Abstract
Background
Communicating abnormal results to requesting clinicians is an essential part of clinical authorisation. Guidance from the Royal College of Pathologists on communication of critical/unexpected results is issued as ‘advice to pathologists’. The 2017 guidelines advise rapid communication of serum potassium results ≤2.5 mmol/L and ≥6.5 mmol/L. Little is known about what happens after the results have been communicated. We wished to establish answers to the following questions: Are phoned results acted on? If so, when? What is the outcome of any action taken?
Methods
A prospective study of primary care potassium results authorised out of hours was undertaken. Potassium requests from primary care were retrieved from the laboratory information management system. The potassium result was recorded, along with other data. Data were analysed for potassium results that were validated out of hours (18:00 h–08:00 h).
Results
Over six months, 220 potassium results <3.1 mmol/L and >5.9 mmol/L from primary care were validated out of hours. A subset of these (27) were phoned to the general practice out of hours ‘hub’, and 16 patients referred to hospital out of hours, on account of the potassium results. The remaining potassium results phoned out of hours were acted on subsequently.
Conclusions
Critical potassium results were phoned urgently and are acted on, although not always out of hours. For potassium results phoned out of hours, the most frequent action was to refer to hospital out of hours. Different actions occurred for similar potassium results, reflecting the fact that actions taken and their urgency depend on the patient, the clinician and the practice policy for handling results.
Introduction
A core function of clinical authorisation is to alert requesting clinicians to abnormal results that may warrant urgent intervention. Credit for the original observation highlighting the importance of such results is widely given to Lundberg, in a somewhat unpromisingly titled 1972 paper: ‘When to panic over abnormal values’.1 In the United Kingdom, guidance on reporting of laboratory results requiring urgent clinical action has been available from the Royal College of Pathologists (RCPath) since 2004. Current RCPath guidance is published as ‘advice to pathologists’,2 acknowledging the absence of underpinning evidence. In the current study, we focused on what happens after a result is phoned, and addressed three questions: (1) Are phoned results acted on? (2) When are they acted on? (3) What is the outcome of any action taken?
Methods
In Tayside, Scotland, out-of-hours (OOH) general practice (GP) services are provided through a single ‘hub’, which operates between 18:00 h and 08:00 h on weekdays, and throughout the weekend. It is staffed at all times by a general practitioner, as well as nursing and administrative staff. Action limits were in accordance with the 2010 RCPath guidance3 for the duration of the study, from 1 December 2015 to 31 May 2016; potassium results above 6.5 mmol/L and below 2.5 mmol/L were phoned. We confined ourselves to results phoned to the OOH hub between 18:00 h and 08:00 h.
We defined an action as any of the following: venepuncture; referral to an acute receiving facility for further assessment; treatment (Telephoning the patient was not considered an action. Although useful, this is done reflexly by the hub on receipt of a phoned result.). We categorised actions according to whether they were taken outside normal working hours or at the next available opportunity within normal working hours; these vary slightly from one practice to another, but were possible to establish in each individual case (The timing of actions was established from the laboratory information management system and/or electronic patient record.). Outcomes were also categorised. For venepuncture, the follow-up potassium result was categorised according to whether it too breached the action limit. For hospital referral, categorised was according to whether or not the patient was admitted.
Results
During the study, 128,618 potassium requests were received from GP sources. Of these, 2112 samples were unsuitable for analysis, and 125,841 had a potassium result of 3.1–5.9 mmol/L. Of the remaining 665 samples, 272 had a result <3.1 mmol/L and 393 had a result of >5.9 mmol/L. Of these, 220 were validated OOH (84 with a result <3.1 mmol/L and 136 with a result >5.9 mmol/L). Of the 220 ‘abnormal’ potassium results which were validated OOH, 89 were autovalidated. Results were autovalidated if the result was between 3.0 and 6.0 mmol/L inclusive, or if there was a ≤ 0.8 mmol/L change in result in the previous seven days, unless the result breached the action limits. Three of the autovalidated potassium results were phoned to the OOH hub on account of other results. The remaining potassium results were viewed by the duty biochemist and one of several actions was taken: 95 results were manually validated only; five samples were manually validated with a comment added; 27 results were phoned to the OOH hub primarily on account of the potassium result; seven (including the three ‘autovalidated’ potassium results) were phoned to the OOH primarily on account of other results. This gave a total of 34 potassium results phoned to the OOH hub.
Of the 27 results phoned to OOH primarily on account of the potassium result, 22 breached the RCPath action limits. Figure 1 summarises whether or not action was taken, as well as the urgency, nature and outcome of the action. One patient had died by the time the result was phoned, and one had already been referred to hospital for other reasons; both of these were categorised as ‘not acted on’. In the case of results which were acted on, all patients except one underwent repeat venepuncture. For urgent (OOH) actions, this was performed in secondary care. For non-urgent actions, it was performed in primary care. Of 16 patients bled urgently, repeat potassium breached the action limit in six; these were admitted for potassium replacement. Three were admitted for other reasons (acute kidney injury, cirrhosis and sepsis, respectively).
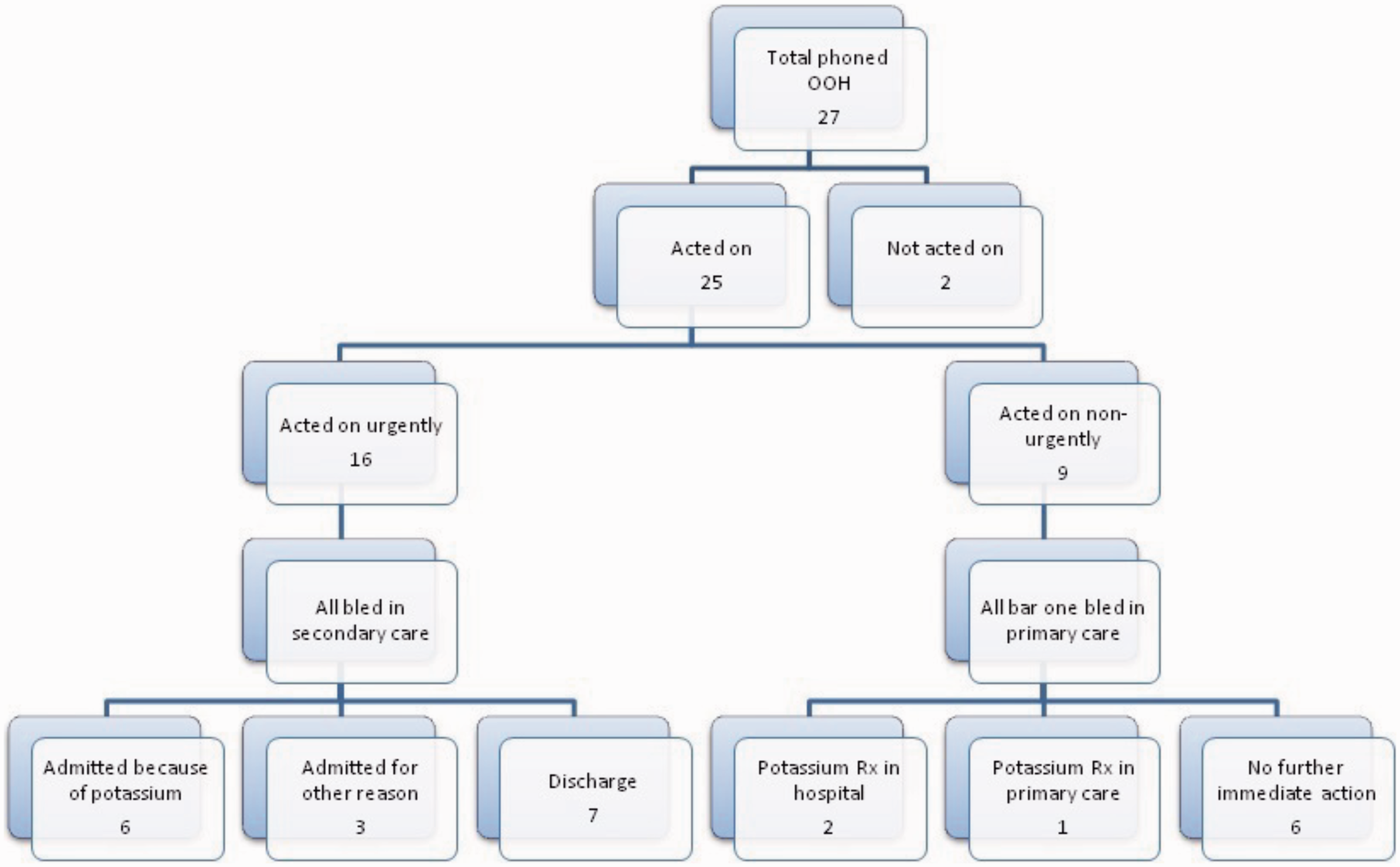
Urgency, nature and outcome of actions taken when potassium results were phoned out of hours. See text for details.
Discussion
It is intuitive that rapid communication of critical laboratory results should improve patient safety. Yet, proving the impact of laboratory results on outcomes is challenging,4 and there is wide variation in how laboratories manage high-risk results.5 In the current study, we examined the actions taken when a result is phoned, and the urgency and outcome of such actions.
Our most striking finding was that, during the six months of the study, only 22 potassium results breached the action limits, out of almost 130,000 potassium requests from primary care (0.016% of requests). These and a further five potassium results just inside the action limits were phoned to the OOH service. The small sample size may have reflected the exclusive focus on results phoned to primary care OOH. Despite the small numbers, potassium results that were the same or very similar were treated differently by different clinicians. Also, practice policy on processing of abnormal results varied, further confounding the assessment of the impact of phoning.
Our study was prospective, enabling prompt and detailed follow-up of individual outcomes in a way that would not be possible with retrospective review. Results were communicated to a single OOH hub, staffed by a small pool of experienced general practitioners, thereby minimising variation from this source. By focusing on one analyte, we did not have to establish whether a result was phoned because of abnormalities in one analyte or another.
Our study has established the feasibility of investigating immediate outcomes of phoned results. The final sample size was too small to permit any firm conclusions to be drawn. However, the impact of confounders could be reduced by performing a larger and longer UK-wide study, which could help to inform future guidance.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
MJM.
Contributorship
Both authors contributed equally to the study.
