Abstract
Cortisol is a steroid hormone produced in response to stress. It is essential for maintaining health and wellbeing and leads to significant morbidity when deficient or present in excess. It is lipophilic and is transported bound to cortisol-binding globulin (CBG) and albumin; a small fraction (∼10%) of total serum cortisol is unbound and biologically active. Serum cortisol assays measure total cortisol and their results can be misleading in patients with altered serum protein concentrations. Automated immunoassays are used to measure cortisol but lack specificity and show significant inter-assay differences. Liquid chromatography – tandem mass spectrometry (LC-MS/MS) offers improved specificity and sensitivity; however, cortisol cut-offs used in the short Synacthen and Dexamethasone suppression tests are yet to be validated for these assays. Urine free cortisol is used to screen for Cushing’s syndrome. Unbound cortisol is excreted unchanged in the urine and 24-h urine free cortisol correlates well with mean serum-free cortisol in conditions of cortisol excess. Urine free cortisol is measured predominantly by immunoassay or LC-MS/MS. Salivary cortisol also reflects changes in unbound serum cortisol and offers a reliable alternative to measuring free cortisol in serum. LC-MS/MS is the method of choice for measuring salivary cortisol; however, its use is limited by the lack of a single, validated reference range and poorly standardized assays. This review examines the methods available for measuring cortisol in serum, urine and saliva, explores cortisol in disease and considers the difficulties of measuring cortisol in acutely unwell patients and in neonates.
Keywords
Introduction
Cortisol
Cortisol is a steroid hormone produced by the zona fasciculata of the adrenal cortex. Its secretion is directly controlled by the release of adrenocorticotrophin (ACTH) from the anterior pituitary which is regulated by corticotrophin-releasing hormone (CRH), and less directly, arginine vasopressin (AVP), secreted by the hypothalamus. Cortisol exerts negative feedback on both ACTH and CRH secretion, while stress acts directly on the hypothalamus to stimulate CRH release.
ACTH secretion is pulsatile and follows a circadian pattern in line with CRH. An unstressed adult produces 18–25 pulses in a 24-h period, starting with high-amplitude pulses released at roughly 90-min intervals between 3 and 9 a.m., which peak between 7 and 11 a.m. and reduce in frequency between 6 p.m. and midnight. Rapid blood sampling is, therefore, required to fully appreciate the dynamic nature of ACTH concentrations in blood. 1 Cortisol mirrors ACTH’s circadian rhythm and pulsatile pattern, with highest peak in the early morning and nadir between midnight and 3 am.1,2 Its rhythm is further affected by multiple factors, including sleep deprivation/shift-working, 3 ethnicity, 4 gender, age, BMI 5 and menstrual cycle phase.6,7
Cortisol is lipophilic and requires a protein carrier for transport around the body. Approximately 80% is bound to cortisol-binding globulin (CBG), 10% is albumin-bound and 10% is free (unbound) and biologically active. Cortisol is involved in energy metabolism through direct effects on the synthesis and breakdown of carbohydrates, protein and lipids. It increases gluconeogenesis by activating key enzymes, e.g. phosphoenolpyruvate carboxykinase and glucose-6-phosphatase and by increasing muscle breakdown and inhibiting amino acid uptake and protein synthesis to provide additional substrate. It reduces peripheral glucose uptake into cells, activates glycogen synthase and inhibits glycogen phosphorylase leading to increased glycogen stores. It activates lipolysis, resulting in increased circulating free fatty acids and enhances adipogenesis through activation of lipoprotein lipase and increased leptin concentration.8,9 The net result is increased blood glucose concentrations mediated by the breakdown of protein and lipids; and cortisol, along with glucagon, adrenaline and growth hormone, is released in response to hypoglycaemia.
Cortisol is involved in water and electrolyte balance and blood pressure control. It increases angiotensinogen synthesis, enhances the vasopressor effects of catecholamines and angiotensin II and reduces nitric oxide-mediated endothelial dilatation. In the kidneys, it increases glomerular filtration rate and acts on the distal renal tubule to increase sodium retention and potassium loss; it also increases sodium transport and free water loss through antagonism of AVP. 9 Its renal effects are mediated through the mineralocorticoid receptor to which it binds with equal affinity to aldosterone. The magnitude of cortisol binding to the receptor is reduced by the action of 11β-hydroxysteroid dehydrogenase type 2 (11β-HSD2) which oxidizes cortisol to cortisone, which has much weaker affinity for the receptor.
Cortisol has both anti-inflammatory and immunosuppressive effects. It reduces circulating lymphocyte counts by redistribution to the spleen, lymph nodes and bone marrow. It inhibits immunoglobulin synthesis, induces lymphocyte apoptosis and prevents inflammatory cytokine (e.g. interleukin-1, 2, 6, tumour necrosis factor [TNF], interferon-γ) production through inhibition of nuclear factor kappa B (NF-κB). Monocyte differentiation into macrophages and subsequent macrophage function are inhibited by cortisol, and local inflammation is suppressed by reduced histamine production and impaired prostaglandin synthesis.
Cortisol is also involved in maintaining healthy pregnancy and in the initiation of birth, although its exact role is still unclear; 10 further study being hindered by differences between humans and animal models. Increased placental production of CRH has been noted in the run up to human labour, resulting in increased foetal cortisol production. Raised serum cortisol and placental CRH, both believed to be manifestations of maternal stress, have been shown to increase the risk of obesity and diabetes in later life.11–13
Measuring cortisol
Acute cortisol deficiency presents clinically as an Addisonian crisis, characterized by catastrophic dehydration and salt wasting. This is potentially fatal so early diagnosis is essential. However, as mild adrenal insufficiency is difficult to detect clinically due to non-specific symptoms such as fatigue and weakness, diagnosis relies on appropriate biochemical testing. Similarly, the early symptoms of cortisol excess (Cushing’s syndrome), which include weight gain, hypertension and impaired glucose tolerance, are easily attributable to other causes, such as simple obesity, leading to delays in diagnosis.
A random serum cortisol concentration rarely gives enough information for a diagnosis of adrenal disease to be made. 14 A recent study using the Siemens Centaur assay demonstrated the extent of inter-individual variation in morning cortisol, with concentrations ranging between 96 nmol/L and 722 nmol/L in samples collected from healthy volunteers between 8.00 and 11.00 am. 15 This wide variation can be partly explained by the timing of the sample relative to a pulse of cortisol.
An early morning cortisol measurement, although rarely diagnostic, can contribute some useful information, with concentrations above 450–550 nmol/L virtually excluding hypoadrenalism, concentrations below this indicating the need for more definitive testing14,16,17 and concentrations below 140 nmol/L suggesting adrenal insufficiency, 18 although requiring confirmation (these quoted concentrations will vary according to the accuracy and bias of the assays used). Plasma ACTH can aid in the diagnosis of adrenal insufficiency; with concentrations >66 pmol/L being highly predictive for disease when combined with low early morning cortisol (<140 nmol/L) or an impaired response to Synacthen (these quoted concentrations will vary according to the accuracy and bias of the assays used). 14 The most useful tests of adrenal function, therefore, are those that stimulate or suppress the HPA axis.
Serum/plasma cortisol assays
Cortisol can be measured in either serum or plasma, 19 with direct comparison between the two showing they are interchangeable (unpublished data). The first plasma cortisol assays were restricted to research laboratories as they required several labour-intensive steps, given the low concentrations being measured and the presence in plasma of numerous other steroid hormones. In 1962, a simple fluorimetric assay utilizing the natural fluorescence of 11-hydroxycorticoids (cortisol and corticosterone) was described, marking the beginning of clinical cortisol assays. 20 However, this method was limited by poor specificity for cortisol and low sample throughput, and radioimmunoassay (RIA) soon replaced it as the method of choice. 21 Further assay development saw the elimination of preanalytical sample extraction steps and the introduction of enzyme and chemiluminescent detection,22,23 and the first automated cortisol immunoassay was described in 1992. 24 These have now become the method of choice in many clinical laboratories despite their limitations which include differences in antibody specificity and affinity for cortisol and other steroid hormones and the need to release the steroid molecule from its carrier protein before it can be measured.
Immunoassays
Endogenous and exogenous steroids
Many steroids are structurally similar to cortisol (Figure 1), which makes the generation of entirely specific antibodies difficult.
25
Cortisol immunoassays are thus hampered by varying degrees of antibody cross-reactivity with other steroids, endogenous and exogenous,
26
and can be unreliable in certain clinical settings such as congenital adrenal hyperplasia
27
(CAH) and in patients treated with synthetic glucocorticoids.
Structural homology of cortisol and various endogenous and exogenous steroid molecules.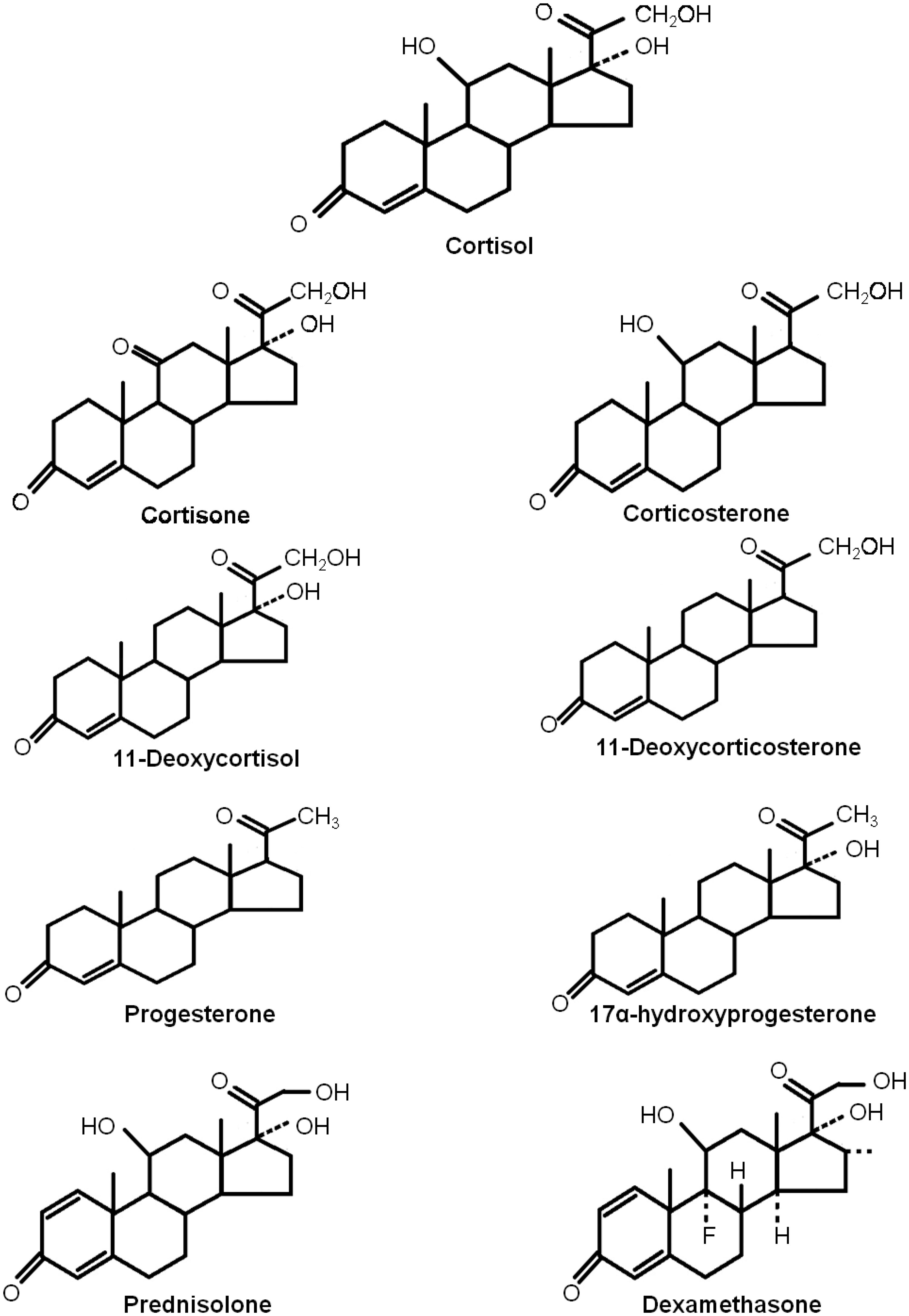
Information provided by assay manufacturers can sometimes help laboratories identify possible interference; however, this information is not exhaustive, and labs are not always aware of patients’ medication. This was highlighted by the recent implementation of a liquid chromatography-tandem mass spectrometry (LC-MS/MS) serum cortisol assay in a laboratory serving a large respiratory unit. Exogenous steroids were found in the serum of up to 50% of patients tested. 28 Synthetic steroids were also found in the urine of 5% of patients investigated for Cushing’s syndrome when an LC-MS/MS assay was used. 29
Binding proteins
Current assays measure total serum cortisol (bound and unbound fractions) and as a result are affected by changes in CBG and albumin concentration. The cortisol response to ACTH in a group of patients with the nephrotic syndrome was shown to be lower, by between 63 and 179 nmol/L, depending on the assay used, than that in healthy volunteers. 30 This translated into a 50% failure rate in interpretation of the response for two of the assays, when a 500 nmol/L cut-off was used. Similar findings were reported in patients with liver disease and mean albumin concentration of 21 g/L, with 46% of patients failing to achieve a total cortisol of 550 nmol/L post-stimulation. 31 Low protein concentration also affects the interpretation of cortisol results in acutely unwell patients.
Mean post-ACTH cortisol concentrations in non-OCP females and OCP females by assay.
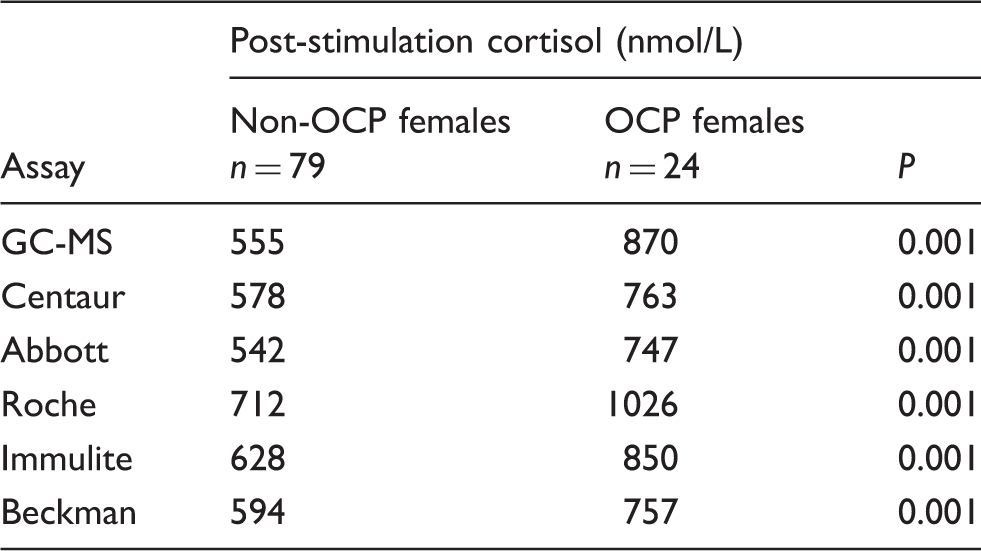
Non-OCP females: women not using an oestrogen-containing oral contraceptive pill; OCP females: women using an oestrogen-containing oral contraceptive pill.
Reproduced with permission of authors and publishers. 15
This difference can be eliminated by heat-treatment 32 and has been attributed to reduced cortisol availability for measurement due to increased protein binding. Most current immunoassays make some attempt to displace cortisol from CBG prior to measurement, using either steroid substitutes, e.g. Danazol (Roche, personal communication) or pH change (Abbott, personal communication), although these measures are not always sufficient when excess CBG is present.
Assay standardization
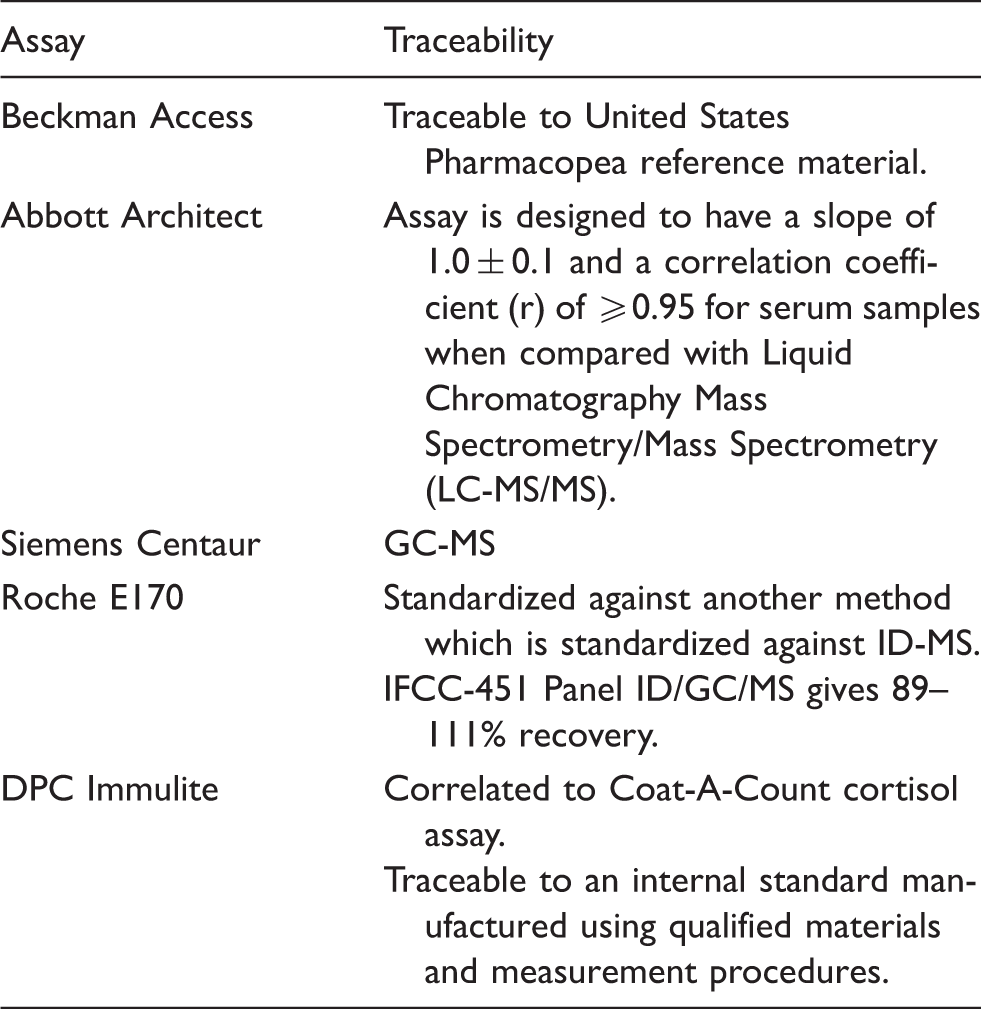
Information provided by assay manufacturers regarding the reference material and method to which each assay was traceable.
The effect of gender and sample matrix
A recent study of the effect of gender and matrix on immunoassay cortisol measurement 36 demonstrated increased inter-assay variation in patients with significant renal disease and those who are critically ill. This was presumed to be a matrix effect due to decreased cortisol clearance in the former group and its increased production and reduced breakdown in the latter. Inter-assay variation in cortisol recovery was also noted to be higher in spiked samples from female patients than in males, presumably due to differences in CBG concentrations.
Divergence in external quality assurance
The majority of UK participants in the UKNEQAS cortisol scheme measure cortisol using one of the following immunoassays: Roche Elecsys (44%), Siemens ADVIA Centaur (20%), Abbott Architect (19%) and Beckman Access (10%).37 The scheme organizers calculate a B score, or specimen percentage bias, for each laboratory to demonstrate how far its result deviates from the target concentration – the all laboratory trimmed mean (ALTM). An arbitrary acceptable limit of performance of 10% above or below the target mean has been set, with the percentage of laboratories failing to meet this target shown in Figure 2.
Proportion of laboratories participating in UKNEQAS cortisol EQA scheme with bias scores outside the acceptable limits of performance.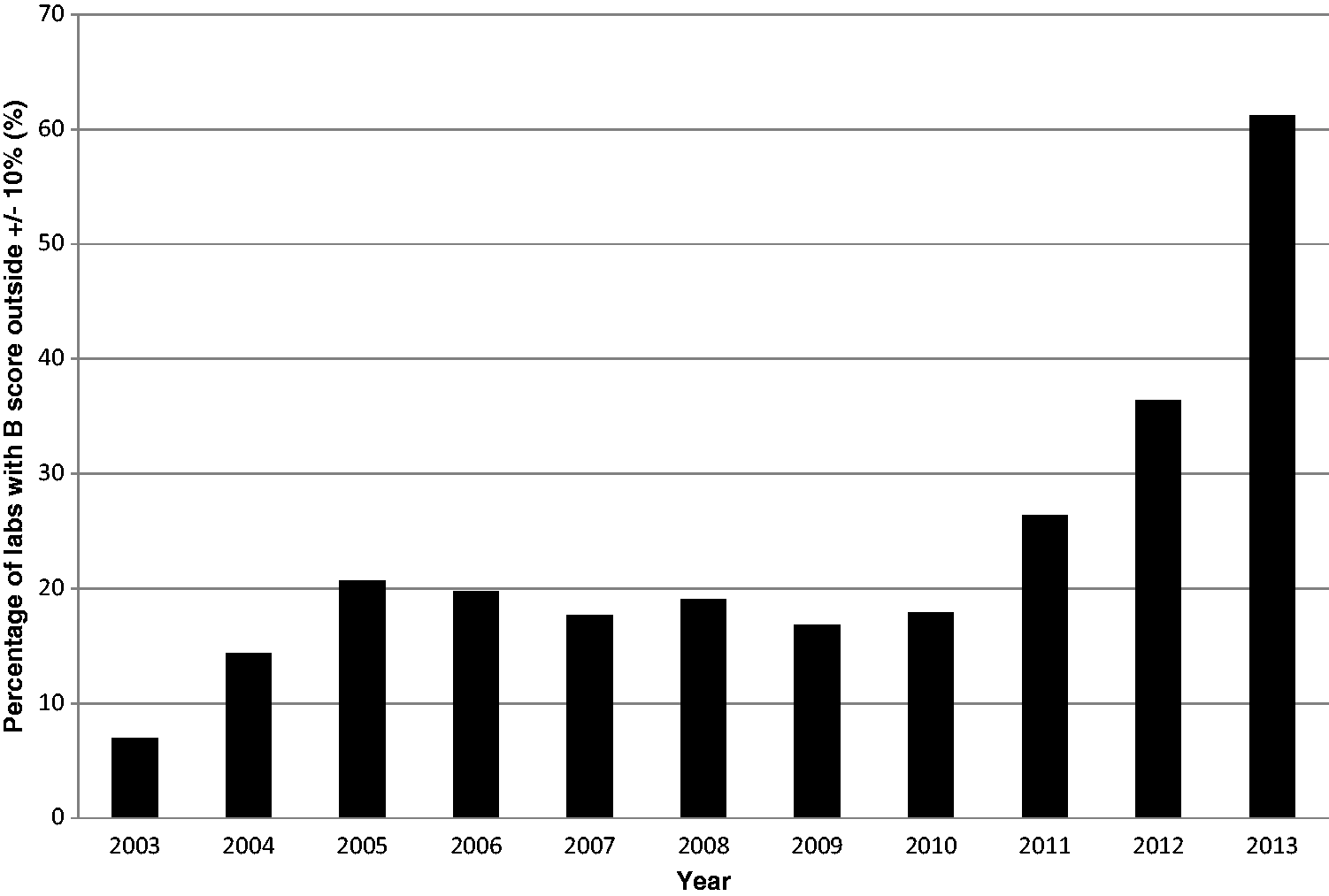
This increasing divergence between cortisol assays37–39 is unlikely to be explained by worsening laboratory performance, but indicates that the differences between current cortisol immunoassays are too great for a single, meaningful mean to be calculated. UKNEQAS plans to address this by replacing the ALTM with the LC-MS/MS method mean (the field method-mass spectrometry (FM-MS) mean) as of April 2017. 40 Occasional comparisons will also be made to an LC-MS/MS reference method (RM-MS) to ensure the validity of the FM-MS mean.
Differences between immunoassays
The significance of differences between automated cortisol immunoassays was not widely considered until a study by Clark et al. 41 Cortisol concentration pre- and post-Synacthen was measured using four widely available immunoassays. Median cortisol concentration 30 min post-Synacthen ranged from 707 nmol/L to 866 nmol/L, with a lower limit (defined as the 5th percentile) between 510 nmol/L and 626 nmol/L. This demonstrated the assay dependence of cortisol cut-offs and the risk of misdiagnosing adrenal insufficiency if the wrong cut-off was used. Nevertheless, most laboratories continued to use the fluorimetrically derived 550 nmol/L cut-off. 42
Diagnosing adrenal insufficiency
Assay-specific estimated lower reference limits for post-ACTH cortisol according to gender and OCP-status.
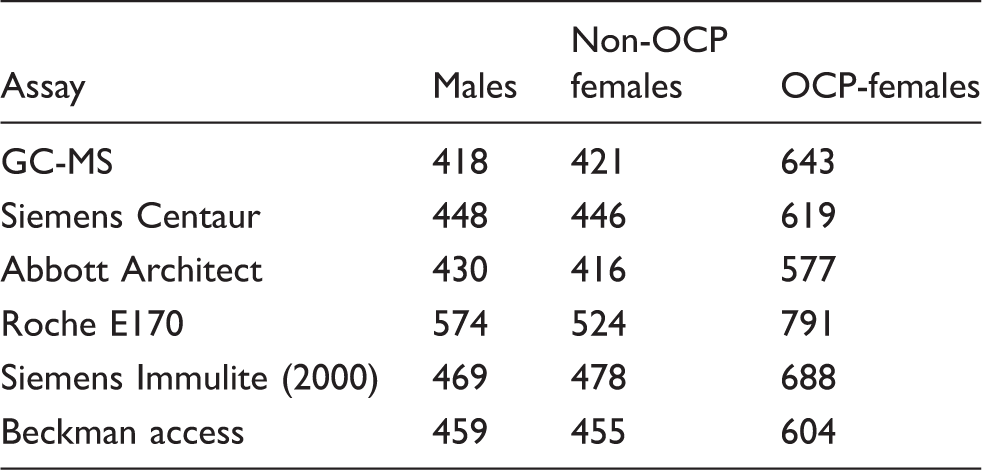
Note: The estimated lower reference limit was determined by back transformation of the 2.5th percentile value (mean – 1.96 × SD) of the log-transformed data. Results are expressed in nmol/L.
Non-OCP females: women not using an oestrogen-containing oral contraceptive pill; OCP females: women using an oestrogen-containing oral contraceptive pill; NA: not applicable.
Reproduced with permission of authors and publisher. 15
Estimated lower reference limits for post-ACTH cortisol according to gender and OCP-status for Siemens Centaur assay pre- and post-assay reformulation.

Note: The estimated lower reference limit was determined by back transformation of the 2.5th percentile value (mean: 1.96 × SD) of the log-transformed data. Results are expressed in nmol/L. The bias ratio was calculated by dividing each sample’s immunoassay cortisol concentration by the corresponding GC-MS concentration. The mean ratio for all subjects at all time points was then calculated.
Non-OCP females: women not using an oestrogen-containing oral contraceptive pill; OCP: women using an oestrogen-containing oral contraceptive pill.
Ongoing assay changes
Mean bias ratios for cortisol immunoassays compared with GCMS in 2010 and 2013.
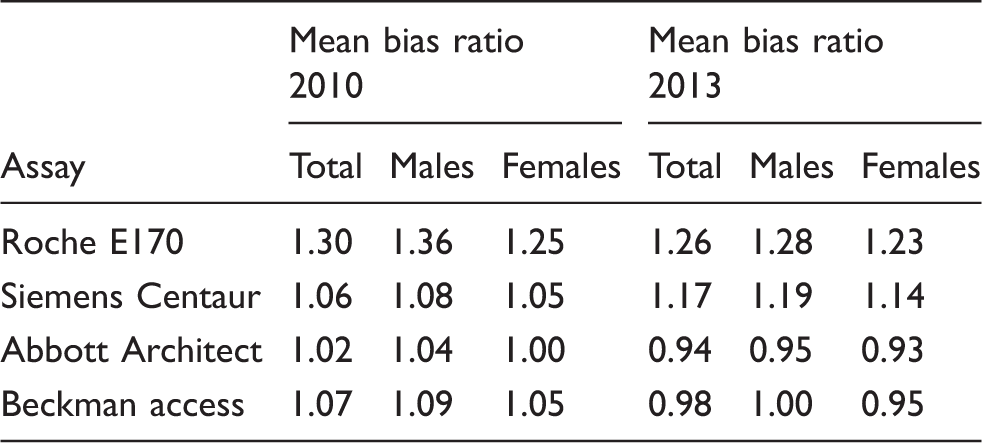
Note: The bias ratio was calculated by dividing each sample’s immunoassay cortisol concentration by the corresponding GC-MS concentration. The mean ratio was then calculated.
Total: mean bias ratio for all subjects at all time points.
Reproduced with permission of authors and publishers. 36
The recent adjustment of the Roche cortisol assay, to align its performance with LC-MS/MS cortisol, has resulted in substantially lower concentrations and will provide some indication of how well laboratories adapt to ongoing changes. Continuing with old cortisol cut-offs for the Synacthen test will result in overdiagnosis of adrenal insufficiency, but simply reducing the cut-off in line with the observed bias of the new assay will not provide a clinically validated cut-off. The best approach would, therefore, be to directly compare post-Synacthen results across the two assays, which would be easier done by the kit manufacturer or an EQA provider than by each kit user individually.
Mass methods
In 1975, the first reference method for measuring cortisol using GC-MS was described. 34 Cortisol was derivatized to its heptafluorobutyrate ester to improve assay sensitivity and tritiated cortisol was added as an internal standard to correct for losses due to steroid adsorption to the chromatography column. Today, GC-MS cortisol assays continue to outperform immunoassays in both specificity and sensitivity, but they remain confined to reference and research laboratories as they are labour intensive and require specialist equipment and highly skilled laboratory staff.
In 2001, an LC-MS/MS method for measurement of multiple steroid hormones in patients with 11 β-hydroxylase and 21-hydroxylase deficiencies and Addison’s disease was described. 43 This combined the improved specificity and definitive analyte identification of mass spectrometry with the ability to perform multiple tests simultaneously in a cost-effective way and offered the potential for mass methods to spread to clinical laboratories. In 2004, an LC-MS/MS reference method, which avoided the need for preanalytical sample derivatization was described as an alternative to GC-MS for routine method comparisons, 35 and in 2016, an LC-MS/MS candidate reference measurement procedure that could be used to assign traceable target concentrations to EQA samples was described. 44
Currently, there are 12 LC-MS/MS cortisol assays registered with the UKNEQAS cortisol scheme 40 and tandem-mass spectrometry is being used globally for routine steroid hormone measurement, either in isolation 28 or as part of a panel of related molecules.45–50 However, due to the greater specificity of LC-MS/MS, the cortisol response to ACTH stimulation and dexamethasone suppression may need to be redefined to ensure correct interpretation. Concerted efforts to standardize LC-MS/MS assays, including traceability to a single reference method and material, and rigorous validation 51 of these assays are also needed to avoid similar inter-assay differences to those affecting immunoassays.
Such differences have already been reported with LC-MS/MS vitamin D and testosterone assays, due to the lack of common calibrators, 52 differences in sample extraction techniques and differences in the type of LC column and/or mass spectrometer used. 53 There have been no direct comparisons between cortisol LC-MS/MS assays as yet, although UKNEQAS data from the past year shows that the variability between these assays (CVs: 3.1–15.8%) is less than differences between methods (CVs: 11.6–24.9%) and not too dissimilar to the performance of a single immunoassay. 54
Sample matrix has also been shown to affect the reproducibility and accuracy of LC-MS/MS assays. Endogenous impurities, e.g. salts (typically in urine) and co-eluting substances, e.g. analyte metabolites, can cause ion suppression or enhancement – a reduction or increase in the efficiency of ion formation.55–57 Careful evaluation of this effect is, therefore, essential before a new LC-MS/MS cortisol assay can be introduced into clinical practice. Careful selection of the internal standard is also necessary, with stable isotopically labelled analogues preferred over structurally similar, but non-identical, analogues 58 and other compounds which have the same retention time but a different structure. 59 The use of isotopically labelled standards ensures co-elution of analyte and internal standard and their subsequent ionization under identical conditions, 59 thus improving analytical accuracy.
Exogenous interferences in LC-MS/MS cortisol assays are few and recent developments have managed to eliminate the interference from prednisolone and fenofibrate which had previously been problematic.60,61 For a more detailed discussion of LC-MS/MS, cortisol assays readers are directed to a recent review by Hawley and Keevil. 58
Free cortisol assays
Direct measurement of serum-free cortisol has been possible since the 1950s, although available methods – equilibrium dialysis, gel filtration and ultrafiltration – are too labour intensive and time consuming for routine clinical use. Attempts to develop a semi-routine assay for free cortisol measurement are ongoing, with ultrafiltration offering the most likely option. 62
Equilibrium dialysis is particularly time consuming. A dialysis membrane separates a small volume of plasma from a buffer solution and the two are incubated for up to 24 h. 63 Free cortisol crosses the membrane until equilibrium is reached and can be measured in the dialysis buffer. However, the accuracy of this method is affected by serum leakage across the dialysis membrane and by changes in cortisol binding equilibrium within the plasma sample due to dilution with dialysis buffer. Nevertheless, an equilibrium dialysis isotope-dilution LC-MS/MS method using a commercially available dialysis cell (Amicon) has been described and offers potential for a clinical assay. 62
Gel filtration relies on unbound cortisol moving down a glass column packed with Sephadex G-25 more slowly than its protein-bound counterpart. 64 As the latter passes through the column, the cortisol dissociates from its binding protein until the concentration of dissociated cortisol in the column equals the unbound fraction in the original sample and no further dissociation occurs. As the protein-bound cortisol elutes first the concentration of serum-free cortisol can be measured in the later fraction. This method requires large sample volumes to create a steady state in the gel column and, although analysis is possible at any temperature, 37℃ is preferred as temperatures below this may affect cortisol-CBG binding equilibrium.
Ultrafiltration is a relatively simple method and benefits from the availability of commercial filters, e.g. Amicon ultracentrifugal filters with a 30 KDa cut-off (Millipore, UK). Plasma, separated from a collection chamber by a dialysis membrane, is centrifuged for 10 min at 37℃ and the cortisol concentration in the resulting filtrate is measured. 65 However, substantial variations in filtrate yield can be seen, presumably due to the forced filtration disturbing cortisol binding equilibrium at the membrane surface. Precise temperature control can also be difficult to achieve, particularly in a routine laboratory setting.
Routine measurement of serum-free cortisol is further hampered by the lack of a single, robust reference range. Most studies are small and have used in-house assays to measure cortisol, resulting in significant differences in the ranges quoted. Low substrate concentration and lack of stable quality control material have also contributed to these differences, although good correlation between serum-free cortisol measured by ultrafiltration and equilibrium dialysis has been reported. 66 In view of the difficulty measuring serum-free cortisol directly, interest has grown in deriving calculated estimates as an alternative.
Coolens et al. 67 derived an equation for calculating serum-free cortisol (CFC), taking into account the binding properties of both CBG and albumin, and demonstrated excellent correlation between this estimate and serum-free cortisol measured by ultrafiltration. The free cortisol index (FCI) is another estimate, defined as serum total cortisol divided by CBG, and has been shown to correlate well with serum free cortisol measured by gel filtration and with Coolens’ CFC. 68 However, calculated free cortisol estimates have not yet been validated in patients with HPA axis disease and Coolens’ CFC has been shown to underestimate measured free cortisol in patients undergoing a short Synacthen test. 69 Substantial bias between calculated and measured free cortisol in critically ill patients has also cast doubt on its role in this population.70,71 Better understanding of how serum-free cortisol behaves in health and disease is, therefore, essential if it is to become a practical alternative to total cortisol.
Urinary and salivary cortisol assays
Urine
Under normal conditions, the kidneys filter, and partially reabsorb, unbound cortisol, and only a very small fraction (∼2%) is excreted unchanged in the urine. Urinary cortisol is unaffected by changes in hepatic metabolism, unlike urinary cortisol metabolites, and better reflects endogenous cortisol concentration.
Hypercortisolaemic patients have significantly higher renal cortisol clearance than patients with normal serum cortisol due to the rapid rise in serum-free cortisol once the binding capacity of CBG has been exceeded (at around 500 nmol/L), and due to continued cortisol excretion overnight. Thus, 24-h urine free cortisol correlates well with mean serum-free cortisol in conditions of cortisol excess in all but severe renal impairment, and is widely used as a screening test. 72 In some instances, e.g. in patients with adrenal incidentalomas or cyclical Cushing’s syndrome, cortisol metabolites perform better than urinary cortisol, 73 despite their variable 24-h excretion pattern. 74
Urinary cortisol was first measured fluorimetrically, with subsequent development of radioimmunoassay (RIA), enzyme-linked immunoassay (EIA) and chemiluminescent assays (CLIA). 75 However, the presence of conjugated cortisol metabolites in urine which can cross-react with immunoassay antibodies, 76 e.g. allotetrahydrocortisol glucuronide, necessitated extraction of free cortisol prior to analysis. By mixing the urine with an organic solvent, e.g. dichloromethane and removing the aqueous layer, cortisol, which is less water-soluble than most of its metabolites, can be separated from the latter prior to analysis.8,25 To further improve the specificity of urine cortisol assays, chromatography was introduced – initially as a purification step prior to immunoassay 77 – and has now become the method of choice.
Early HPLC assays were not ideal due to the long run times needed to ensure adequate separation; and by coupling HPLC or GC to mass-spectrometry for detection, run times were significantly reduced, although low throughput and high costs precluded these assays from widespread use.78,79 More recently, LC-MS/MS has provided highly sensitive and specific assays which are easy to use, with high sample throughput and which compare well to the gold standard GC-MS method. 78 These assays, however, can be more susceptible than serum assays to ion suppression due to the variable salt content and numerous steroid metabolites present in urine or sample extract.
Saliva
Cortisol enters saliva by diffusion, independently of salivary flow rate. 80 It reflects cortisol’s circadian rhythm and early morning peak, and responds to changes in plasma cortisol concentration quickly and reliably. 81 Interest in measuring salivary cortisol began in the 1960s but was hampered by the lack of sensitive cortisol assays as concentrations in saliva are less than one tenth those in serum. 80
Salivary cortisol was first measured by direct RIA in 1978 82 and, more recently, automated serum cortisol immunoassays have been successfully adapted to measure cortisol in saliva.83,84 However, these assays are limited by their poor specificity, particularly where there is significant antibody cross-reactivity with cortisone, which is present in high concentration in saliva. Assay sensitivity can also present a problem as late night salivary cortisol concentrations often fall close to, or below, the functional limits of detection.83,85
The first LC-MS/MS assay for measuring salivary cortisol was described in 2003 and promised improved specificity over existing assays. 86 Efforts have since concentrated on adapting LC-MS/MS to provide a quick, high-throughput service, 28 with numerous successful assays now available.87–90 However, widespread application of salivary cortisol measurement is limited by poorly standardized assays 85 and the lack of a single, validated reference range.89,90
Thus, mean salivary cortisol concentrations in the early morning of 3.6 nmol/L 87 and 8.3 nmol/L 88 have been reported in healthy laboratory workers in two studies using different in-house LC-MS/MS assays. Similarly, the late night salivary cortisol cut-off for diagnosing Cushing’s syndrome has been reported as 2.95 nmol/L and 2.1 nmol/L,91,92 and although it is likely to lie somewhere below 3 nmol/L, a single cut-off is yet to be determined. A further study of late night salivary cortisol showed significant overlap between concentrations in healthy volunteers and obese subjects, with ranges of <0.1–116 nmol/L and <0.11–17.7 nmol/L respectively. 89 Laboratories will, therefore, need to define their own assay-specific cut-offs until better assay standardization has been achieved and should be aware of the potential overlap between patients and healthy volunteers.
Sample collection technique may also contribute to the differences observed between salivary cortisol assays. Saliva is often collected by passive drooling into a container but can be collected by chewing an absorbent cotton pad, typically the Sarstedt Salivette®, which is later centrifuged to release its liquid content for analysis. Steroid recovery from cotton pads has been shown to differ from other collection devices, e.g. Whatman® foam-tip applicator and blood collection cards, 92 resulting in different concentrations simply due to the collection technique used. Thus, the development of any assay should include an evaluation of the chosen collection device.
Other potential interferences with salivary cortisol measurement include sample contamination with blood during collection and smoking before sample collection; both of which can lead to falsely elevated cortisol concentrations. 89 Avoiding the analysis of visibly pink saliva is, therefore, important and most authors suggest collecting saliva at least 30 min after eating, drinking or teeth-brushing,84,93 although there is little evidence to support this approach. 94
Salivary cortisol is now widely used in clinical laboratories: it has been suggested as a convenient alternative to serum for monitoring hydrocortisone replacement, 95 there is interest in it as an alternative to serum total cortisol in the interpretation of the Synacthen test,96,97 particularly in patients with altered protein concentrations 98 and late night salivary cortisol is a recommended first-line screening test for Cushing’s syndrome. 99 Salivary cortisol has also been used to research basal cortisol patterns in neonates and in evaluating their response to stress. 100
An alternative marker of serum-free cortisol is salivary cortisone, which shows excellent correlation with serum-free cortisol concentration88,101 and is found in higher concentration than salivary cortisol,101,102 due to the presence of 11β-HSD2 in parotid tissue. 103 Further work is now needed to establish suitable reference ranges and cut-offs for salivary cortisone and to investigate the effect of altered 11β-HSD2 activity on its diagnostic utility before it can be used in a clinical setting.
Cortisol in disease
Addison’s disease
The gold standard test of HPA axis function is the insulin tolerance test (ITT) in which insulin-induced hypoglycaemia stimulates the hypothalamus, resulting in CRH release and an increase in serum cortisol.
14
The short Synacthen test (SST) is a quick and safe alternative to the ITT and has replaced it as the first line test,
104
although as it does not assess the entire HPA axis, it can give false negative results in acute pituitary disease and in the early weeks post-pituitary surgery.17,105,106 It involves parenteral administration of a supraphysiological dose (250
Defining the most appropriate cortisol cut-off for use with the Synacthen test continues to pose some difficulty.105,110 Historically, a 30-min cut-off of 550 nmol/L was used, as it correlated well with results from the insulin tolerance test,111,112 but because of differences between cortisol immunoassays, a single cut-off is no longer applicable and this should be replaced by method-specific alternatives.15,30,41 These are likely to gain better clinical acceptance if they are shown to correlate well with the cortisol response to insulin-induced hypoglycaemia (which will itself need to be redefined).
Cushing’s syndrome
In 2008, the American Endocrine Society’s Clinical Guidelines Subcommittee published a guideline on the diagnosis of Cushing’s syndrome. 99 The authors collated evidence from studies that had demonstrated significant treatment benefit in patients diagnosed with Cushing’s syndrome to identify the tests that best detected the disease. They recommended using 24-h urine free cortisol, late night salivary cortisol, the 1 mg overnight dexamethasone suppression test (DST) or the 48 hour low-dose dexamethasone suppression test (LDDST) (0.5 mg of dexamethasone administered every 6 h for 48 h, beginning at 9 a.m. on day 1, with serum cortisol measured at 9 a.m. on day 3, 6 h after the final dose) for initial screening; the former two on at least two separate occasions. Confirmation of an abnormal screening test using a second test from the list is required and patients with discordant or positive results should be evaluated by an endocrinologist, while those with negative screening tests, reassured. The guideline also identifies population groups at greater risk of diagnostic confusion and makes specific recommendations about the most suitable tests to use. 99
The effect of immunoassay differences on interpretation of the DST, and hence the diagnosis of Cushing’s syndrome, has recently been studied, 113 and the current serum cortisol cut-off of 50 nmol/L, which was first defined in 1989 using a Diagnostic Products Corporation (DPC) cortisol RIA, 114 was shown to perform well with the Abbott assay and LC-MS/MS, with diagnostic sensitivities of 87.5% and 93.8% and specificities of 92.5% and 91.2%, respectively. 113
Diagnosing Cushing’s syndrome can be challenging, as evidenced by the multiple screening tests on offer and cyclical hypercortisolism contributes further diagnostic challenge.115,116 Cycles of cortisol hypersecretion may be regular or irregular with no discernible pattern, and cortisol concentrations in between each episode may be normal or even low. Repeated testing is, therefore, necessary. 116 Pseudo-Cushing’s presents another source of diagnostic confusion. It is a state of cortisol excess caused by increased HPA axis activity due to poorly controlled diabetes, obesity, alcoholism and certain psychiatric disorders.117,118 Patients present with clinical features of Cushing’s syndrome and positive screening tests despite only mild hypercortisolism. 118
In a study designed to distinguish patients with Cushing’s syndrome from those with Pseudo-Cushing’s, all recommended first-line tests showed excellent sensitivity but limited specificity, 119 leading several authors119,120 to recommend the desmopressin stimulation test and combined dexamethasone suppression – CRH stimulation (DST-CRH) test as more discerning alternatives.
The desmopressin stimulation test involves measuring ACTH before and, at intervals, after IV administration of 10
The DST-CRH test is recommended as a confirmatory test by the Endocrine society. It consists of the LDDST followed by intravenous (IV) CRH and subsequent measurement of cortisol and ACTH. 122 It relies on the premise that patients with pseudo-Cushing’s retain sensitivity to glucocorticoid suppression and fail to respond to CRH stimulation. In contrast, patients with true Cushing’s fail to respond to the LDDST, with those with an ACTH-producing tumour subsequently responding to CRH stimulation.
Adrenal incidentalomas (AI) and subclinical Cushing’s syndrome (SC) present yet further diagnostic confusion. AI are masses discovered by imaging studies performed for reasons other than the investigation of adrenal pathology and have a reported postmortem prevalence of 1.4–2.9%. 123 Identifying and treating those who require intervention because of hormonal activity or malignancy presents a significant challenge, particularly as the absence of clinical signs does not exclude a secretory mass. 123 SC is autonomous glucocorticoid production without the associated signs and symptoms of Cushing’s. It is found in 5 to 20% of patients with AI and carries a risk of progressing to overt Cushing’s syndrome.124,125
Accurately diagnosing SC remains a challenge. The DST is considered the test of choice,99,126–128 although there is disagreement over the best cut-off to use, partly due to assay differences and partly a lack of clinical consensus.127,128 Urinary free cortisol excretion and late night salivary cortisol perform relatively poorly as screening tests on their own129–131 and most diagnostic algorithms recommend combining the DST with a second measure of hypercortisolism, e.g. 24-h urinary cortisol excretion, failure to respond to CRH stimulation or loss of diurnal rhythm.128,131
Acute disease
Cortisol concentrations increase in acute illness via a stress-induced rise in CRH and reduced negative feedback from cortisol. 132 The diurnal pattern of cortisol secretion is lost and CBG concentrations fall, leading to increased circulating free cortisol. These changes are mediated by an increase in circulating cytokines, including tumour necrosis factor α, interleukin-1 and interleukin-6, which also increase cortisol at tissue level by increasing receptor affinity for cortisol and reducing its peripheral metabolism.132,133 The HPA axis can also be impaired by the disease process, resulting in a functional adrenal insufficiency, i.e. a transient (for the most part) episode of subnormal corticosteroid production.
Evaluating the HPA axis in acutely ill patients is complicated by increased synthesis of endogenous steroids by an activated HPA axis and a high prevalence of heterophilic antibodies. 134 The low protein concentrations typical of these patients affect the serum total cortisol response to Synacthen stimulation, resulting in a diagnosis of adrenal insufficiency in up to 40% of patients with no HPA axis disease. 135 Assay differences are accentuated, with one study showing concordance between the results of the Synacthen test in only four of the nine patients studied when samples were analysed using four different assays. 136
Similar results were found in an offshoot of the multinational CORTICUS (corticosteroid therapy of septic shock) study by Briegel et al. 134 in which the correlation coefficient for the concordance between serum total cortisol measurements from 12 different cortisol assays and the Roche Elecsys assay was 0.60 and the correlation with LC-MS/MS was as low as 0.43 for some assays. Furthermore, although the correlation between the Roche assay and LC-MS/MS was excellent for a group of outpatients, this fell when samples from critically ill patients were used instead. This has led to concerns that the clinical management of these patients is being determined by the cortisol immunoassay being used by their local laboratory rather than their actual adrenal status, and that serum-free cortisol or total cortisol measured by LC-MS/MS should be used to evaluate their HPA axis.
Cortisol in neonates
Basal cortisol
Early studies of neonatal serum cortisol identified high concentrations in cord blood at birth (880 nmol/L), 137 which started to fall within the first 24 h, plateauing at concentrations between 50 and 400 nmol/L by day 3. 138 More recently, a cortisol range of 4 to 588 nmol/L, with a median concentration of 93 nmol/L, was reported in infants born between 24 and 32 weeks’ gestation, 139 with concentrations falling with increasing gestational age. 140 This suggests that early reports of high neonatal cortisol concentration were overestimates, which may have arisen due to the relatively high cortisone concentrations in neonates cross reacting in cortisol assays.
Attempts to define a reference range for salivary cortisol in healthy infants have largely met with failure due to high intraindividual variability. 141 Nevertheless, Ivars et al. reported a median early morning salivary cortisol concentration of 5.1 nmol/L in one-month old infants, increasing to 10.9 nmol/L at 12 months. 142 Strict adherence to sample collection time was essential for meaningful interpretation of results.
Further studies in serum have shown that cortisol’s circadian rhythm is absent at birth, although its secretion, and that of ACTH, is pulsatile, and it settles into a circadian rhythm by around three months of age. 143 In contrast, studies in saliva have demonstrated a discernible circadian rhythm in full-term neonates at 1 month of age, 142 albeit with significant intra and interindividual variability in terms of how early the rhythm appears and how persistent it is once it has appeared.141,142,144 Further study is now needed to differentiate genuine differences from those arising due to non-standardized assays, poor specificity and matrix effects.
Conclusions
Early diagnosis of disorders of the HPA axis is essential and historically, serum total cortisol has been the analyte of choice. Most current cortisol immunoassays lack specificity and are poorly standardized, and in some clinical scenarios, e.g. in patients with altered protein concentrations and the critically ill, their performance is simply not good enough. LC-MS/MS provides a suitable alternative to immunoassay for measurement of serum total cortisol in all clinical scenarios. However, mass spectrometers are not yet ubiquitous in clinical laboratories, and even those that have the equipment may struggle with the extensive validation required to ensure LC-MS/MS methods are fit-for-purpose. 51
Both salivary cortisol and cortisone show promise as alternatives to serum cortisol, although further work to define reference ranges and their response to dynamic testing is needed before they can be used in routine clinical practice. There has also been interest in steroid hormone profiles for diagnosing adrenal disease, particularly congenital adrenal hyperplasia, although their role in diagnosing adrenal insufficiency and Cushing’s syndrome is not yet clear. The role of serum free cortisol measurement in clinical practice remains to be established.
Further improvements to serum and salivary cortisol assays will depend on the development and use of a single cortisol reference material and method to achieve standardized assays. EQA schemes will need to develop a more robust system for evaluating assay performance using samples with MS-assigned target concentrations.
Footnotes
Acknowledgements
We would like to thank Finlay MacKenzie, UKNEQAS for reading and commenting on the manuscript and for permission to use UKNEQAS data. This article was prepared at the invitation of the Clinical Sciences Review Committee of the Association of Clinical Biochemistry and Laboratory Medicine.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
NE.
Contributorship
NE wrote the first draft of the manuscript. All authors reviewed and edited the manuscript.
