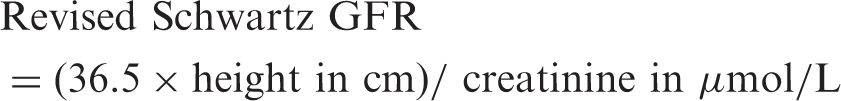Abstract
Background
The use of serum creatinine equations for estimating glomerular filtration rate is well known in adults and children. We evaluated the revised Schwartz creatinine-based estimated glomerular filtration rate prediction equation in Black African children in KwaZulu-Natal, South Africa.
Methods
Review of medical records of all Black African patients aged 2–18 years old who have had glomerular filtration rate determined by intravenous Technetium-99 m-diethylene-triamine-pentaacetate, for the period 1 January 2010 to 31 December 2014 at the Inkosi Albert Luthuli Central Hospital, Durban, South Africa was performed. Estimated glomerular filtration rate result obtained using the revised Schwartz equation was compared to Technetium-99 m-diethylene-triamine-pentaacetate plasma clearance measured glomerular filtration rate. Accuracy of the estimated glomerular filtration rate equations within 10% (P10) and 30% (P30) of the measured glomerular filtration rate, sensitivity and specificity for predicting glomerular filtration rate < 60 mL/min/1.73 m2 and < 30 mL/min/1.73 m2 was determined.
Results
Results from 148 African children between 2 and 18 years old were analysed. P10 and P30 values were 16 and 49%, respectively. Sensitivity of 92.9% (95% CI: 80.5–85), specificity of 95.3 (95% CI: 89.3–98.5) and AUC of 0.96 (95% CI 0.92–0.99) were obtained for measured estimated glomerular filtration rate < 60 mL/min/1.73 m2. Sensitivity of 88.2% (95% CI: 63.6–98.5), specificity of 90.8 (95% CI: 84.5–95.2) and area under the curve of 0.93 (95% CI 0.88–0.96) were obtained for measured estimated glomerular filtration rate < 30 mL/min/1.73 m2.
Conclusions
The revised Schwartz equation did not meet the National Kidney Foundation Kidney Disease Outcomes Quality Initiative guidelines of 90% of estimated glomerular filtration rate results within 30% of measured glomerular filtration rate.
Introduction
Several published equations for estimated glomerular filtration rate (eGFR) in paediatric populations exist; however, most apart from the revised Schwartz were not developed using an isotope dilution mass spectrometry (IDMS) traceable creatinine assay. 1 There is a paucity of data on the use of paediatric eGFR equations on the general healthy population globally as well as in the local Black African population in South Africa with kidney disease. The aim of this study was to determine if the use of the revised Schwartz equation would be valid in our local Black African paediatric population.
Materials and methods
The study was performed at the Inkosi Albert Luthuli Central Hospital, an academic hospital servicing the province of KwaZulu-Natal in South Africa.
Medical records were reviewed from the hospital information system and laboratory information system.
Clinical history, age, gender height, weight and serum creatinine (S-Cr) results (measured within 24 h prior to the measured GFR) for patients <18 years old were obtained for the period from 1 January 2010 to 31 December 2014.
The Biomedical Research Ethics Committee (BREC) of the University of KwaZulu-Natal (Clearance certificate number BE425/15) approved this study, in accordance with the Declaration of Helsinki.
GFR measurement
Measured GFR (mGFR) was performed at the Nuclear Medicine Department using Sodium Technetium-99 m-diethylene-triamine-pentaacetate (99mTc-DTPA). During the period of the study, there were no changes to the formulation used. 99mTc-DTPA clearance is based on the estimation of the plasma clearance of 99mTc-DTPA. Dose is calculated according to patient body weight. A dose vial is prepared for each patient. A standard is prepared and measured under the same conditions as the injected dose. All doses are measured by activity in the dose calibrator. Background reading on the calibrator is noted and subtracted. Samples are taken at 2 and 3 h post injection. The plasma samples are counted with appropriate standards and blanks for background in a well counter. The slope intercept method with correction for body surface area (using the Dubois and DuBois method) was used to determine the GFR. We compared mGFR to published age related normative values. 2
Creatinine measurement
Serum samples were analysed for creatinine using the modified Jaffe kinetic method on the Siemens Advia 1800 analyser (Siemens Healthcare Diagnostics, Tarrytown, USA), which is traceable to an IDMS reference creatinine method (laboratory bias= −3.6% total CV=4.1% derived from proficiency testing data during period of study). During this time, there were no changes to the methodology. The paediatric creatinine reference ranges were based on a publication. 3
Estimation of GFR
The revised Schwartz equation was used to estimate GFR using the equation
Statistical analysis
Bias plots were used for method comparison studies. Mean percentage bias, SD of the difference and 95% limits of agreement were calculated. Wilcoxon matched pairs signed ranks test was used to test for significance of bias compared to reference mGFR. Accuracy within 10% (P10) and accuracy within 30% (P30) were also calculated. Receiver operating characteristic (ROC) curve analysis was used to calculate the sensitivity and specificity to correctly predict eGFR <60 and <30 mL/min/1.73 m2. Statistical analysis was performed using the MedCalc Statistical program (MedCalc Version 11.6.1, Mariakerke, Belgium). A p value of <0.05 was considered significant.
Results
A total of 148 children and adolescents of African ancestry were included in this study. Fifty one per cent were male and median age was 12 years (IQR: 5–15). The indication for GFR measurement for the majority of the patients was related to dose adjustment for therapeutic drugs (n = 87), with 36 (24%) having a haematological malignancy, 51 (34%) solid tumour, 43 (29%) CKD (Stage 1:1 [2.3%], Stage 2:5 [11.6%], Stage 3:20 [46.5%], Stage 4:12 [27.9%], Stage 5:5 [11.6%]), 14 (9%) Type 1 diabetes mellitus and 4 (3%) other pathologies.
Results of S-Cr showed 43% to have concentrations within the relevant age-specific reference ranges, with 20% below and 37% above the reference range. Forty nine per cent showed an mGFR below the age-related normative values.
mGFR ranged from 7 to 244 mL/min/1.73 m2. Forty-three patients (29%) had mGFR < 60 mL/min/1.73 m2. The median mGFR was 100 mL/min/1.73 m2 (IQR: 46–143.5). The median mGFR for the n = 87 subgroup of children undergoing GFR for drug dosing purposes was 156 mL/min/1.73 m2. The median eGFR result was 90 mL/min/1.73 m2 (QR: 37.0–126.5).
The SD of the difference was 48 mL/min/1.73 m2 for the revised Schwartz equation. Following analysis using the Wilcoxon matched pairs ranks test, the revised Schwartz showed a statistical significant difference from mGFR (p < 0.0001). P30 was 49% and P10 was 16 %. Refer to Figure 1 for percentage difference plot of the revised Schwartz equation against mGFR.
Percentage difference plot: mGFR versus eGFR. eGFR: estimated glomerular filtration rate; mGFR: measured glomerular filtration rate.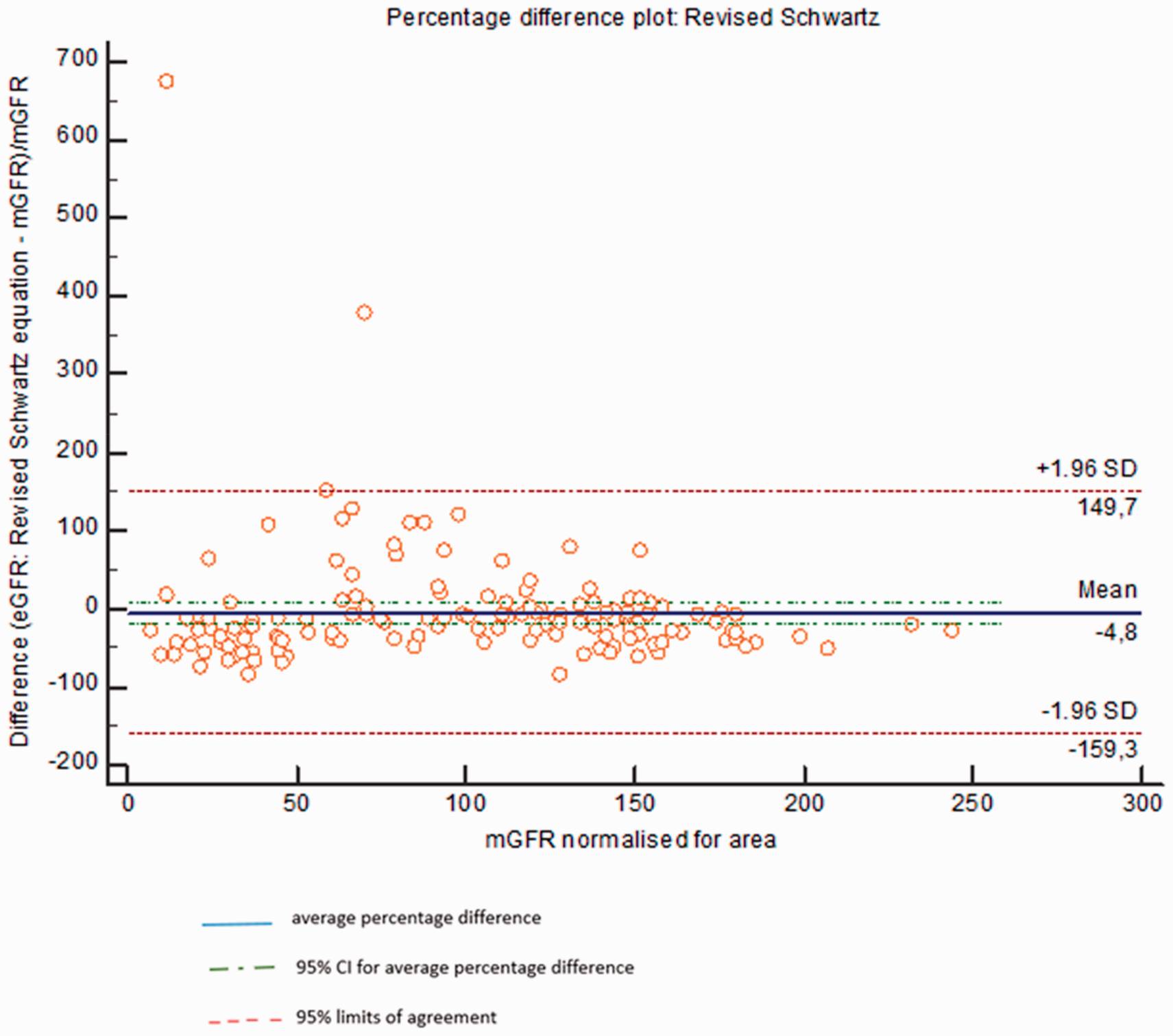
The revised Schwartz equation underestimated mGFR by 4.8% (95% CI: −17.5 to 8%). ROC curve analysis was used to calculate the sensitivity, specificity and area under the curve (AUC) in the prediction of mGFR <60, and <30 mL/min/1.73 m2. Sensitivity of 92.9% (95% CI: 80.5–85), specificity of 95.3 (95% CI: 89.3–98.5) and AUC of 0.96 (95% CI 0.92–0.99) were obtained for mGFR<60 mL/min/1.73 m2. Sensitivity of 88.2% (95% CI: 63.6–98.5), specificity of 90.8 (95% CI: 84.5–95.2) and AUC of 0.93 (95% CI 0.88–0.96) were obtained for mGFR<30 mL/min/1.73 m2.
Discussion
We report on the performance of the revised Schwartz eGFR for estimating paediatric GFR using an IDMS traceable S-Cr.
The revised Schwartz equation was able to differentiate diseased from non-diseased patients at a GFR of 60 mL/min/1.73 m2 with an AUC of 0.96 (95% CI 0.92–0.99). In this study the revised Schwartz equation underestimated GFR compared to mGFR. An enzymatic assay was used to derive the revised Schwartz equation, whilst we used a modified Jaffe creatinine assay in our lab. The enzymatic creatinine assays have decreased interference by non-creatinine chromogens, thus a lower value of creatinine may be obtained compared to the Jaffe assays. As per instructions for use, all results obtained for creatinine on the Siemens Advia 1800® are subjected to automatic correction by subtracting 26.5 µmol/L from each result, to correct for non-specific serum/plasma protein interactions. In young children however, where serum total protein is generally lower than in adults, the magnitude of the influence of the non-specific protein reactions may be regarded as negligible, thus an underestimation of creatinine can occur.
Jaffe assays are known to be imprecise at lower concentrations which could lead to erroneous results. In addition to the methodology employed, the assay’s coefficient of variation also directly impacts on that of the eGFR equation. The revised Schwartz equation was derived in a multiracial study population with CKD. In contrast, our study had an African only population with varying degrees of kidney function but with the majority having normal S-Cr and mGFR values. These issues highlight the shortcomings of applying an equation to a population which may not be truly representative, or the application of an equation by a laboratory in which the creatinine method employed is different to that which the equation was derived and validated. It has thus been advised that laboratories perform studies on their own populations using the creatinine assays which will ordinarily be used to determine the applicability of the various equations to their population. 4 To our knowledge, this has not been evaluated in our local paediatric population. Van Deventer et al. 5 have evaluated the eGFR equations in black South African adults.
We calculated bias as an expression of systematic error in eGFR (mean differences between estimated and measured GFR). The evaluated equation did not reach the 2002 KDOQI benchmark of P30 greater than 90%. 6 Although this guideline is for adults and no guideline is available for children. This highlights the fact that while standardization of creatinine assays has reduced bias due to differences in creatinine assays, it has not eliminated it. Limitations of the study include: (1) creatinine method used; (2) limitations of the use of Tc DTPA as a reference GFR method such as extrarenal elimination with subsequent overestimation of mGFR and its potential for dissociation of 99mTc from DTPA and binding to plasma proteins, leading to underestimation in GFR 7 ; (3) small sample size; (4) only one geographical site, in one race group; (5) no biopsy-proven data and (6) other parameters of physiological interest as an indicator of kidney function (e.g. albumin excretion) were not evaluated.
Strengths of our study are as follows: (1) patients with a variety of pathologies were studied and (2) patients with a wide range of GFR values were evaluated.
Conclusions
We recommend more studies using IDMS traceable enzymatic creatinine assays, encompassing the diverse racial populations of South Africa, as well as varying stages of kidney function be performed in order to validate local use of this equation.
Footnotes
Acknowledgements
We would like to thank Dr John C Stanfliet, Dr Magdalena J Turzyniecka and Dr Ravindra Sirkar for their assistance and guidance in this research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
University of KwaZulu-Natal BREC reference number BE 425/15.
Guarantor
VG.
Contributorship
YR and VG researched literature, conceived the study, was involved in protocol development, gaining ethical approval, data analysis and wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.