Abstract
Background
Diagnosis of Wilson’s disease is currently performed using caeruloplasmin as a first-line screening test; however, this test has well-described limitations. Monitoring of known Wilson’s disease patients often uses 24-h urine collection; however, this is inaccurate in children. Methods for directly measuring plasma free copper have been described, but no reference interval data exist for a paediatric population.
Methods
An inductively coupled plasma mass spectrometry method for measuring free copper was developed and validated, using ultracentrifugation. A paediatric reference interval was generated using 85 plasma samples from children attending outpatient clinics at University Hospital Southampton.
Results
Results showed no significant contamination of copper using the ultracentrifugation technique, and validation showed the method was accurate and precise with an analytical coefficient of variation between 5 and 7% depending on the concentration of free copper.
Conclusions
We describe the use and validation of an ultrafiltration inductively coupled plasma mass spectrometry method for plasma free copper with the first published paediatric reference interval. Free copper could provide much needed assistance for the monitoring of Wilson’s disease in children and also for adults.
Keywords
Introduction
Wilson’s disease (WD) is an autosomal recessive disorder with an estimated prevalence of 1 in 30,000. 1 It results from mutations in ATP7B, a gene that encodes a copper-transporting P-type ATPase responsible for copper excretion. Homozygosity for ATP7B mutations leads to toxic accumulation of copper and deposition predominantly in the liver and the brain, but also in the periphery of the iris and the kidney. Defective ATP7B gene product function prevents effective incorporation of copper into caeruloplasmin resulting in the generally low copper and caeruloplasmin levels seen in patients with WD. In normal people, less than 5% of copper exists as free copper in the circulation with the rest being bound to caeruloplasmin, transcuprein, albumin and amino acids. 2 In children, WD usually presents after three years of age with either incidental findings of abnormal liver function tests or evidence of chronic liver disease. In rare instances, it can present with acute hepatic failure and haemolytic anaemia. 3 Neurological presentation in childhood is rare, but patients may present as young as eight years of age.
Diagnosis of WD remains problematic, and a rapid and reliable diagnostic test is lacking. At present, serum caeruloplasmin is used as a first-line screening test. However, it is known that a significant proportion of patients with WD have caeruloplasmin levels within the reference interval. 4 Most laboratories use immunonephelometric methods and this will measure both caeruloplasmin and the biologically inactive apo-form which does not contain any copper atoms. Caeruloplasmin may also be measured enzymatically via determination of its oxidase activity, and this is thought to be superior as only the active form is measured. 4 Low caeruloplasmin values can be found in liver disease, nephrotic syndrome, protein-losing enteropathy and in acaeruloplasminaemia. 4 A 24-h urinary copper excretion is almost always elevated in patients with WD; however, this relies on a complete collection which can be very difficult to obtain in children. 5 Additionally, concentrations can be artificially high if there is acute hepatic inflammation, and there is considerable overlap in values with other causes of liver disease. WD can also be diagnosed by assessing dry weight copper content of liver biopsy samples; however, this can be problematic due to the often marked heterogeneity of levels of copper deposition within the liver. 6 Diagnosis of WD is particularly difficult in children due to its more obscure and variable clinical presentations. 3 Delayed diagnosis is a significant clinical issue as Wilson’s can be fatal if left untreated. 1
The organ dysfunction in WD occurs due to the toxicity associated with the accumulation of unbound free copper. Determining the free copper concentration in plasma could be a useful solution to overcoming the difficulties associated with the current diagnostic tests available. 7 It is possible to calculate estimated free copper, or non-caeruloplasmin copper (NCC), using an equation; however, there are significant limitations associated with this approach. 8 A number of assumptions are made, and it has been shown that use of this calculation generates biologically impossible negative results as well as elevated results in the majority of healthy people. 9 NCC is therefore unreliable as a diagnostic test and of limited use as a test to monitor known cases of WD.
More recently, there has been interest in direct measurement of free copper including ‘exchangeable copper’ as a tool to aid with the screening, diagnosis and monitoring of WD. 7 There is evidence that ‘exchangeable copper’ is superior to free copper for diagnosis of WD. 10 Free copper, however, may have an important role in monitoring in patients with WD due to its ease of measurement, as it can be performed on routine plasma samples without the need for cumbersome 24-h urine collections or the strict sample and analytical requirements of ‘exchangeable copper’(samples require the addition of ethylenediaminetetraacetic acid (EDTA) so that copper is cleaved from the exchangeable pool of non-caeruloplasmin bound copper to provide an indication of all the copper not associated with caeruloplasmin). A method has been previously described using inductively coupled plasma mass spectrometry (ICP-MS) with an initial ultrafiltration step, along with generation of a reference interval for an adult population. 2 There are currently no data available to guide clinicians regarding normal values for free copper in children. Here, we describe the use and validation of an ICP-MS method for free copper as described by McMillin et al., 2 validated with paediatric samples, and the generation of a reference interval for free copper for the first time in a normal paediatric population.
Materials and methods
For direct free copper analysis
Patient samples were stored at 4℃ for a minimum of 48 h to allow a steady state to occur in free copper due to exchanges between amino acids and plasma proteins. The samples were then centrifuged in a fixed angle microfuge (Beckman Coulter Allergra 6R Microfuge [Beckman Coulter California, USA]) at 10,000 r/min for 5 min, and transferred to the Ultrafiltration device at a 90° angle to prevent air locks. Ultrafiltration was then performed using Centrifree Ultrafiltration Devices (Millipore, Cork Ireland) with a molecular weight cut-off of 30 kDa and centrifugation at 2000 × g for 60 min. Test samples were prepared with 50 µL 1 mg/L rhodium standard solution (British Drug Houses, Poole, UK), 100 µL 1000 mg/L copper standard solution (Fisher Scientific, Loughborough, UK), 100 µL sample and 1.2 mL distilled and de-ionized water. They were then vortex-mixed and analysed on the Elan 6100 ICP-MS (PerkinElmer Elan 6100 DRC Plus, ICP-MS [PerkinElmer SCIEX, Beaconsfield, UK]).
For total copper analysis
Patient samples were stored at 4℃, and then centrifuged in a fixed angle microfuge at 10,000 r/min as described above. Test samples were prepared with 50 µL 1 mg/L rhodium standard solution, 20 µL sample plasma and 1.48 mL distilled and de-ionized water. They were then vortex-mixed and analysed on the Elan 6100 ICP-MS.
Inductive-coupled plasma mass spectrometry
Samples were analysed on the PerkinElmer Elan 6100 DRC Plus ICP-MS platform. ICP-MS conditions were optimized using TS, which was aspirated to establish the optimum nebulizer argon gas flow at ionic mass 114.904 to achieve a maximum intensity with a low cerium oxide (CeO) level. The instrument settings for copper analysis were radio frequency power: 1200 W; argon gas flow rates: nebulizer 0.9–1.0 L/min, auxiliary 1.2 L/min and plasma 15 L/min; data acquisition parameters: scanning mode: peak hopping; mass: masses Cu 65 and rhodium internal standard 103; dwell time: 50 ms, sweeps: 100 sweeps per reading, replicates: 2 replicates for total copper and dwell time: 100 ms, sweeps: 100 sweeps per reading replicates: 2 replicates for UF copper.
Sample collection
Plasma samples were collected from children aged between 1 and 16 from outpatient clinics. Total copper concentrations are stable throughout this age group with a single reference range quoted in the SAS Trace Element Centres handbook. 11 An assumption was made that free copper behaves in a similar manner. Blood samples were obtained as part of routine clinical care, and the results were anonymized. Medical notes and clinical details were assessed to exclude samples from patients with potential copper abnormalities. Patients with acute phase conditions such as infections or chronic conditions known to potentially affect copper absorption such as cystic fibrosis were excluded. In total, samples from 85 subjects were used to generate the reference intervals, 50 were male and 35 were female.
Validation
Copper contamination analysis
Copper contamination of devices was assessed through the measurement of copper by ICP-MS before and after distilled and de-ionized water had been centrifuged through the ultrafiltration device.
Ultrafiltrate limit of detection
Detection limit was achieved through the analysis of blank samples of DDW (n = 10). The values were averaged, and the detection limit was calculated as three times the standard deviation.
Copper recovery and linearity
Copper recovery and linearity were assessed through measurement of the calibration standards after centrifugation through the ultrafiltration device, and then copper analysis was performed using the ICP-MS.
Accuracy
Accuracy was measured through the use of IQC materials. Initially, all the IQC materials were analysed once the calibration curve had been performed. Throughout each batch of samples analysed, IQC materials were then analysed before, in-between and at the end of analysis of 10 samples for both total and direct free copper were measured.
Precision
Precision for direct free copper was assessed through the measurement of IQC material 10 times on 7 separate occasions. For direct free copper analysis, precision was also assessed by the analysis of spiked foetal bovine serum over five separate occasions.
Functional sensitivity
Functional sensitivity, defined as an inter-assay coefficient of variation (CV) of 20% that is consistently above the analytical detection limit, was assessed by analysing spiked plasma after ultrafiltration at two different concentrations: 0.080 µmol/L and 0.200 µmol/L. The spiked plasmas were analysed eight times over four occasions. The copper result at a CV 20% would be the functional sensitivity of the assay.
Statistical analysis
Statistical analysis was performed using Analyse-it (Analyse-it Software Ltd 2009) for Microsoft Excel 2007 (Microsoft, Redmond, WA). Data normality tests were performed using the Anderson-Darling method for total copper, direct free copper and direct free copper as a percentage of total copper values.
Reference interval determination
The reference interval for direct free copper was determined using log parametric Anderson-Darling normality tests. The reference interval for direct free copper as a percentage of total copper was determined using log parametric Anderson-Darling normality tests after calculating the percentage of free copper from the total copper. Identification and removal of erroneous values were performed by visual inspection of the histograms, and then checked via the Dixon’s range Q test.
Results
A key concern with any analysis of trace elements is the possibility of contamination, and to address this, copper was measured in distilled de-ionized water before ultrafiltration and was then centrifuged through the ultrafiltration device and analysed again with each new batch of devices. Results showed that the devices were copper-free, with before and after values showing no significant differences.
Limit of detection for free copper was determined by the analysis of 10 blanks of distilled de-ionized water. Values were averaged with 3 × SD taken as the detection limit which was calculated at 0.007 µmol/L.
Copper recovery and linearity were assessed through centrifugation of the calibration standards through the ultrafiltration device followed by measurement of free copper via ICP-MS. Figure 1 demonstrates the standard calibration curve obtained for free copper.
Standard calibration curve for free copper, measured values of known standards versus copper recovery.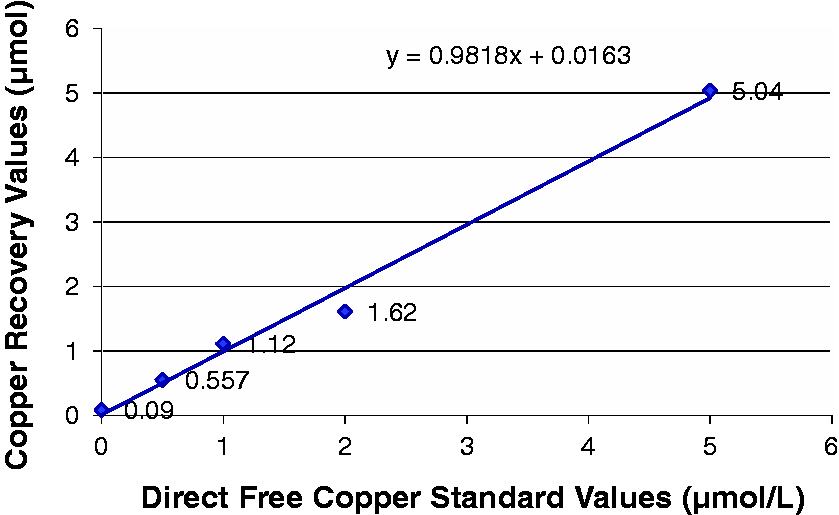
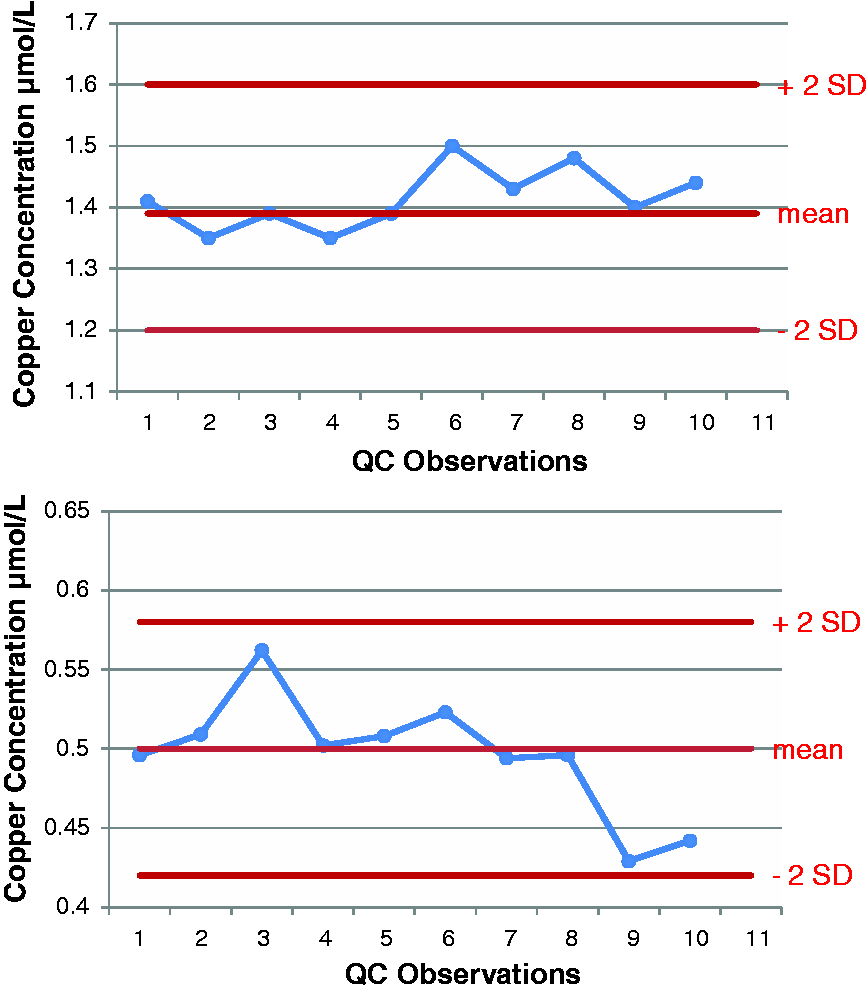
Accuracy was assessed by the repeated analysis of IQC samples of known concentration with results plotted on Levy Jennings plots for both total copper and free copper. Figures 2 and 3 show Levy Jennings plots for two levels of IQC for each analyte. Total imprecision, as expressed by the co-efficient of variation, was 7% at 0.5 µmol/L, 5% at 1.39 µmol/L and 7% at 2.22 µmol/L. The functional sensitivity of the free copper assay was 0.12 µmol/L.
Levy Jennings plots for two levels of QC for total copper ICP-MS method. Levy Jennings plots for two levels of QC for free copper ICP-MS method.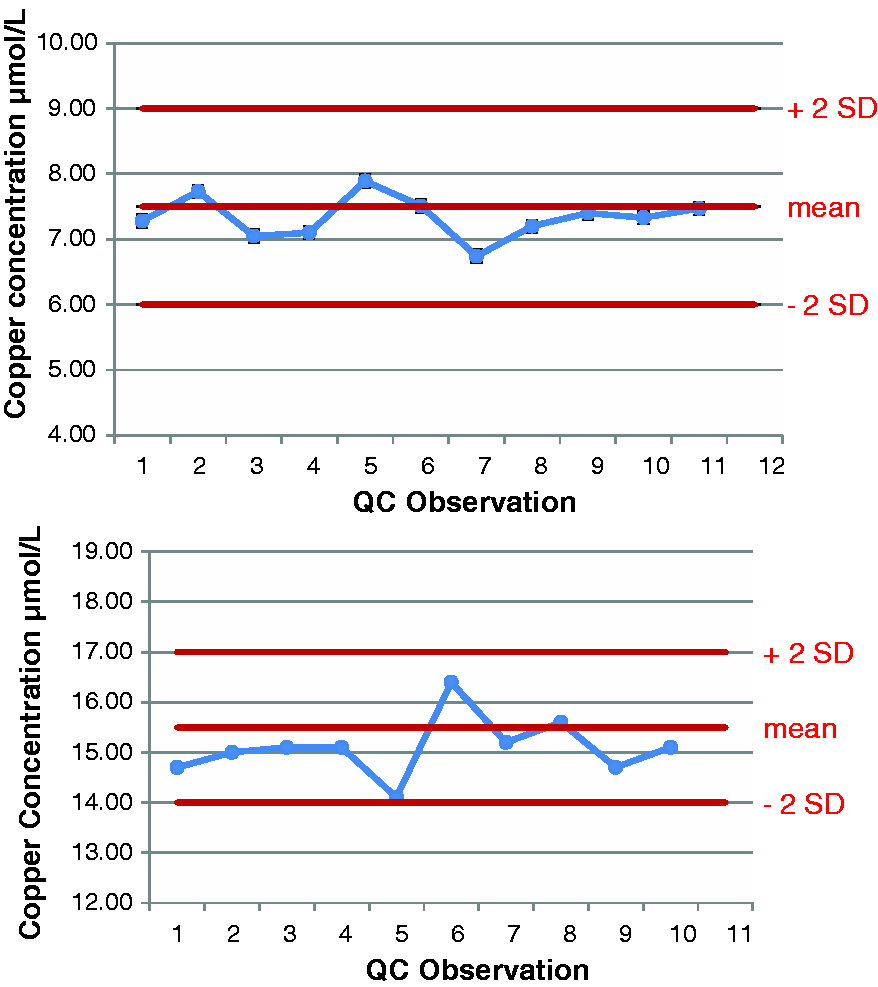
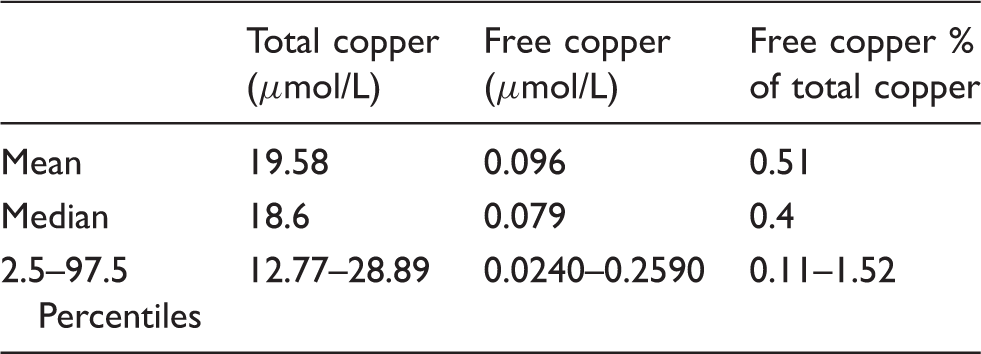
Reference interval data for total copper, free copper and free copper as % of total.
Anderson-Darling normal probability plot for total copper (µmol/L).
Anderson-Darling normal probability plot for free copper as a percentage of total copper (%).
Discussion
We have described and validated a method for determining free copper in plasma samples using ultrafiltration and ICP-MS. Results show that the method is accurate and precise with an appropriate linear working range. The method CV was shown to be between 5 and 7% depending upon free copper concentration. We have used this method to generate a reference interval for directly measured free copper in a paediatric population.
There is currently an unmet clinical need for a biomarker for monitoring patients with WD that is accurate and easy to use in a paediatric population. There exist significant issues with caeruloplasmin as a biomarker for the diagnosis of WD, and it has no role in disease monitoring. Free copper is a useful biomarker in the management of people with WD, and this is traditionally obtained by measuring copper from 24-h urine collections. In a paediatric population, these are notoriously inaccurate.
Free copper has several practical advantages over existing tests, as well as exchangeable copper methods, for monitoring known cases of WD. Exchangeable copper methods described thus far require pretreatment with EDTA within a short period postvenesection. 12 The free copper method here requires no such preanalytical preparation and can be performed on simple, routine plasma samples. Additionally, disease activity and treatment response in WD are commonly monitored with 24-h urinary collections for copper. Free copper represents a far simpler monitoring method than 24-h urine collection.
In conclusion, we describe the use and validation of an ultrafiltration ICP-MS method for plasma free copper with the first published paediatric reference interval. Free copper could provide much needed assistance for the monitoring of WD in children and also for adults. Further studies are required to better characterize the clinical utility of this test in cases of proven disease.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
PC.
Contributorship
PC devised the research question, planned the experiments, supervised the project and assisted with writing the paper. DW carried out the experiments and statistical analysis. PW wrote the manuscript and assisted with statistical analysis and interpretation of the data.
