Abstract
Background
Numerous studies demonstrate the potential of circulating microRNAs as non-invasive biomarkers for several diseases. Circulating microRNAs are much more stable than mRNAs and remain largely intact even after prolonged incubation at room temperature. However, recent reports show that microRNAs in serum or plasma samples have diverse stabilities. The aim of this pilot study is to evaluate the stabilities of miR-92a, miR-122 and miR-145 in serum during transient storage at 4℃ before freezing.
Methods
Serum samples were stored for 24 h at 4℃, and then RNA was extracted from whole serum or extracellular vesicles in serum. Total Exosome Isolation Reagent (from serum) was used for the fractionation of extracellular vesicles. Reverse transcription and real-time PCR of microRNAs were performed using the TaqMan MicroRNA Assays for miR-92a, miR-122 and miR-145.
Results
MiR-122 and miR-145 were degraded rapidly in serum; the concentrations dropped to 35.9% (P < 0.001) and 29.3% (P < 0.0001), respectively. These microRNAs in extracellular vesicles exhibited similar instability; the concentrations were 52.2% (P < 0.05) and 56.5% (P < 0.01), respectively. On the other hand, no significant degradation of miR-92a was observed (whole serum: P = 0.052, extracellular vesicles: P = 0.196).
Conclusions
MiR-122 and miR-145 in serum are extremely unstable and could be degraded during transient storage of serum at 4℃ prior to freezing.
Introduction
MicroRNAs are ∼22-nucleotide non-coding RNAs that bind the 3′-untranslated regions of target mRNAs to negatively regulate their stability or translation. MicroRNAs are present in various body fluids and these are collectively termed circulating microRNAs. Circulating microRNAs have been evaluated as non-invasive biomarkers for several diseases.
Early studies of circulating microRNAs suggested that most are remarkably stable. 1 However, Köberle et al. 2 recently reported diverse stabilities of microRNAs miR-1, miR-16, miR-21, miR-122 and miR-142-3p, and observed marked degradation of miR-122 and miR-1 over several hours at room temperature. Yamada et al. 3 reported that the concentrations of the circulating microRNAs miR-21, miR-29a, miR-125b and miR-16 in serum samples clearly decreased after serum samples were stored at 4℃ for seven days. 3 We previously reported that miR-223 is less stable than miR-21 at 4℃. 4
In general, serum samples used to quantify circulating microRNAs can be stably stored at −80℃; however, they are often transiently stored at 4℃ prior to freezing. In this study, we analysed the stabilities of miR-92a, miR-122 and miR-145 in extracellular vesicles in serum samples at 4℃.
Materials and methods
The study protocol was approved by the Ethical Committee of the Faculty of Health Sciences, Kyorin University. All participants signed informed consent. Peripheral blood was collected in VP-AS109K Vacutainer tubes (Terumo, Japan) from three healthy donors. Samples were left at room temperature for 30 min, and then centrifuged at 1500 g for 10 min at 4℃ to separate serum. The serum was stored for 0 h or 24 h at 4℃, and then centrifuged again at 20,000 g for 10 min at 4℃ to remove cell debris. The serum samples were pooled, divided into 200 μL aliquots, and stored at −80℃ until use.
Extracellular vesicles were extracted from 200 μL of pooled serum prior to freezing using the Total Exosome Isolation Reagent (from serum) (Thermo Fisher Scientific, USA). The precipitant was re-suspended in 200 μL of 1 × PBS. Extracellular vesicles were observed by transmission electron microscopy as previously described. 5 RNA was extracted from 200 μL of the pooled serum or the extracellular vesicle fraction using the miRNeasy Serum/Plasma Kit (Qiagen, Netherlands) according to the manufacturer’s protocol with a minor change: the volume of ultra-pure H2O used to elute RNA was 28 μL. RNeasy serum/plasma spike-in control (Qiagen), cel-miR-39, was added (5.6 × 108 molecules/200 μL of sample) after addition of QIAzol.
Five microlitres of the RNA eluate were used in reverse transcription. The TaqMan MicroRNA Reverse Transcription Kit (Thermo Fisher Scientific); TaqMan Universal Master Mix II, no UNG (Thermo Fisher Scientific) and TaqMan MicroRNA Assays for cel-miR-39, hsa-miR-92a, hsa-miR-122 and hsa-miR-145 were used for reverse transcription or real-time PCR. RNA extraction and microRNA quantification were performed three times independently. The 2−ΔΔCt method was used for relative quantification of microRNAs. ΔCt = (Ct of target microRNA)−(Ct of cel-miR-39). ΔΔCt = (ΔCt in time after storage)−(ΔCt in time 0). Relative quantity (RQ) = 2−ΔΔCt. Differences were considered significant when P < 0.05.
Results
The concentrations of miR-122, reported by Köberle et al.
2
to be short-lived at room temperature, decreased to 35.9% (P = 0.00023) over 24 h in whole serum (Figure 1(a)). The concentration of miR-145 dropped to 53.3% (P = 0.0118) over 6 h (data not shown) and 29.3% (P = 0.000047) over 24 h (Figure 1(a)). By contrast, no significant reduction in the concentration of miR-92a was observed (P = 0.052).
Quantification of miR-92a, miR-122 and miR-145 in serum samples after 24 h at 4℃. Open column: 0 h, shaded column: 24 h. Asterisks indicate P < 0.05 (*) or P < 0.01 (**) (Student’s t-test). (a) Concentrations of the microRNAs in whole serum. (b) Image of extracellular vesicles obtained by transmission electron microscopy. Scale bar, 200 nm. (c) Concentrations of the microRNAs in extracellular vesicles from serum samples. Quantitative data are presented as mean ± standard error.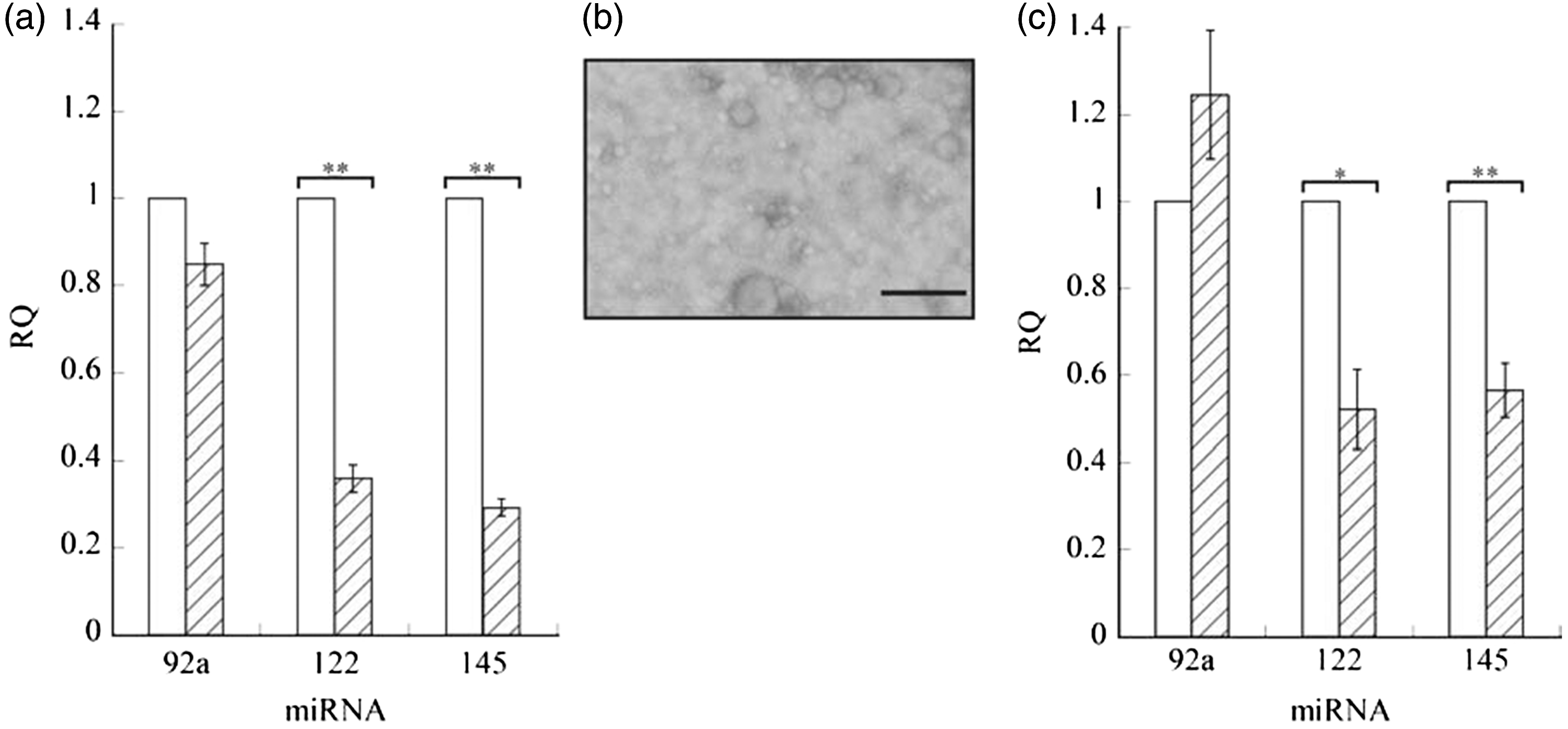
Next, we analysed the degradation of these microRNAs in extracellular vesicles during storage of serum at 4℃. Several vesicles of approximately 60–120 nm in diameter were observed in the extracellular vesicle fraction used (Figure 1(b)). The concentrations of miR-122 and miR-145 decreased to 52.2% (P = 0.014) and 56.5% (P = 0.0059), respectively (Figure 1(c)). No significant degradation of miR-92a was observed (P = 0.196).
Discussion
Circulating miR-122 is a potential marker for liver injury, and miR-145, a tumour suppressor and post-transcriptional regulator of c-Myc, is also a regulator of vascular smooth muscle cell differentiation, suggesting that it holds promise as a candidate biomarker for cardiovascular diseases. The results presented in this study show that serum miR-122 and miR-145 are unstable at 4℃ even if they are present in extracellular vesicles. The amount of total RNA extracted from extracellular vesicles in 200 μL of serum did not significantly decrease over 24 h (data not shown), suggesting that the reduction in miR-122 and miR-145 did not result from a reduction in the number of extracellular vesicles caused by vesicle rupture, but from the degradation of miR-122 and miR-145 in extracellular vesicles.
Circulating microRNAs are considered promising candidate biomarkers for cancer. However, little agreement was achieved on specific cancer markers. 6 One of the likely sources of discrepancy is related to the reproducibility in microRNA quantification. Köberle et al. 2 showed diverse stabilities of microRNAs, indicating that it is necessary to consider stability of each candidate microRNA in the quantification. We evaluated the stabilities of some candidate microRNAs for lung cancer biomarker in a preliminary study, and miR-92a and miR-145 were the most and the least stable microRNAs of them, respectively. We clearly showed in this study that miR-122 and miR-145 undergo degradation during storage of serum at 4℃ for 24 h. Our results suggest that microRNA degradation during transient storage of serum at 4℃ prior to freezing could lead to low reproducibility, especially for relatively unstable microRNAs such as miR-122 and miR-145. In conclusion, miR-122 and miR-145 in serum are extremely unstable and, to accurately quantify them, it is essential to avoid degradation during transient storage of samples at 4℃.
Footnotes
Acknowledgements
We thank Ms Sachie Matsubara (Laboratory for Electron Microscopy, Kyorin University School of Medicine) for assistance with electron microscopy.
Declaration of competing interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported in part by JSPS KAKENHI Grant Number 25460698.
Ethical approval
Ethical Committee of the Faculty of Health Sciences, Kyorin University (ref: 28-5).
Guarantor
TA.
Contributorship
TA, ST, AY and HO designed the study. TA, ST and AY performed experiments. TA and HO analysed the data and wrote the paper.
