Abstract
Background
The insulin tolerance test is the gold standard for diagnosis of cortisol insufficiency. However, it is cumbersome, invasive, requires supervised hospital facilities and has unpleasant side-effects. A non-invasive outpatient-based test will be useful. We hypothesized that free cortisol concentrations in multiple spot urine samples can be used to diagnose cortisol insufficiency in patients with normal renal function (eGFR > 60 mL/min).
Method
Patients and controls provided urine samples at bedtime (S1), and first (S2) and second (S3) void the next day. Cortisol and creatinine were measured in all three samples, and cortisol:creatinine ratio (S1, S2 and S3) was used for further analysis. The sum of S1 + S2 + S3 was used to calculate total cortisol secretion (T). Variation (V) in cortisol secretion in response to circadian rhythm was calculated as the modulus of the difference between S1 and S2 and S2 and S3.
Results
Samples were collected from 96 controls and 11 patients. S1 was significantly lower vs. S2 and S3 in controls (P < 0.0001) but not in patients. S2, S3, T and V were significantly lower in patients vs. controls (P < 0.0001). ROC curve analysis using insulin tolerance test as gold standard showed that S2, S3, T and V were all equally accurate diagnostic markers for cortisol insufficiency (AUC: 0.87, NPV: 100%). The best balance of sensitivity and specificity was achieved using T (sensitivity: 100%, specificity: 58%).
Conclusion
Multiple spot urine samples test is an accurate, relatively inexpensive, non-invasive, convenient outpatient-based screening test for exclusion of cortisol insufficiency.
Background
The insulin tolerance test (ITT) is the gold standard for the diagnosis of cortisol insufficiency (CI).1,2 Insulin-induced hypoglycaemia stimulates adrenocorticotropic hormone (ACTH) and cortisol secretion in patients with intact hypothalamo–pituitary–adrenal function and sufficient cortisol reserves. CI, either due to ACTH deficiency or primary adrenal deficiency, results in a failure to mount an adequate cortisol response to ITT.1,2
The ITT needs to be conducted under medical supervision in hospital facilities, results in unpleasant neuroglycopaenic symptoms and is contraindicated in children and in patients with epilepsy or heart disease. The short synacthen test (SST) is the most widely used test for adrenal insufficiency, is a safer alternative, but requires hospital admission and does not test the integrity of the entire hypothalamo–pituitary–adrenal axis. 2 A negative result following an SST may require confirmation using an ITT in some cases. 2 The metyrapone, glucagon and arginine tests advocated as safer alternatives are also invasive and require hospital admission. 3 Therefore, a reliable non-invasive outpatient-based screening test for CI would be of great clinical value.
Cortisol secretion follows a circadian rhythm. Circulating cortisol concentrations are lowest at midnight and peak on awakening in the morning. 3 CI results in a decrease in total cortisol secretion and in a blunting of both early morning cortisol peak and diurnal variation. 4
Urinary free cortisol (UFC) is a good surrogate for circulating cortisol levels and is routinely measured as a diagnostic marker of cortisol excess. 5 We therefore proposed the hypothesis that UFC could also be used to exclude CI. 6
This study assessed the utility of measuring UFC in a multiple timed spot urine cortisol test (MTSUCT) to detect a decreased cortisol secretion and a blunting of diurnal variation and thus to exclude CI. 6
Method
Ethical approval for the study was obtained, and participants were recruited following informed consent. Hospital staff, friends and family were given information about the study and were invited to participate in the control arm provided they met the inclusion criteria (>18 years of age and no history of renal or other underlying disease). The patient arm of the study included patients with ITT (done as part of routine clinical investigations) confirmed CI. Both controls and patients provided three timed spot urine samples: S1 (bedtime day 1), S2 (first void on waking the next day – day 2) and S3 (second void – day 2). Patients on hydrocortisone replacement were advised to take the last dose of hydrocortisone no later than 4 pm on day 1 and to delay first dose on day 2 to after collection of S3.
Urine samples received in the laboratory by post (in crash-proof packaging provided) were aliquoted and stored at −20℃ until further analysis. Urinary cortisol was measured by a routine laboratory method (Roche Elecsys 2010 electrochemiluminescence immunoassay). Briefly, cortisol from 600 µL of sample was extracted manually using 3 mL of dichloromethane. 7 Once mixed, the sample was centrifuged and the top layer discarded. The organic layer (1.5 mL) was dried and reconstituted and incubated with 300 µL of Roche Universal Diluent at room temperature for 30 min before being analysed on the Roche Elecsys 2010 cortisol assay ([recovery: 99%; range: 94.2–104.3%]; [inter-assay imprecision (coefficient of variation (%CV)) reported by the manufacturer, K021218 Elecsys®, ranged from 1.8% to 3.2% across the analytical range of 1–1758 nmol/L]). Urinary creatinine was measured on the Roche/Hitachi Cobas C system using an enzymatic method. Results were expressed as cortisol:creatinine ratio (Ccr) in order to correct for variation in urine concentration. Ccr in samples S1, S2 and S3 is denoted hereafter as S1, S2 and S3. The sum (T) of S1 + S2 + S3 was used to estimate the total cortisol secretion. The diurnal variation (V) in cortisol concentration (and hence secretion) between each consecutive sampling time was estimated by calculating the sum of the modulus of the difference between S1 and S2 and S2 and S3.
Gaussian distribution was not assumed; therefore, non-parametric statistics (Mann Whitney U test) were used to compare groups. Receiver-operator curve (ROC) analysis was used to assess the utility of S1, S2, S3, T and V as diagnostic marker(s) of CI.
Results
Samples from 96 controls (45 female [age (years): range: 20–84; median: 51; number > 50 = 25), 51 male [age (years): range 19–81; median: 29; number > 50 = 17]) and 11 patients (4 female [age (years): range: 51–79; median: 67] and 7 male [age (years): range: 30–74; median: 50]) were available for analysis.
S1, S2 and S3 were significantly different in healthy controls (P < 0.0001) but not in patients (P = 0.36). While there was no significant difference between S1 in patients vs. controls (P = 0.19), S2 and S3 were significantly lower (P < 0.0001). Both T and V were significantly lower in patients vs. controls (P < 0.0001) (Figure 1).
Scatter plot of S1, S2, S3, T and V in controls and patients. Patient values are denoted as S1p, S2p, S3p, Tp and Vp.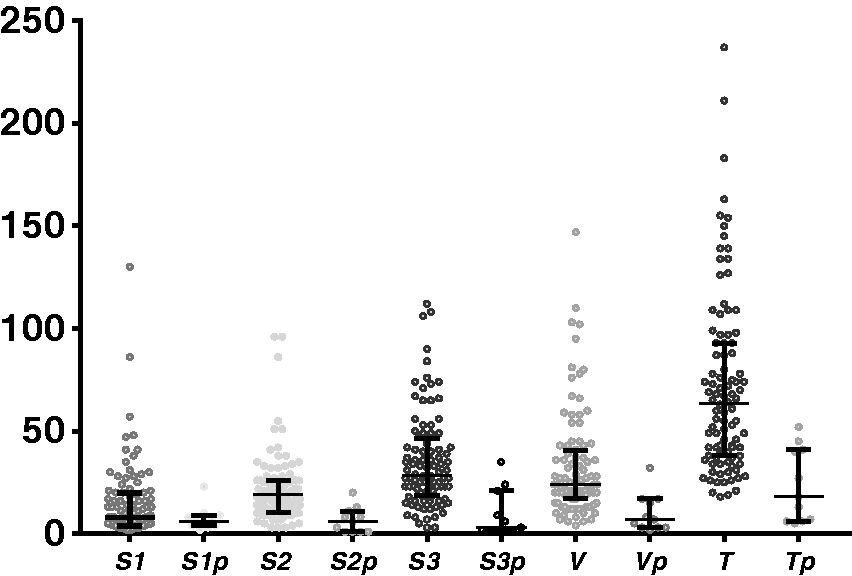
No significant gender differences were noted. S2 (P < 0.05) was significantly lower in both male and female controls aged over 50 years vs. controls aged below 50 years. Male controls aged over 50 years also had significantly lower S3 (P < 0.05) compared with males aged below 50 years and female controls aged over 50 years had significantly lower T compared with females aged below 50 years (P = 0.002).
ROC curve analysis using ITT results as the reference standard showed that there was no significant difference in the area under the curve (AUC) obtained for S2, S3, T or V, all of which provided a high diagnostic accuracy for diagnosis of CI (V [AUC: 0.87; 95% confidence interval (CI): 0.75–0.98], S3 [AUC: 0.87; CI: 0.75–0.99], S2 [AUC: 0.87; CI: 0.76–0.97], T [AUC: 0.87; CI: 0.77–0.97], S1 [AUC: 0.62; CI: 0.48–0.77]). However, the best balance of sensitivity and specificity was obtained for T with sensitivity of 100%, specificity 58%, positive predictive value 20% and negative predictive value (NPV) 100% at a diagnostic cut-off T < 53 nmol/mmoL of creatinine.
Discussion
CI results in a decrease in total (T) and early morning peak cortisol (S2, S3) and in a blunting of response to circadian rhythm/sleep as a physiological stimulus (V). 5 We have reliably demonstrated all three using non-invasive MTSUCT.
ITT was used as the reference standard in our ROC analysis. Our results suggest very good concordance of MTSUCT with ITT. Any discrepancies between the tests may be because ITT is by far the most potent stimulus for cortisol secretion. 8 It is possible that subtle deficiencies resulting in blunted cortisol response to physiological stimulus (such as sleep or emotional stress), which remain undiagnosed by ITT, are unmasked by the MTSUCT.
Urine cortisol secretion decreases in older patients (as seen in our study) and in patients with renal impairment. The specificity of MTSUCT is decreased in these patients, due to an increase in false positives. As the decrease in cortisol secretion will not cause an increase in false negatives, the sensitivity is likely to remain unaffected. A missed diagnosis is unlikely given the high NPV of this test. A larger study to improve specificity by (i) assessing the effect of renal impairment on MTSUCT and (ii) establishing the utility of age-related diagnostic cut-offs will be useful.
Conclusion
Our study suggests that MTSUCT is an accurate, relatively inexpensive outpatient screening test that is able to rule out suspected CI. It not only eliminates the cost of hospital staff time and day unit beds but is also more beneficial for both the workforce and the workplace in general, by eliminating the need for time off work for hospital investigations. T offers the best balance of sensitivity and specificity (sensitivity: 100%, specificity: 58%). However, T, V, S2 and S3 are all equally accurate in diagnosing CI (AUC: 0.87, NPV: 100%), and using these markers in conjunction can offer further diagnostic assurance, just as both basal and peak cortisol concentrations are used to improve overall diagnostic accuracy in ITT.
Footnotes
Acknowledgement
The authors thank Viapath pathology services for providing the laboratory facilities. Most importantly, they thank the volunteers for their help without which this study would not have been possible.
Competing interests
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Viapath Innovation Grant awarded to Radha Ramachandran and Arun Sankaralingam.
Ethical approval
REC reference: 13/ES/0100; IRAS project ID; 131806; East of Scotland Research Ethics Service.
Guarantor
AW.
Contributorship
RR, JAZ and ASW: study design, data analysis and manuscript preparation; SAK, MT, CF, MS, NM: sample and data collection; AS: sample assays and analysis; PVC, BM: patient access and help with study design.
