Abstract
Background
To assess the vitamin D nutritional status, serum total 25-hydroxyvitamin D (25(OH)D) concentration is measured. We used six automated 25(OH)D immunoassays (AIAs) available in Japan and certified by the Vitamin D Standardization Program (VDSP) at the U.S. Center for Disease Control and Prevention to assess the concordance of the assay results.
Methods
Serum total 25(OH)D concentrations in SRM 972a and 20 serum samples from patients were determined using three liquid chromatography-tandem mass spectrometry (LC-MS/MS) and six AIAs (pilot study), and an additional 110 serum samples were assessed by the six AIAs (surveillance study). The assay bias from the results of LC-MS/MS by Chiba University or consensus values (i.e. average of six AIAs) was estimated using the procedure described in CLSI document EP09-A3.
Results
LC-MS/MS at Chiba University could completely separate 25(OH)D2, 25(OH)D3 and 3-epi-25(OH)D3, and the observed values including total 25(OH)D in SRM 972a were all within ±1·SD of the assigned values. All AIAs produced results greater than ±3·SD. In the pilot study, four of the six AIAs had an average percentage bias, as estimated by confidence interval (CI), larger than ±5% (acceptance criterion in CLSI); the bias converged from −6.5% to 3.2% after adjustment by LC-MS/MS. In the surveillance study, 25(OH)D concentrations in AIAs all adjusted to LC-MS/MS converged within ±5% from consensus values. However, some AIAs showed negative or positive bias from the consensus values.
Conclusions
Current AIAs in Japan continue to lack standardization. Manufacturers should implement quality assurance strategies so that their values more closely align to those of standard reference material 972a.
Keywords
Introduction
One of the many roles of vitamin D is to regulate the calcium concentrations in the blood. Among various D vitamers, 1,25-dihydroxyvitamin D2 and D3 are the hormonally active metabolites of vitamin D, both of which are produced in the kidneys via enzymatic hydroxylation of their inactive precursors, 25-hydroxyvitamin D2 and D3 (25(OH)D2 and 25(OH)D3). 1,25-Dihydroxyvitamin D has a short serum half-life of approximately 4–6 h. In contrast, 25(OH)D (i.e. the sum of 25(OH)D2 and 25(OH)D3) has a long serum half-life of approximately two to three weeks. Thus, the estimation of vitamin D nutritional status can be done by measuring 25(OH)D concentrations in the serum. 1
Although serum 25(OH)D concentrations have been analysed using many methods – (radioimmunoassay [RIA], competitive protein-binding assay [CPBA], automated immunoassay and high-performance liquid chromatography [HPLC]) – the intermethod variability, especially in the automated immunoassay, confounds the diagnosis of hypovitaminosis D. 2 In this situation, a reference measurement procedure (RMP) based on liquid chromatography-tandem mass spectrometry (LC-MS/MS) was proposed for serum total 25(OH)D measurement, 3 and the National Institute of Standards and Technology (NIST) developed a standard reference material (SRM 972a), 4 in which certified values were assigned with the use of RMP. Most manufacturers providing automated immunoassay use SRM 972a to make their assay values traceable to those obtained with RMP (LC-MS/MS).5,6
In addition, these manufacturers have joined the Vitamin D Standardization Program (VDSP),7,8 which is conducted as a collaboration between the NIST and the Center for Disease Control and Prevention (CDC). In the VDSP, assay values for 40 serum samples from healthy subjects measured with each automated immunoassay are adjusted to values measured by NIST/CDC with RMP (LC-MS/MS), which are independent from those measured by automated immunoassay. Herein, however, those assay values are not validated to provide concordant values, i.e. the same result from the same sample, when serum samples from patients were measured with each automated immunoassay. Therefore, this study aims to evaluate for the first time in Japan the standardization of assay values for serum total 25(OH)D concentration measured with automated immunoassays.
Materials and methods
Specimens
A total of 130 serum specimens were randomly selected from residual serum samples from adult patients submitted for routine laboratory testing at two academic medical centres (Chiba University Hospital and University of Toyama Hospital). Each serum specimen was divided into nine aliquots. These were distributed frozen on dry ice to the laboratories of six manufacturers and three universities in order to measure serum 25(OH)D concentration using the laboratory's own assay procedure. In accordance with the Helsinki Declaration, the study protocol was approved by the Protection of Human Subjects Committee of Chiba University Hospital (Ethics Committee approval number 263, 25 June 2014) and University of Toyama Hospital (Ethics Committee approval number 23–51, 5 September, 2011).
SRM 972a was purchased from NIST, and consisted of four vials (levels 1 through 4) of frozen serum from healthy donors with different concentrations of 25-hydroxyvitamin D (25(OH)D2, 25(OH)D3, 3-epi-25(OH)D3 and total 25(OH)D). Levels 1, 2 and 3 were prepared from pools of human serum with endogenous concentrations of 25(OH)D. Level 4 was prepared from a pool of human serum that was fortified with 3-epi-25(OH)D3. Here, the total 25(OH)D concentration is the sum of 25(OH)D2 and 25(OH)D3 and is expressed as nmol/L in terms of 25(OH)D3 equivalent. For conversion to mass units (ng/mL), molar units (nmol/L) were multiplied by a factor of 0.40065.
Assays
Serum concentrations of 25(OH)D2, 25(OH)D3 and 3-epi-25(OH)D3 were measured by three LC-MS/MS in the laboratories of Chiba University, University of Toyama and Kobe Pharmaceutical University. At the Chiba University, serum samples were extracted by supported liquid extraction and derivatized by a Cookson-type reagent (DAPTAD, 4-(4′-dimethylaminophenyl)-1,2,4-triazoline-3,5-dione). The derivatized three vitamin D metabolites (25(OH)D3, 3-epi-25(OH)D3 and 25(OH)D2) were subjected to LC–MS/MS, ionized by electrospray ionization (positive-ion mode) and detected by selected reaction monitoring. 9 At the University of Toyama, serum samples were extracted by supported liquid extraction and subjected to LC-MS/MS, electrospray ionization in the positive-ion mode (ACQUITY TQD, Waters, Co., Ltd, MA, USA). The hexa-deuterated vitamin D3 (MEDICAL ISOTOPES, Inc., NH, USA) was used as an internal standard. 10 At Kobe Pharmaceutical University, serum concentrations of vitamin D metabolites were measured using a previously reported modified LC-APCI-MS/MS method.11,12 Internal standard d6–25(OH)D3 and acetonitrile were added to serum samples. Vitamin D metabolites were extracted with ethyl acetate and derivatized by DMEQ-TAD(4-(2-(6,7-dimethoxy-4-methyl-3-oxo-3,4-dihydroquinoxalinyl)ethyl)-1,2,4-triazoline-3,5-dione). The derivatized three vitamin D metabolites (25(OH)D3, 3-epi-25(OH)D3 and 25(OH)D2) were subjected to LC-MS/MS (API-3000; Applied Biosystems, Inc.) ionized by atmospheric pressure chemical ionization (APCI), MS data were collected in the positive ion mode and quantitative analysis was carried out using MS/MS-MRM of the precursor/product ion. The analytical column was a CAPCELL PAK C18 UG120 (5 μm; 4.6 I.D. × 250 mm; SHISEIDO, Tokyo, Japan). The mobile phase consisted of (A) acetonitrile and (B) distilled water (0–5 min A = 30%, 5–35 min (A)=30→95%) at a flow rate of 1.0 mL/min. In this analytical condition, separation of 3-epi-25(OH)D3 and 25(OH)D3 was incomplete.
On the other hand, serum total 25(OH)D concentrations were measured by six automated 25(OH)D immunoassays (ARCHITECT i2000SR [ARCHITECT 25-OH Vitamin D; Abbott, Abbott Park, IL, USA], UniCel DxI 800 [Access 25(OH) Vitamin D Total; Beckman Coulter, Inc., Brea, CA, USA], Cobas e601 [Elecsys Vitamin D total II, electrochemiluminescence; Roche Diagnostics, Basel, Switzerland], Liaison XL [LIAISON 25 OH VITAMIN D TOTAL; DiaSorin S.p.A., Saluggia, Italy; distributed by Kyowa Medex Co., Ltd], ADVIA Centaur XP [ADVIA Centaur Vitamin D Total; Siemens Healthcare Diagnostics, Tarrytown, NY, USA] and Lumipulse G1200 [Lumipulse G 25-OH Vitamin D; Fujirebio Inc., Tokyo, Japan]) in the manufacturer's laboratories. Because this study does not aim to rank the automated immunoassays, we assigned the labels AIA-A to F to each automated assay. Each immunoassay was calibrated and performed according to the manufacturer's instructions. Good analytical characteristics required by the VDSP (i.e. ≤ 10% imprecision, < 20 nmol/L limit of quantitation [LoQ], 100 ± 10% recovery of 25(OH)D3 and cross-reactivity for 25(OH)D2 and 3-epi-25(OH)D3)5,6,7 have been declared by each manufacturer and/or reported in previous literature (Table 1).13–17 In this surveillance, within-day reproducibility (CV at 32.4 nmol/L) was 8.9%, 5.7%, 6.6%, 9.2%, 3.9% and 7.4% in AIA-A, AIA-B, AIA-C, AIA-D, AIA-E and AIA-F, respectively.
Characteristics of current automated 25(OH)D immunoassays provided from the manufacturers' information.
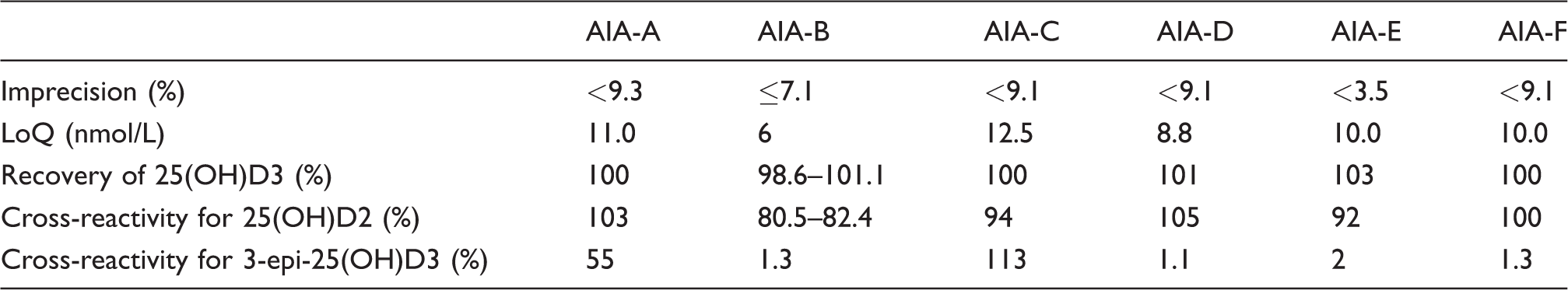
AIA: automated immunoassay.
Statistical analysis
In a pilot study, total 25(OH)D concentrations in 20 serum specimens (randomly selected from the above 130 serum specimens) were measured by LC-MS/MS (at the University of Toyama and Kobe Pharmaceutical University) and automated immunoassay, and the assay values were compared with those of LC-MS/MS at Chiba University (Figure 1). In the LC-MS/MS, the total 25(OH)D concentration was calculated as the sum of 25(OH)D2 and 25(OH)D3. Bias from the assay values of Chiba University was estimated by the procedure described in CLSI document EP09-A3 ‘Measurement Procedure Comparison and Bias Estimation Using Patient Samples; Approved Guideline-Third Edition’, 18 and accepted bias was interpreted as ±5%.5–7,19 This estimation is the same procedure used in the VDSP. In CLSI document EP09-A3, the 95% confidence interval (CI) of the accepted bias was calculated as mean ± 1.984·SE of percentage difference between results by each measurement and the assay values of LC-MS/MS by Chiba University.
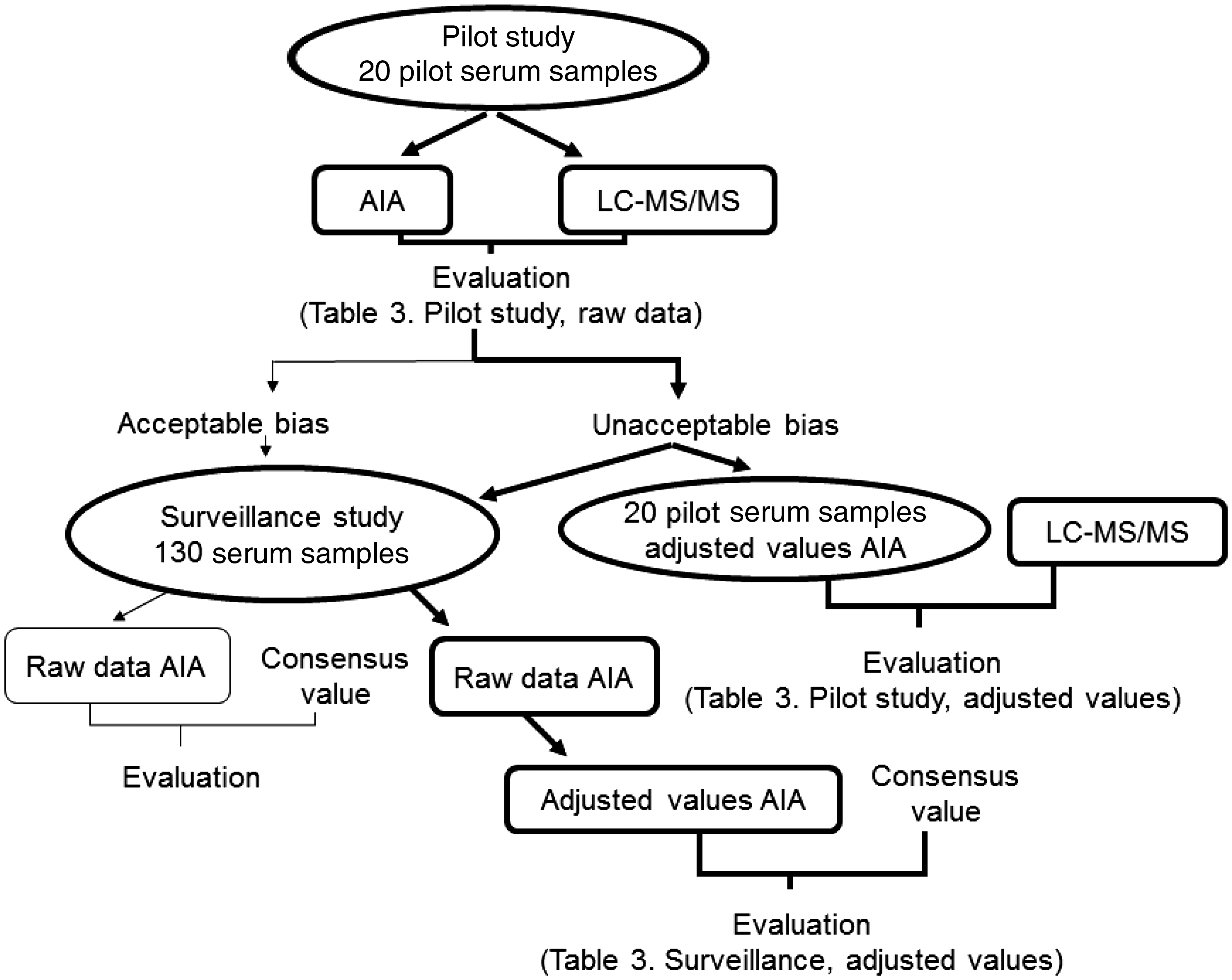
Outline of the assay process for surveillance evaluation of assay values for serum total 25-hydroxyvitamin D concentration in Japan. Bold line indicates this study.
In the next step, total 25(OH)D concentrations in 130 serum specimens determined by automated immunoassay were compared with consensus values (Figure 1), which were derived by averaging the results from the six automated immunoassays. Before averaging, the results from each automated immunoassay (raw data) were adjusted to those of LC-MS/MS by Chiba University, because automated immunoassay had bias from LC-MS/MS as described later. Bias from the consensus values was estimated using the procedure described in CLSI document EP09-A3 in the same manner as for the pilot study.
Results
Measurement of SRM 972a
SRM 972a was measured by three LC-MS/MS and six automated immunoassays (Table 2). LC-MS/MS at Chiba University could completely separate 25(OH)D2, 25(OH)D3 and 3-epi-25(OH)D3, and the observed values of 25(OH)D2, 25(OH)D3, 3-epi-25(OH)D3 and total 25(OH)D in SRM 972a were all within ±1·SD of the assigned values. Total 25(OH)D concentrations measured at University of Toyama were within ±2·SD of the assigned values, but this measurement did not separate 25(OH)D2 and 3-epi-25(OH)D3. There was some indication that LS-MS/MS at Kobe Pharmaceutical University gave slightly lower estimates than other measurements for SRM 972a. When the overall means and the inter-laboratory (i.e. between laboratories) variability of 25(OH)D3 and 25(OH)D2 concentrations in each level for the different LC-MS/MS were expressed as %CV, there was very good agreement on 25(OH)D3 concentration and overall %CVs ranged from 3.9% to 5.7%, except for level 4 (40.7%). There was poor agreement on 25(OH)D2 concentration.
Observed values of SRM 972a using three LC-MS/MS and six automated immunoassays.
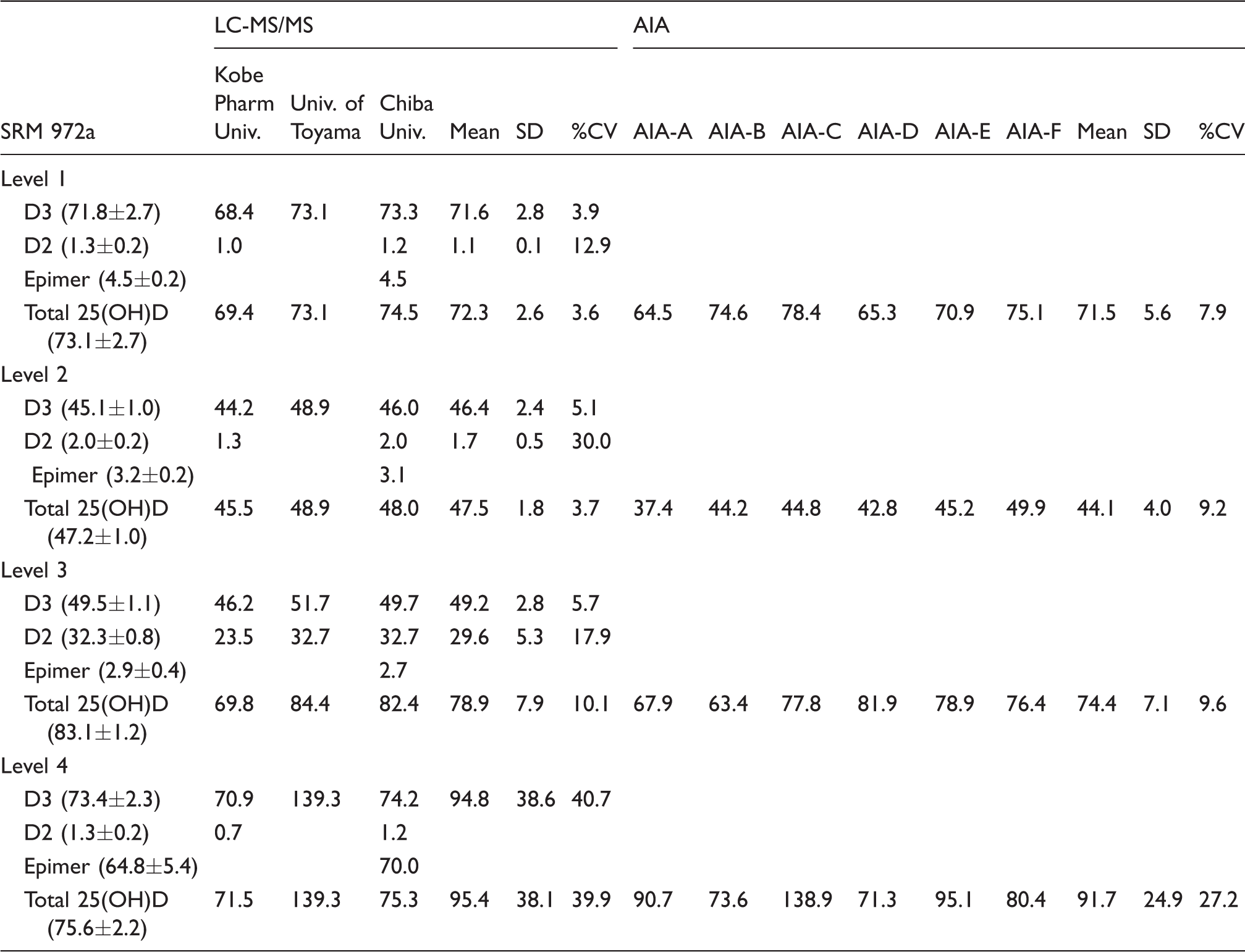
AIA: Automated immunoassay; LC-MS/MS: liquid chromatography-tandem mass spectrometry; D3: 25(OH)D3; D2: 25(OH)D2; Epimer: 3-epi-25(OD)D3.
Note: All values are presented as nmol/L. Assigned value of SRM 972a in parentheses.
On the other hand, the observed values of SRM 972a (levels 1–4) for total 25(OH)D measured by most of the automated immunoassays were distributed beyond ±3·SD. The inter-laboratory variability expressed as %CVs ranged from 7.9% to 9.6%, up to 27.2% in level 4, and there was less agreement for the total 25(OH)D concentration, which was twice that of LC-MS/MS. The automated immunoassays were found to react with both 25(OH)D2 and 25(OH)D3, but not with 3-epi-25(OH)D3 (except for AIA-A, AIA-C and AIA-E).
Comparison of total 25(OH)D concentrations between automated immunoassay and LC-MS/MS in a pilot study
As a pilot study, serum total 25(OH)D concentrations in SRM 972a (levels 1 to 4) and 20 serum specimens assessed by six automated immunoassays (AIA-A to AIA-F) and two LC-MS/MS (University of Toyama and Kobe Pharmaceutical University) were compared with those by LC-MS/MS of Chiba University (Figure 2(a)). Significant relationships were observed in all automated immunoassays and LC-MS/MS (University of Toyama and Kobe Pharmaceutical University) against LS-MS/MS at Chiba University (r = 0.847 to 0.975: Table 3). Serum total 25(OH)D concentrations in 20 serum specimens determined by LC-MS/MS at Chiba University were distributed from 27.4 nmol/L to 48.5 nmol/L. These serum samples contained 0.3–2.1 nmol/L of 25(OH)D2 (0.9–7.2% of total 25(OH)D) and 1.1–2.1 nmol/L of 3-epi-25(OH)D3. In the Passing-Bablok regression analysis, assay values of SRM 972a were excluded, because percentage differences from the LC-MS/MS in six automated immunoassays were significantly higher than those in 20 serum specimens (Figure 2(b)). Percentage differences in serum specimens were distributed within ca. ±24%, ranging from −22.1% to 23.6%, but those of SRM 972a exceeded ±24%.
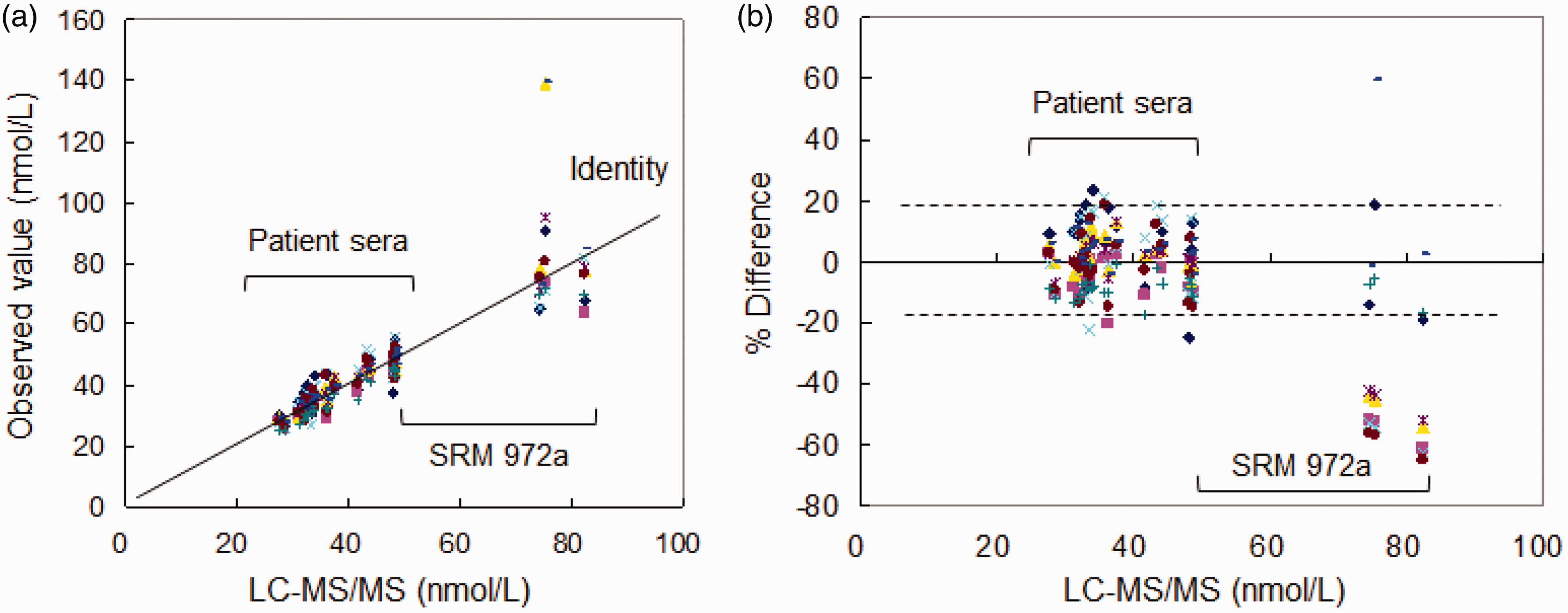
Comparison of total 25(OH)D results before adjustment in 20 serum samples from patients and SRM 972a levels 1–4 between LC-MS/MS by Chiba University and six automated immunoassays and two LC-MS/MS. (a) Passing-Bablok plot. (b) Percentage difference plot (dashed line indicates 95% limits of agreement in all measurements).
Concordance correlation analysis of serum specimens by LC-MS/MS and automated immunoassays.
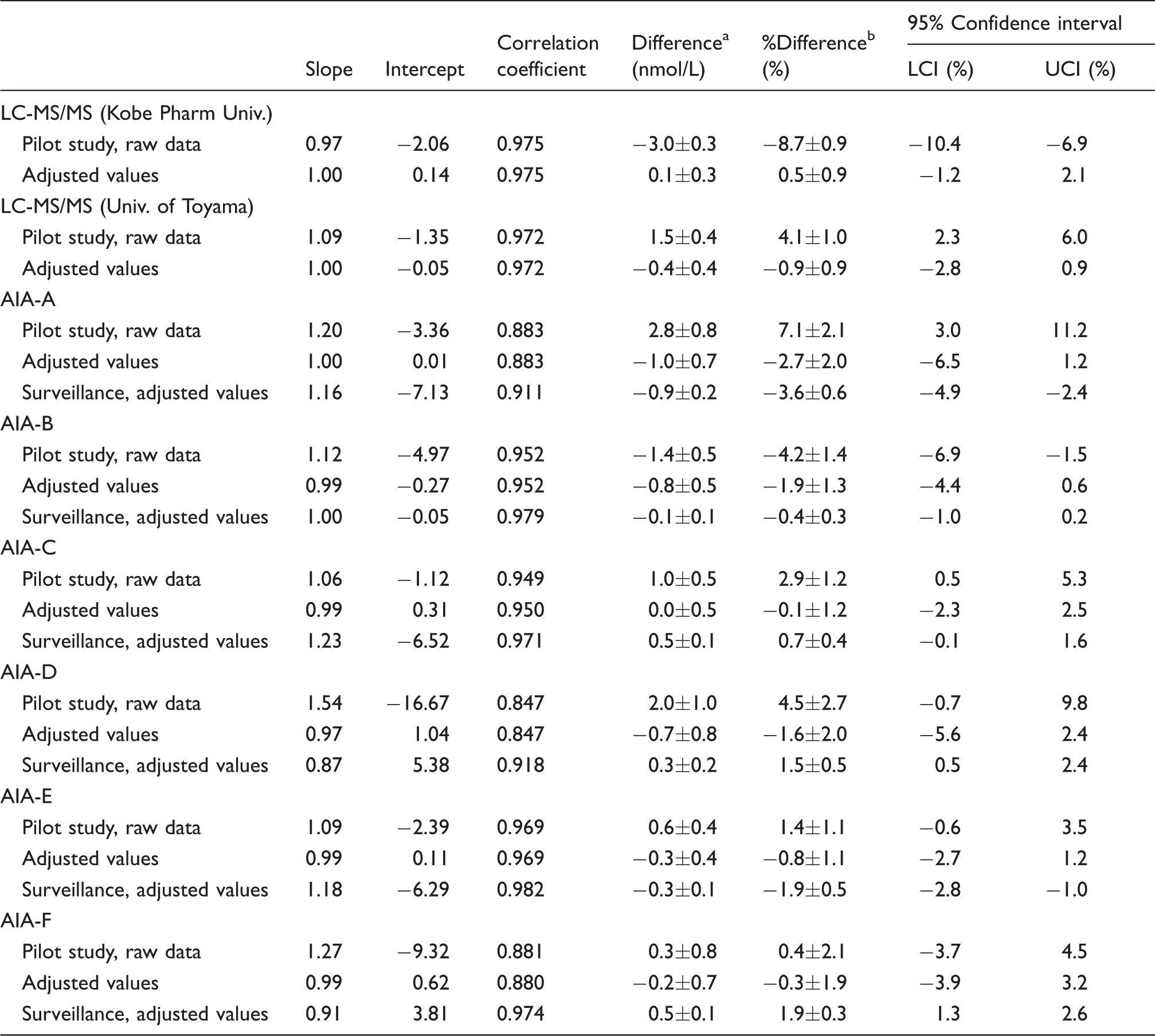
Note: All values were compared with those of LC-MS/MS by Chiba University (pilot study) or consensus values (surveillance study). Slope and intercept were calculated by Passing-Bablok regression analysis against LC-MS/MS by Chiba University or consensus values.
AIA: Automated immunoassay; LC-MS/MS: liquid chromatography-tandem mass spectrometry; LCI: lower confidence interval; UCI: upper confidence interval.
aDifference means average (±SE) of absolute difference from that of LC-MS/MS by Chiba University or consensus values.
b% Difference means average (±SE) of percentage difference from that of LC-MS/MS by Chiba University or consensus values.
When LCI and UCI were calculated from the percentage difference according to the procedure of CLSI document EP09-A3, both LCI and UCI distributed outside of ±5% in LC-MS/MS and automated immunoassays, except for AIA-E and AIA-F (Table 3). Assay values of SRM 972a were excluded from this calculation.
Next, we attempted to adjust the assay values of two LC-MS/MS (University of Toyama and Kobe Pharmaceutical University) and six automated immunoassays using an equation derived from Passing-Bablok regression (i.e. slope and intercept) against LC-MS/MS by Chiba University. After the adjustment, the LCI converged to > −6.5%, and the UCI converged to < 3.2% in all analyses (Table 3).
Comparison studies on consensus value for total 25(OH)D concentrations adjusted to LC-MS/MS in 130 serum specimens among automated immunoassays
In the next step, serum total 25(OH)D concentrations in 130 serum specimens, including 20 serum samples used in the pilot study, were assayed by six automated immunoassays and observed concentrations were adjusted using the equation obtained from the Passing-Bablok regression in the pilot study. Then, the adjusted values were compared with consensus values.
Adjusted values by six automated immunoassays showed significant relationships with the consensus values (r= 0.911 to 0.982: Table 3 and Figure 3(a)). Percentage differences from the consensus values in six automated immunoassays varied from −22.8% to 19.9% (Figure 3(b)). When LCI and UCI were calculated from the percentage differences according to the procedure of CLSI document EP09-A3, both LCI and UCI of the adjusted values were distributed within ±5% (Table 3). However, the slope and intercept of the regression lines in six automated immunoassays varied from 0.87 to 1.23 and −7.13 to 5.38, respectively.
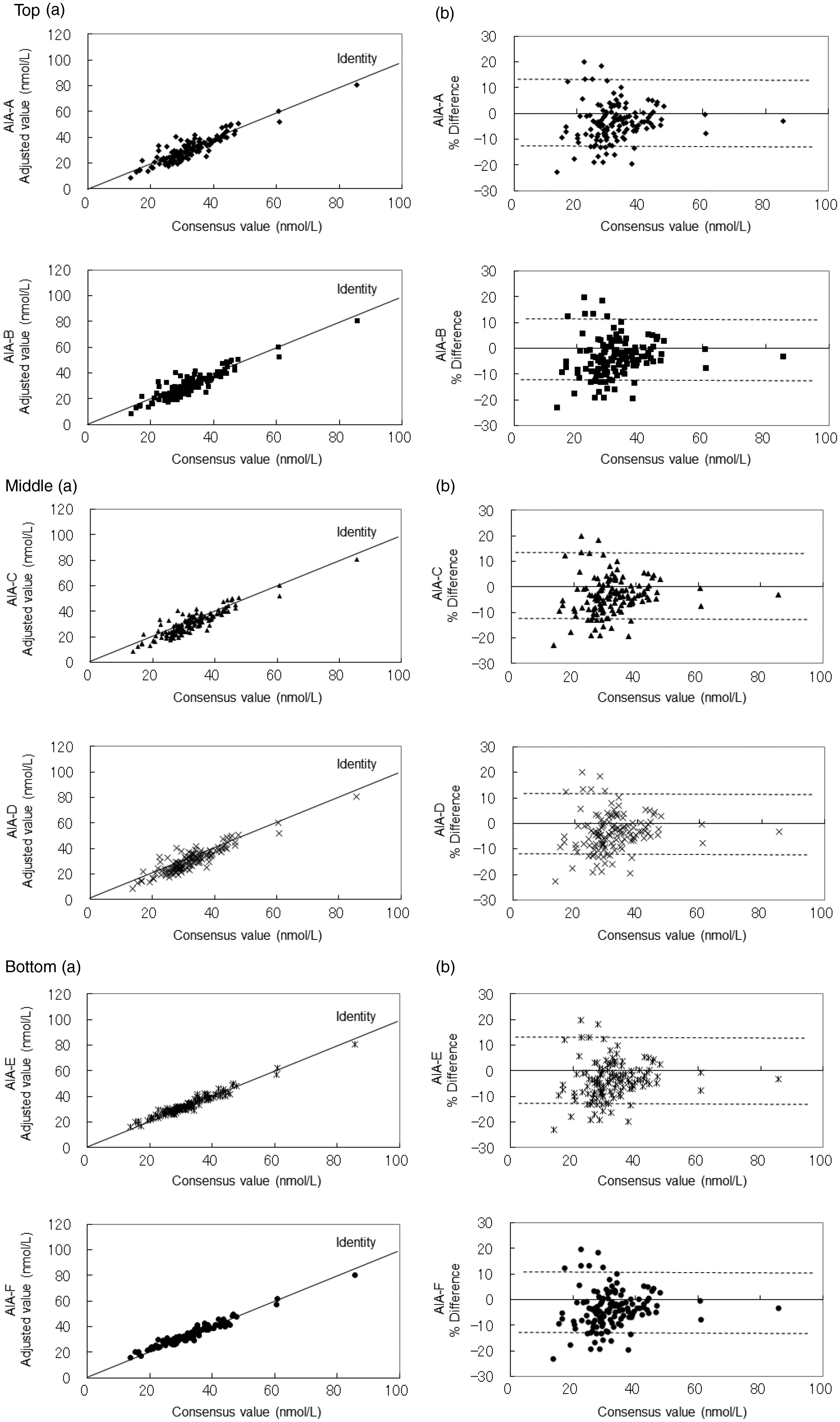
Top, middle and bottom: Comparison of total 25(OH)D results in 130 serum samples from patients between consensus values adjusted to LC-MS/MS by Chiba University and six automated immunoassays. (a) Passing-Bablok plot. (b) Percentage difference plot (dashed line indicates 95% limits of agreement in all measurements).
Discussion
In the development of reagents for serum total 25(OH)D measurement, each manufacturer providing automated immunoassay used SRM 972a to ensure that their values are traceable to RMP and also joined the VDSP (Figure 1). Although LC-MS/MS in Japan (University of Toyama, Kobe Pharmaceutical University and Chiba University) did not join the VSDP, the interlaboratory CV of 25(OH)D3 in SRM 972a levels 1, 2 and 3 of LC-MS/MS converged to less than 5.7%. Further, they used their own calibrator instead of SRM 2972a. In the VDSP, each manufacturer assayed 40 serum specimens (obtained from healthy donors) to perform a bias assessment and adjust their calibration (Phase 1). When the bias (LCI and UCI) was within ±5.0%, an additional 10 serum specimens, also from health donors, were assayed to reconfirm that their values matched the reference values (Phase 2). However, the VDSP did not ensure that the assay values of each automated immunoassay were consistent with the others.
We measured SRM 972a using six automated immunoassays (AIA-A to AIA-F), and found that the observed values for total 25(OH)D in most of the automated immunoassays were distributed beyond ±3·SD of the assigned value (Table 2). Moreover, assay values of the 20 serum specimens in the pilot study exceeded ±5% (outside of LCI and UCI) in AIA-A, AIA-B and AIA-D as calculated from the percentage difference from LC-MS/MS by Chiba University, although these assays had joined the VDSP. In this study, LC-MS/MS by Chiba University showed concordant results with the assigned values of SRM 972a levels 1 to 4 within ±1·SD. Therefore, we selected LC-MS/MS by Chiba University as the reference method in this study.
In automated 25(OH)D immunoassays that have been guaranteed by the VDSP, factors present in patient serum that influence assay values are reported to be 25(OH)D213,14 and 3-epi-25(OH)D3.16,17 We speculated that these factors may have been responsible for the variation in AIA-A, AIA-B and AIA-D. Le Goff et al. 14 suggested that cross-reactivity of 25(OH)D2 in the total 25(OH)D measurement declared by the manufacturer should be verified by each end-user (i.e. clinical and research laboratories). They also showed that the Architect underestimated 25(OH)D2 by 36.6%, and the Centaur overestimated 25(OH)D2 by 30%. In our study, we were able to estimate the cross-reactivity of endogenous 25(OH)D2 (32.3 nmol/L) in the total 25(OH)D measurement by subtracting assay values of SRM 972a level 2 from level 3. As a result, AIA-B and AIA-F would be considered to underestimate 25(OH)D2, while AIA-D overestimated 25(OH)D2 (i.e. 67.9–37.4 = 30.5 nmol/L for AIA-A, 63.4–44.2 = 19.2 nmol/L for AIA-B, 77.8–44.8 = 33.0 nmol/L for AIA-C, 81.9–42.8 = 39.1 nmol/L for AIA-D, 78.9–45.2 = 33.7 nmol/L for AIA-E and 76.4–49.9 = 26.5 nmol/L for AIA-F in Table 2). Although the 20 pilot serum samples contained < 2.1 nmol/L of 25(OH)D2 (<7.2% of total 25(OH)D), these results partially explained the negative bias in AIA-B and the positive bias in AIA-D observed in the pilot study.
While 3-epi-25(OH)D3 is a stereoisomer of 25(OD)D3, it can be converted to 3-ep-1,25-dihydroxyvitamin D3, which does not exert hormonal activity to raise serum calcium concentrations. 17 We estimated the cross-reactivity of 3-epi-25(OH)D3 (64.8 nmol/L) in the total 25(OH)D measurement by subtracting the assay value of SRM 972a level 1 from level 4 (i.e. 26.2 nmol/L for AIA-A, −1.1 nmol/L for AIA-B, 60.5 nmol/L for AIA-C, 6.0 nmol/L for AIA-D, 24.2 nmol/L for AIA-E and 5.3 nmol/L for AIA-F, Table 2). Large amounts of 3-epi-25(OH)D3 seemed to influence total 25(OH)D measurement in AIA-A, AIA-C and AIA-E; however, cross-reactivity of exogenously added 3-epi-25(OH)D3 was reported to be different from that of endogenous 3-epi-25(OH)D3. 17 Some automated immunoassays reacted 51% with exogenous 3-epi-25(OH)D3, but minimally with endogenous 3-epi-25(OH)D3.16,17 Therefore, we cannot conclude at this time that the positive bias in AIA-A results from 3-epi-25(OH)D3.
Considering these observations, discrepancies in the assay values of AIA-A, AIA-B and AIA-D from those of LC-MS/MS by Chiba University could be caused not only by 25(OH)D2 and 3-epi-25(OH)D3, but also other factors not revealed in this study. However, the assay values of automated immunoassays including AIA-A, AIA-B and AIA-D converged to an acceptable bias (i.e. < ±5%) when their values were adjusted to LC-MS/MS by Chiba University (Table 3). These convergences were also observed in the surveillance study, in which assay values (raw data) adjusted to LC-MS/MS by Chiba University were compared with their consensus values. However, the slope and intercept of the regression lines in six automated immunoassays varied from 0.87 to 1.23 and −7.13 to 5.38, respectively. This variation was not observed in the pilot study.
Hence, in the context of the findings of this preliminary study, there is a lack of standardization in current automated 25(OH)D immunoassays in Japan. Recent studies reported that current automated 25(OH)D immunoassays still remain unstandardized.20,21 Although adjustment using the Passing-Bablok regression equation against LC-MS/MS revealed a converged bias (i.e. LCI and UCI as < ±5%) from consensus values in each automated immunoassay, this was an apparent standardization. This was because the bias in AIA-A and AIA-E tended to a negative bias, and that of AIA-D and AIA-F tended to a positive bias (Table 3). Furthermore, the high percentage bias (>±10%) indicated a high absolute bias as >±3 nmol/L (maximally to ±10 nmol/L) (Figure 3(b)). These biases could mislead clinical diagnosis of hypovitaminosis D. Furthermore, adjustment by regression parameters by the end-users broke the traceability chain of measurement. In addition, adjustment by standardization resulted in an unresolved variation in the slope-intercept form.
Considering these limitations in automated 25(OH)D immunoassays, we suggest that the respective manufacturers should implement quality assurance measures, so as to, at a minimum, more closely align their assay values to the assigned values of SRM 972a. After that, we are planning to conduct a survey as was done in this study. As reported by Sempos et al., 6 when unacceptable bias from LC-MS/MS is found in automated methods in the periodic survey, there is a need to warn the manufacture to revise their quality assurance.
Footnotes
Acknowledgements
We deeply thank all the subjects for participating in this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Research Foundations of the Japan Society of Clinical Chemistry and Chiba Institute of Science.
Ethical approval (including reference number)
In accordance with the Helsinki Declaration, the study protocol was approved by the Protection of Human Subjects Committee of Chiba University Hospital (Ethics Committee approval number 263, 25 June 2014) and University of Toyama Hospital (Ethics Committee approval number 23–51, 5 September 2011).
Guarantor
HI.
Contributorship
HI, TO, MT, NH, KH and MM designed the research; SK, TI, KM, MS and NT performed the research and interpretation of dada; MN, FN, MY and IK were involved in gaining ethical approval; HI, SK, MN and MY wrote the first draft of the manuscript; and all authors reviewed and edited the manuscript and approved the final version of the manuscript.
