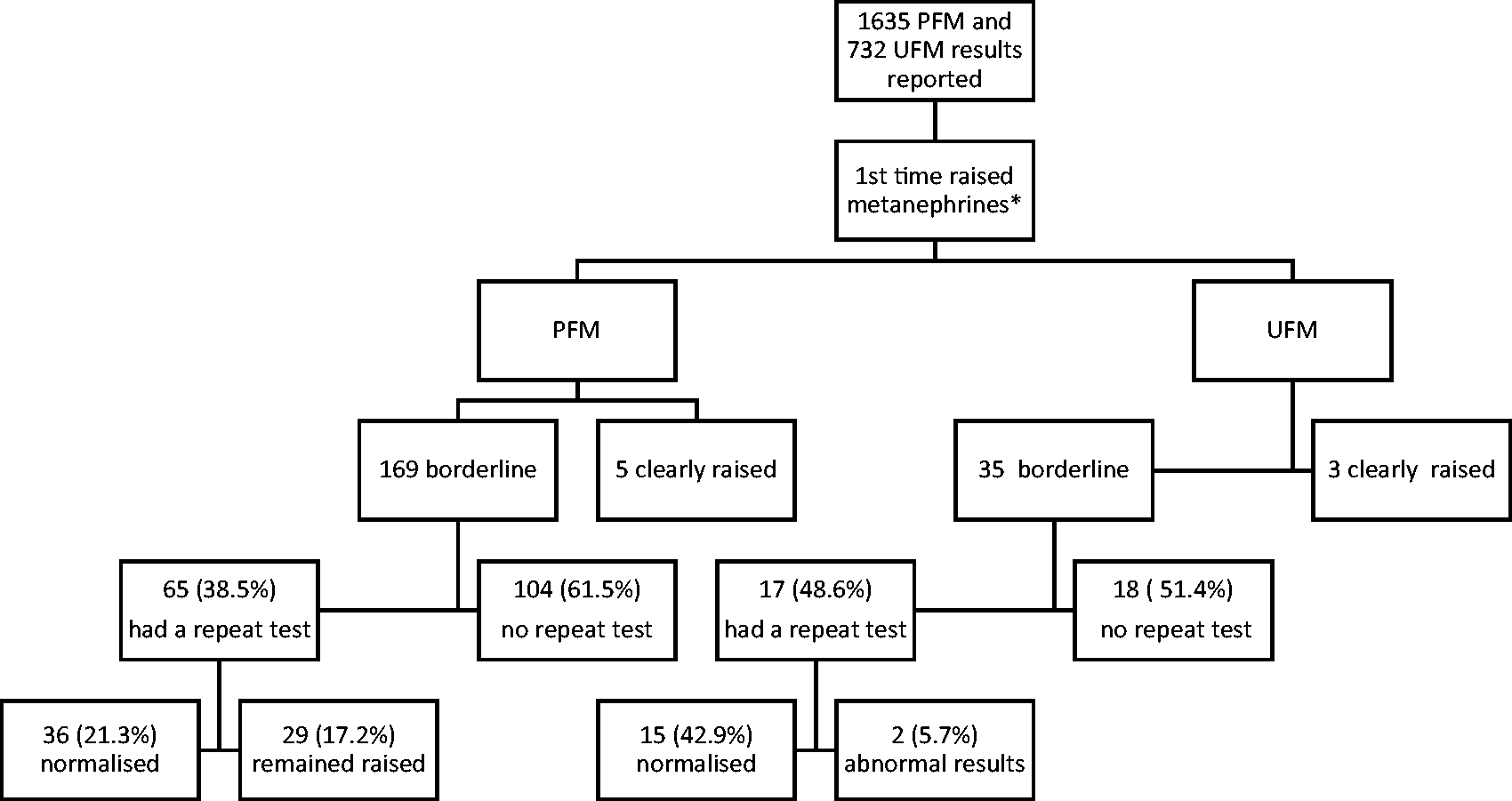
Follow-up of patients with raised metanephrines.
Of 732 UFM results, 52 (7.1%) were borderline increased, 35 for the first time. Of the latter, 30 (85.7%) requests came from general practitioners, two (5.7%) from private specialist clinics and three (8.6%) were hospital inpatients. Seventeen (48.6%) patients had a subsequent PFM or UFM measurement of which 15 were normal (Figure 1). The other two patients had borderline increased PFM on follow-up, with subsequently normal clonidine suppression tests.
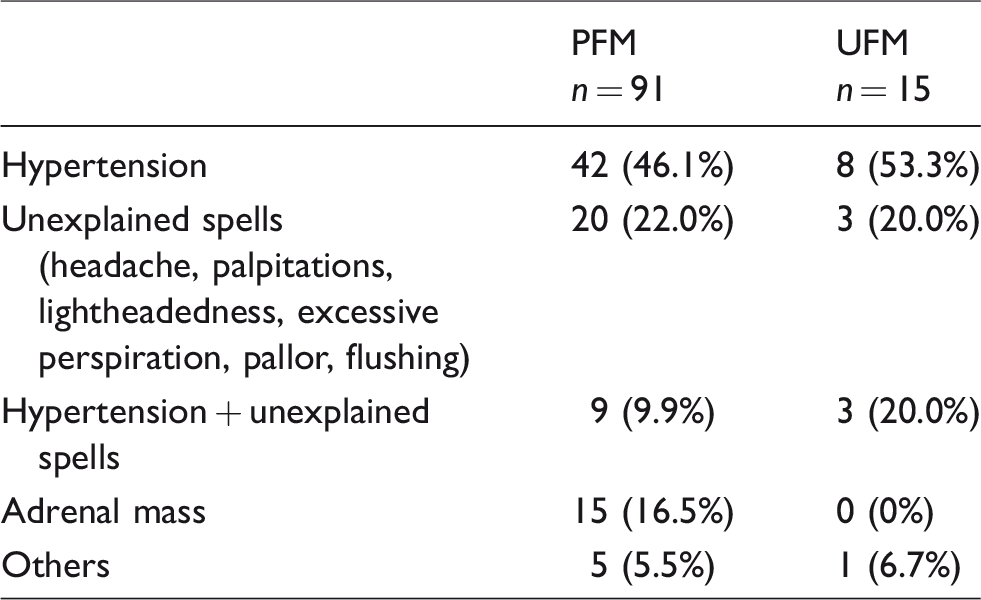
Ninety-one of 169 (53.8%) questionnaires were completed for PFM and 15 of 35 (42.9%) for UFM giving a total response rate of 52%. The most common indication for requesting metanephrines measurement was hypertension, followed by investigation of an incidental adrenal mass (Table 2).
Indications for requesting metanephrines.
Borderline PFM or UFM results in patients with test not repeated
Of 91 patients with borderline PFM results for whom we received a completed questionnaire, 54 (59.3%) patients did not have a repeat PFM. The reasons for not repeating testing are shown in Table 3. In one case for which we could not find follow-up request, the requesting doctor stated that repeat testing had been done, which we confirmed was done in another laboratory. For UFM, out of 15 patients with borderline results for whom we received a completed questionnaire, three patients did not have a repeat PFM as recommended: of these three, one patient’s symptoms spontaneously resolved and two were referred to specialists for further diagnostic work-up. One of these two patients was subsequently diagnosed with primary aldosteronism, and the other with essential hypertension in the context of sleep apnoea.
Reasons for not repeating PFM.

Borderline PFM results in patients with adrenal mass
Borderline increase in PFM or UFM in the presence of an adrenal mass constitutes a high probability of PPGL. Of 15 patients with an incidental adrenal mass, 11 had repeat PFM measurements, 6 of which were normal. Diagnosis of non-functioning adrenal adenoma was made in two patients, two had a diagnosis of lipid-rich adenoma, one patient had an adrenalectomy where histopathological diagnosis revealed a benign schwannoma, while the diagnosis of one remained undetermined at the time of writing. Of the five patients who had a borderline increased result on repeat measurement, two had adrenalectomy with histopathologically proven phaeochromocytoma, one had surgery for moderately to poorly differentiated adenocarcinoma, one was determined to have a non-functioning adrenal adenoma and the other patient was lost to follow-up.
Of the four patients with initial borderline PFM and an adrenal mass who did not have repeat PFM, one patient was followed up with UFM which returned a normal result and a final diagnosis of non-functioning adrenal adenoma was made. Three patients had no further laboratory testing.
Clearly increased results
Three patients with clearly increased first-time UFM underwent subsequent testing with PFM, which confirmed the diagnosis of phaeochromocytoma and adrenalectomy was performed in all three.
Five patients had clearly increased first-time PFM; two of these underwent adrenalectomy, the other three had their hypertension medically managed as a diagnosis of phaeochromocytoma was not confirmed.
Comparison with previous audit
Thirty-eight (38.5) per cent of borderline increased PFM were followed up with a repeat test following the introduction of new interpretative comments compared with 27.6% in the 2010 audit (difference 10.8% [95% confidence interval (CI) −2.8–23.2%];
Discussion
This retrospective audit confirms that a substantial proportion of patients with borderline increased PFM results are not followed up with a repeat test. Other groups have also shown a low follow-up rate of abnormal metanephrine and normetanephrine results, with a figure of 42.7%. 7 However, there has been a modest but statistically non-significant increase in rates of repeat testing since our previous audit which led to changes to our laboratory’s reporting of these results. There may be valid reasons in some cases for not repeating the test. The responses to the questionnaire helped ascertain why repeat testing was not performed in some cases. However, for nearly 30% of cases, we do not know why repeats were not requested, and they could be possibly harbouring undiagnosed PPGLs. It is possible that some repeat testing was done outside of our laboratory network; however, as our laboratory is the sole public provider of these tests in a geographically isolated jurisdiction, this is unlikely to account for a substantial number of cases. Most private laboratories (with one exception where metanephrines are referred interstate) also refer their work to our laboratory. Twelve out of the 15 patients with an adrenal adenoma and borderline PFM were followed up with further testing, and two cases of phaeochromocytoma were identified. Three out of the 15 were not followed raising the possibility of a missed diagnosis in this high-risk group.
We could hypothesize that a potential contributing factor to the complacency of clinicians to the follow-up of borderline positive results could be related to high rates of false-positive results. High rates of false-positive results can be reduced by both adherence to appropriate preanalytical precautions and optimal cut-offs.6,8 As indicated in the supplemental data, we have been using age-specific cut-offs for plasma NMN, which should help reduce false-positive results. 6 An optimal cut-off for plasma metanephrine was described to be higher than that indicated by 97.5 percentiles (446 vs. 330 pmol/L). 6 Further optimization of cut-offs may be possible and may result in reducing the number of false-positives. Non-adherence to appropriate sampling condition (supine position) results in nearly six times more false-positive results especially if the upper reference interval was determined using the strict collection conditions. 8 Although we recommend that fasting samples are collected after 30 min of supine rest, whether this was practised cannot be determined for all samples collected. We may in future enforce strict sample collection after an overnight fast and a 30-min rest in supine position. Lenders et al. recommend that if initial samples were collected in a seated position, the raised results should be followed up using samples collected in supine condition or by using alternative tests. 9 False increases due to medications may be another factor, accounting for about 20% of false increases in one series. 10 A number of medications can cause falsely elevated PFM, and, in such cases, follow-up testing should be done after their exclusion as also recommended in our interpretative comment. 11
Case series have demonstrated that patients with histologically proven phaeochromocytoma may have only borderline increased metanephrines or normetanephrines.7,12 Garrahy et al. reported that two of 12 subjects with phaeochromocytoma had borderline elevated UFM. 7 Similarly, Lee et al. reported that 3 out of 10 patients with adrenal incidentaloma and borderline metanephrines had phaeochromocytoma. 12 In our series, two of 15 patients followed up with an incidentaloma and a borderline increased metanephrine result had a proven phaeochromocytoma. Clearly, patients with a high pretest probability due to the presence of family history, or an incidentally discovered adenoma should be treated with a higher degree of suspicion when the metanephrine increases are borderline. 1 These reports support close follow-up of patients with borderline increased results. Although phaeochromocytoma is uncommon, it is nonetheless a potentially dangerous and curable cause of hypertension. Although hypertension is the most common indication for testing, metanephrines are also requested for the investigation of other potential presenting features of PPGL such as episodic headache, palpitations, sweating and anxiety, as well as those with adrenal incidentaloma and positive family history of PPGL. In those with adrenal incidentaloma, the prevalence of PPGL is reported to be between 1.5 and 14%, with other important differential diagnoses caused by functional tumours including primary aldosteronism and Cushing’s syndrome.13,14
Often the diagnosis is straightforward when the results of PFM and UFM are greater than three or four times the upper reference limit. Of such cases in our audit, one patient who had a final diagnosis of phaeochromocytoma and another who had paracaval paraganglioma, had metanephrine concentrations more than 70 and 5 times the upper reference limit, respectively, while normetanephrine was more than 10 times for both. In the remaining three not diagnosed with PPGL but with concentrations above the borderline range, plasma normetanephrine was less than four times their age-specific cut-offs while plasma metanephrine was borderline increased in two and normal in one patient. In one patient, an interfering medication was identified, while in another an equivocal radiological finding and a negative clonidine suppression test effectively ruled out the diagnosis. All three patients with clearly increased UFM also had a subsequent clearly increased PFM with a final diagnosis of phaeochromocytoma.
Of note, the number of requests for PFM has more than tripled from our last audit in 2010, 4 reflecting the increased awareness among clinicians of the indications for testing. There were also more requests for PFM compared with UFM, likely due to the known superior sensitivity of PFM over UFM, as well as convenience.
Limitations of our study include a response rate of 52% for returned questionnaires, and the lack of long-term outcomes data for patients with borderline increases in PFM and UFM who did not receive repeat testing. Some repeat testing may have been done in other laboratories, but we expect that this would have been communicated to us in the returned questionnaires, as appears to have been true in only one case. Our geographical isolation is in fact a strength in undertaking this type of audit. We were unable to ascertain the reason for not repeating testing in 15 cases (Table 3). This again is a limitation of the retrospective laboratory based audit.
In conclusion, there was no significant improvement in the follow-up testing of abnormal metanephrine and normetanephrine results; the repeat test rate remained suboptimal. The interpretative comment attached to borderline PFM results has been further modified as a result of this audit (Table 1, last row). Strict enforcement of patient preparation and sample collection procedures, further studies to optimize cut-offs to reduce false-positives, changes to the interpretative comment accompanying borderline increased PFM and UFM results as mentioned above as well as education of clinicians will be considered, in order to increase the repeat testing rate. Case review discussions and continuing education activities among clinicians heighten awareness amongst clinicians of the challenges in diagnosis and management of PPGL. 15
Supplemental Material
Supplemental material for The challenge of improving the diagnostic yield from metanephrine testing in suspected phaeochromocytoma and paraganglioma
Supplemental material for The challenge of improving the diagnostic yield from metanephrine testing in suspected phaeochromocytoma and paraganglioma by Intan Samsudin, Michael M Page, Kirsten Hoad, Paul Chubb, Melissa Gillett, Paul Glendenning and Samuel Vasikaran in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
Assistance by the PathWest Fiona Stanley Hospital Network staff is acknowledged.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Approved by the Safety, Quality & Risk Committee, South Metropolitan Health Service, Western Australia (Quality Activity No: 13553).
Guarantor
SV.
Contributorship
All authors contributed to the planning and reporting of the audit. IS collected the data and wrote the initial manuscript. SV designed the audit and contributed to writing of the manuscript. All authors reviewed and approved the final version of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
