Abstract
Aims
The prevention of acute kidney injury can be lifesaving for the intensive care unit patients. However, conventional methods are not sufficient for the prediction of the risk of future acute kidney injury. In this study, the promising biomarker, neutrophil gelatinase-associated lipocalin, was compared with cystatin C as an indicator for the risk of future acute kidney injury.
Methods
One hundred and eighty-three adult patients without chronic kidney disease or renal replacement therapy were included in this study. The plasma and urine concentrations of neutrophil gelatinase-associated lipocalin and cystatin C were assessed on the second day after intensive care unit admission and were followed for seven days to monitor the development of acute kidney injury. Acute kidney injury diagnosis was based on the risk, injury, failure, loss, end-stage renal failure criteria.
Results
Thirty-four per cent of the patients had acute kidney injury; 17 patients who did not fulfil criteria at the beginning, developed acute kidney injury from days 3 to 7 after admission. The mean serum creatinine on admission did not significantly differ between this and control groups (0.72 ± 0.20 and 0.83 ± 0.21; P = 0.060); however, the serum and urinary neutrophil gelatinase-associated lipocalin concentrations on the second day were significantly different (median: 75.69 [54.18–91.18] and 123.68 [90.89–166.31], P = 0.001; and median: 17.60 [8.56–34.04] and 61.37 [24.59–96.63], P = 0.001). Notably, the 48-h serum cystatin C concentration did not differ.
Conclusion
Neutrophil gelatinase-associated lipocalin concentrations in the urine and serum on the second day of intensive care unit admission could be used to predict the development of acute kidney injury in the following three to seven days in the intensive care unit; however, the cystatin C concentration did not have predictive value.
Introduction
The development of acute kidney injury (AKI) in critically ill patients is an unwelcome complication, and its diagnosis and treatment are controversial, despite the recent improvements in therapeutic techniques. After AKI is established in these patients, the mortality and morbidity become worse and increase the risk of permanent renal failure, which causes patients to require lifelong renal replacement therapy (RRT).1–3 Therefore, identifying patients who may develop AKI is crucial and can be lifesaving in the ICU.
The serum creatinine concentration and other urinary indicators have been used to diagnose AKI for many years. However, renal function must decrease by approximately 50% for the detection of a change in the serum creatinine concentration; therefore, this change in concentration does not reflect small changes in the renal function. In addition, the serum creatinine concentration may be affected by body mass index, muscle mass, gender, diet, fluid status and some drugs. These factors prevent it from being an ideal biomarker for use in diagnosing early-stage AKI. 2 Therefore, many studies have attempted to identify new and more accurate biomarkers for the diagnosis of early-stage AKI.
Cystatin C (CysC) and neutrophil gelatinase-associated lipocalin (NGAL) are the most promising biomarkers for AKI in critically ill patients. CysC is a low-molecular weight protein that originates in nucleated cells and functions as a cysteine protease inhibitor. It is released into the blood at a constant rate. In addition, it is filtered through the glomerular membrane and reabsorbed by the tubular epithelium, where it is almost completely catabolized. It is not filtered into the urine under normal circumstances.3,4 However, in the presence of tubular injury, it is not completely reabsorbed and may be released into the urine. The CysC concentration has been used for many years as an alternative to creatinine for the calculation of the steady-state glomerular filtration rate (GFR) in patients. The CysC concentration is more sensitive and specific than the creatinine concentration, possibly because it is less affected by extra-renal factors compared with creatinine.3–5 However, some reports have indicated that age, male gender, current smoking status, an elevated C-reactive protein (CRP) concentration, thyroid dysfunction, glucocorticoid use, aortic aneurism and atherosclerosis may affect the serum CysC (sCysC) concentration.4,6,7 sCysC is also affected by obesity, because it is released by adipose tissues.3,7–9
NGAL is a 25 kDa protein that is a well-known, highly sensitive marker of AKI but is not sufficiently specific to detect it. 10 It is predominantly expressed in the proximal tubule of the kidney after ischaemia, toxic injury, or septic insult. NGAL expression begins within 2 h after cardiopulmonary bypass surgery, which is much earlier than changes in the serum creatinine concentration can be detected. 11
The use of biomarkers should assist in the risk stratification and should indicate which patients are more prone to the eventual development of AKI. This approach would enable physicians to protect patients from further insults and apply the necessary interventions as early as possible.
Patients in intensive care units (ICUs) represent the most difficult population for the prediction of the development and outcome of AKI using biomarkers.12,13 These patients are not at a steady state and can exhibit many co-morbidities. Further, the time of insult and baseline GFR are not known. The ideal time for the assessment of the biomarker levels in these patients is unknown. Therefore, two days after admission was selected as the criterion to determine the level of biomarkers. It was predicted that critically ill patients would be sufficiently stabilized for further evaluation during this time interval. We previously studied NGAL in serum and urine and determined that this biomarker effectively predicts the eventual development of AKI in patients. 14 In this study, we evaluated CysC on the second day after ICU admission to determine whether it could predict the development of AKI as well as NGAL after seven days in the ICU.
Materials and methods
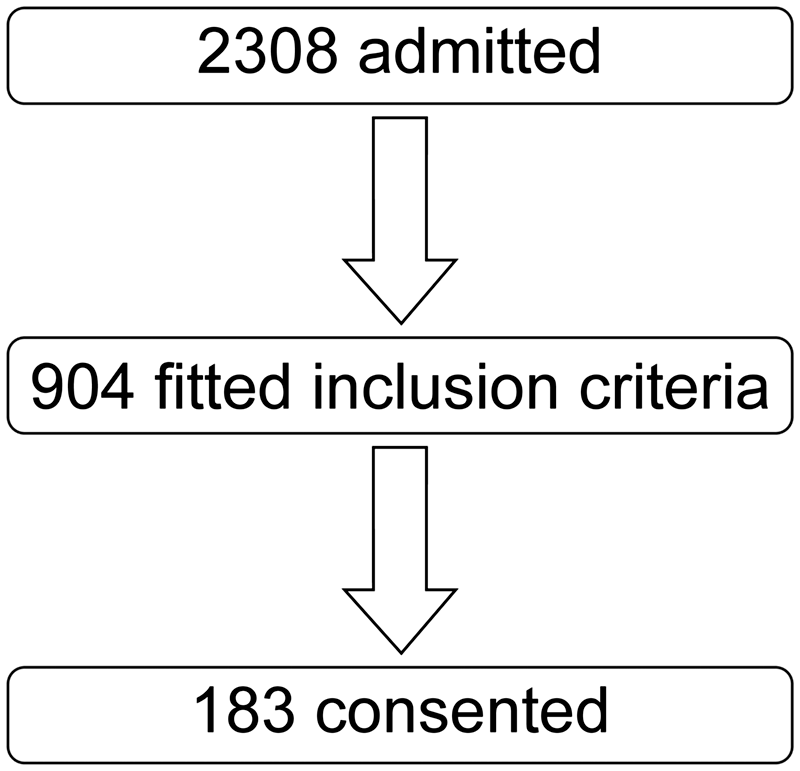
This was a prospective, observational, clinical study conducted in the ICU of a primary educational hospital between October 2011 and December 2013. One hundred and eighty-three of 2308 adult (over 17 years of age) critically ill patients, who had no known chronic kidney disease, were not undergoing RRT, had not received a previous kidney transplant, had no thyroid dysfunction, glucocorticoid use, or aortic aneurism, and remained in the ICU for more than two days, were enrolled in this study (Figures 1 and 2). The demographic features, medical histories and clinical features of the patients and the results of biochemical and haematological analyses were prospectively evaluated. The patients were followed up for seven days or until death or discharge from the ICU. The presence of diabetes mellitus was defined as the requirement of an antidiabetic drug or insulin treatment within the past year, and cardiovascular problems were defined as the presence of an ischaemic heart condition, cardiomyopathy, heart failure, or peripheral vascular disease. Chronic obstructive lung disease was defined as requiring a bronchodilator at any time. Patients with an immunosuppressive disease or oncologic or haematologic malignancy and those who had been treated with corticosteroids or cytotoxic or immunosuppressive drugs were considered immunosuppressed. A medical history of hepatic failure indicated liver cirrhosis.
15
Sepsis and systemic inflammatory response syndrome (SIRS) were defined based on the American College of Chest Physicians and Society of Critical Care Medicine (ACCP/SCCM) criteria.16,17 The APACHE II and SOFA scoring systems were used to determine the severity of the cases.18,19 AKI was diagnosed based on the risk, injury, failure, loss, end-stage renal failure (RIFLE) criteria at admission and each day during the following seven days in the ICU.
20
When the baseline serum creatinine (one day before the admission) concentration was not available, the lowest creatinine concentration measured during the previous three months was used.
21
Primary diagnosis at admission to the ICU. Flow chart of patients.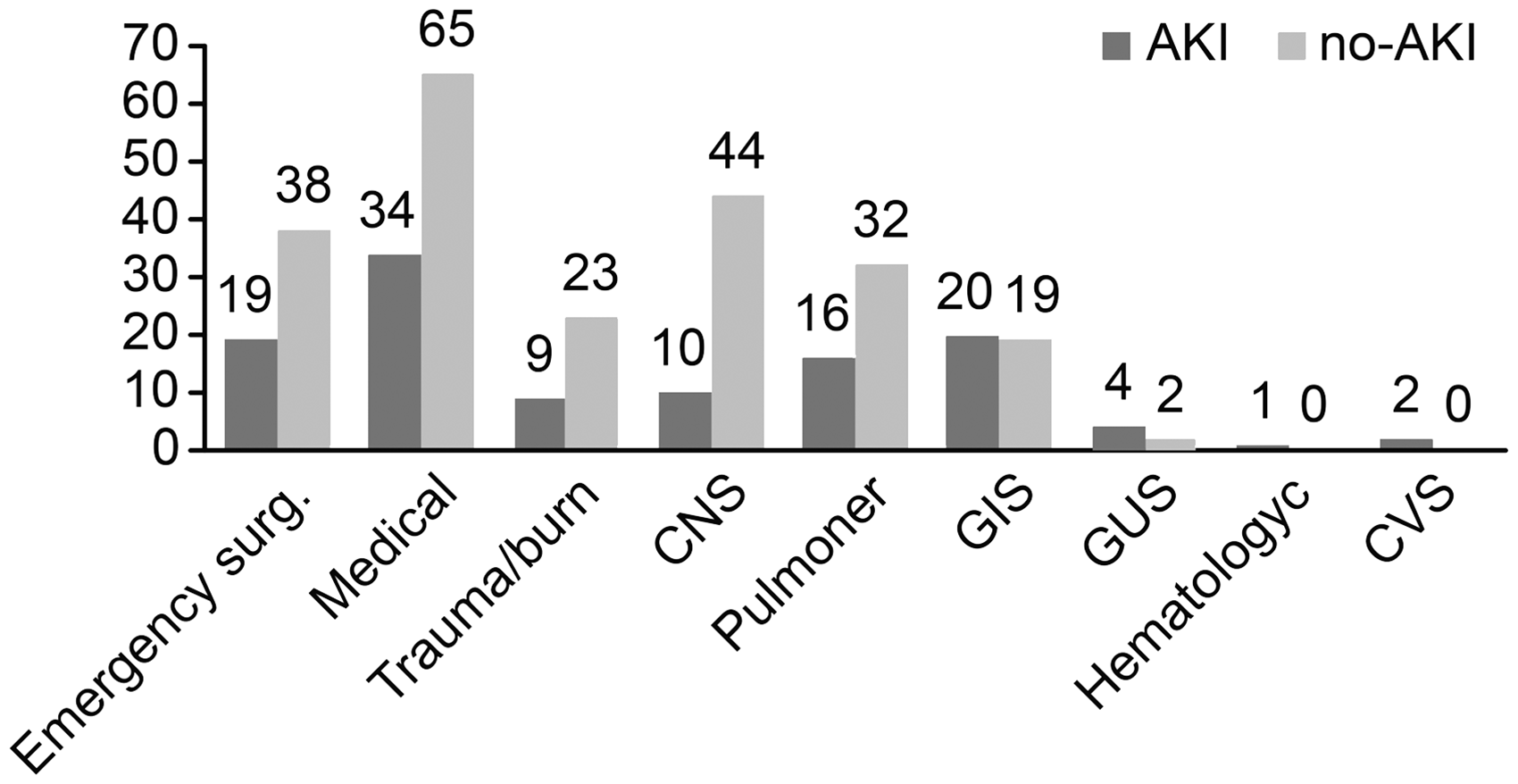
The patients were classified into the following four groups according to the time at which AKI developed during the ICU stay: Group 1: 16 patients who developed AKI within the first 2 days of admission to the ICU and then recovered from AKI. Group 2: 17 patients who did not fulfil the RIFLE criteria in the first two days but developed AKI between days 3 and 7 in the ICU. Group 3: patients who presented with AKI from admission to day 7 of follow-up (30 patients). Group 4: patients who did not develop AKI during the follow-up in ICU (control group; 120 patients).
The NGAL and CysC concentrations were measured in blood and urine samples collected on the second day of admission. Blood samples were centrifuged for 10 min, and the serum was separated and stored at −80℃ until the determination of the NGAL and CysC concentrations by micro-ELISA (BioVendor R&D, Czech Republic).
Written informed consent was obtained from each participant or their next of kin after they were clearly informed of the study details. This study was approved by the Kocaeli University Ethical Committee (KOU KAEK) (2011/39-14/3) (2015/219).
Statistical analysis
Statistical analysis was conducted using IBM SPSS 20.0 (SPSS Inc., Chicago, IL, USA). Comparisons of continuous variables between two groups were performed using Student’s t-test. The distribution of data was analysed using the Kolmogorov-Smirnov test. If the distribution was not normal, then the Mann-Whitney U test, Kruskal-Wallis one-way analysis of variance and Tukey’s multiple comparisons test were used. Pearson’s, Fisher’s exact, and χ2 tests were used to analyse categorical variables. Numeric variables are presented as the mean ± standard deviation, median (25th percentile–75th percentile) and frequency (%). Receiver operating characteristic (ROC) curves were generated, and the areas under the curves (AUCs), including the 95% confidence intervals, were calculated. The AUCs were compared using non-parametric tests, and the sensitivities, specificities, positive predictive values and negative predictive values were calculated. Linear regression analysis was performed to determine the factors that potentially affected the levels of the biomarkers. The two-tailed significance level was set at P < 0.05.
Results
Clinical and laboratory features of all patients.
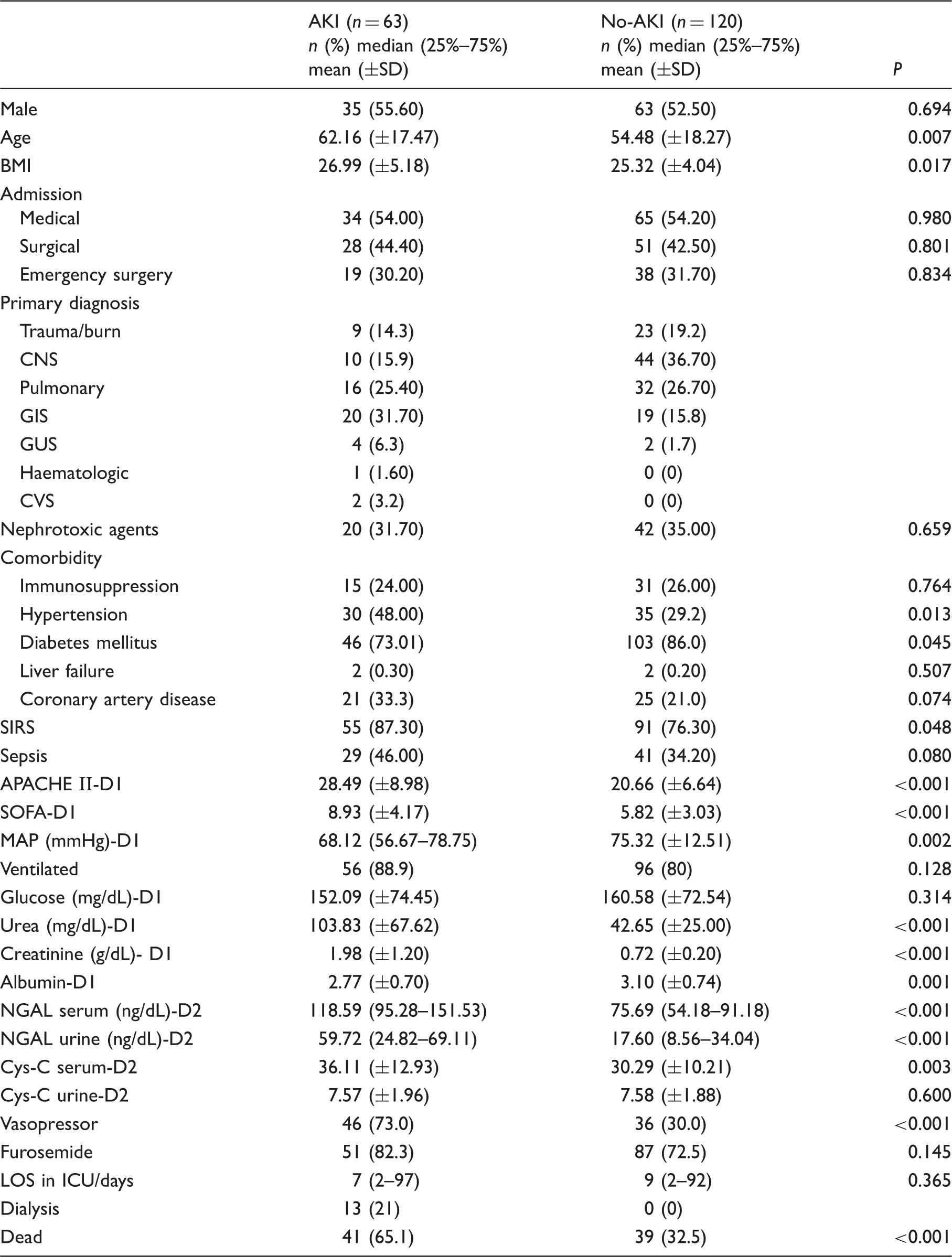
BMI: body mass index; CNS: central nervous system; GIS: gastro intestinal system; GUS: genitourinary system; CVS: cardio vascular system; SIRS: systemic inflammatory response syndrome; MAP: mean arterial pressure; NGAL: neutrophyl gelatinase associated lipocalin; LOS: length of stay in hospital; D1: day of admission; D2: 48 h after admission.
Sixty-three patients (34%) were diagnosed with AKI based on the RIFLE criteria. Gender and exposure to nephrotoxic agents did not differ patients with and without AKI, but those with AKI were significantly older than those who were without AKI. The rate of sepsis did not differ between patients with and without AKI, but SIRS was observed more frequently in those with AKI (87.3% and 76.3%, P = 0.048). Furthermore, a history of hypertension was more common in AKI patients, but the incidence of diabetes mellitus was higher in non-AKI patients. As expected, the APACHE II and SOFA scores were also higher for AKI patients than for non-AKI patients. The serum NGAL (sNGAL), serum CysC (sCysC), and urinary NGAL (uNGAL) concentrations significantly differed between AKI and non-AKI patients (Table 1). The urinary CysC (uCysC) concentration did not differ between the groups of patients; however, the assessment of this concentration was considered unreliable in this study because no adjustment for the urinary creatinine concentration was performed.
AKI patients were further classified according to the number of days in the ICU prior to fulfillment of the RIFLE criteria. Within the first two days, 46 (73%) patients fulfilled these criteria. In addition, 16 patients recovered from AKI after two days, and 17 new patients were added to the AKI group during the three- to seven-day follow-up. The mean serum creatinine concentrations at the first day of admission in the non-AKI patients (group 4) and in the 17 patients who did not fulfil RIFLE criteria during the first two days but were diagnosed from days 3 to 7 during follow-up (group 2) did not differ significantly (0.72 ± 0.20 and 0.83 ± 0.21; P = 0.060). However, the sNGAL and uNGAL concentrations differed significantly between these groups (median: 75.69 [54.18–91.18] and 123.68 [90.89–166.31], P = 0.001; and median 17.60 [8.56–34.04] and 61.37 [24.59–96.63], P = 0.001 in the non-AKI and AKI patients, respectively) (Figure 3).
(a) Serum NGAL concentrations based on the time of AKI development. (b) Urinary NGAL concentrations based on the time of AKI development. (c) Serum CysC concentrations based on the time of AKI development. (d) Urinary CysC concentrations based on the time of AKI development. First column: no AKI (group 4); second column: the development of AKI on the second day of admission to the ICU (group 1); third column: the development of AKI within three to seven days after admission to the ICU (group 2); and fourth column: the presence of AKI from day one to seven days of follow-up in the ICU (group 3).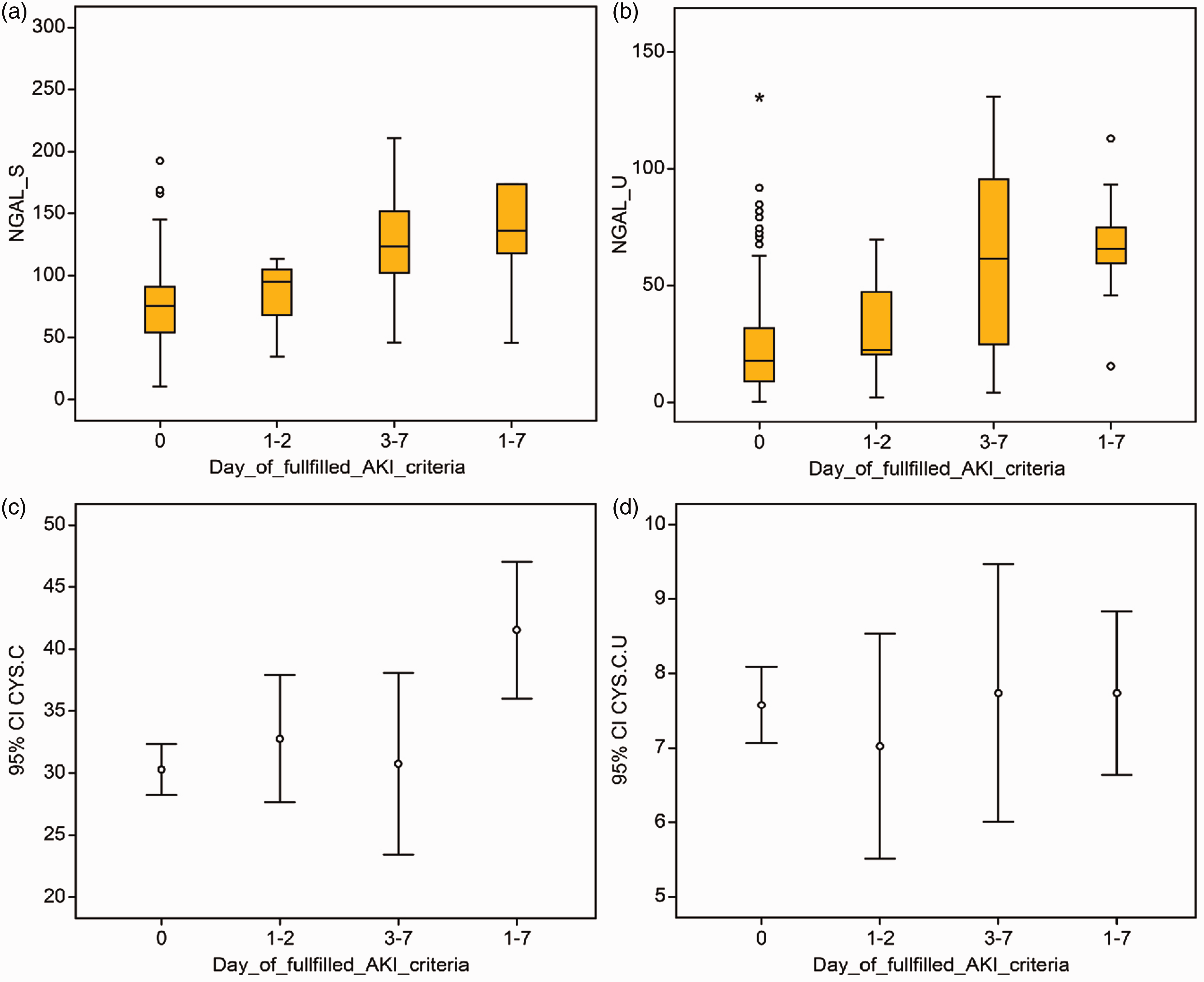
ROC analysis was performed to compare the efficacies of the sNGAL, uNGAL, sCysC and uCysC concentrations determined on the second day in the ICU for the prediction of the development of AKI after three to seven days. The sNGAL and uNGAL concentrations, but not the sCysC concentration, on the second day were useful for the prediction of the development of AKI after three to seven days in the ICU (AUC-ROC: 0.77 [0.66–0.86], P = 0.005; 0.78 [0.67–0.86], P = 0.002 and 0.54 [0.44–0.63], P = 0.657, respectively) (Figure 4). For all AKI patients, the AUC-ROC values for sCysC, sNGAL and uNGAL were 0.66 (0.58–0.74, P < 0.001), 0.76 (0.66–0.83, P < 0.001) and 0.77 (0.68–0.58, P < 0.001), respectively.
ROC plots showing the predictive abilities of biomarkers for the detection of AKI based on the RIFLE criteria during follow-up. (a) Serum CysC: AUC-ROC: 0.66 (0.58–0.74) with a cut-off of 38.29; specificity: 0.80; sensitivity: 0.52; and P = 0.0009. (b) Serum NGAL: AUC-ROC: 0.76 (0.66–0.88) with a cut-off of 93.70; specificity: 79.41; sensitivity: 76.92; and P < 0.0001. (c) Urinary NGAL: AUC-ROC: 0.77 (0.68–0.85) with a cut-off of 41.78; specificity: 79.41; sensitivity: 74.36; and P < 0.0001.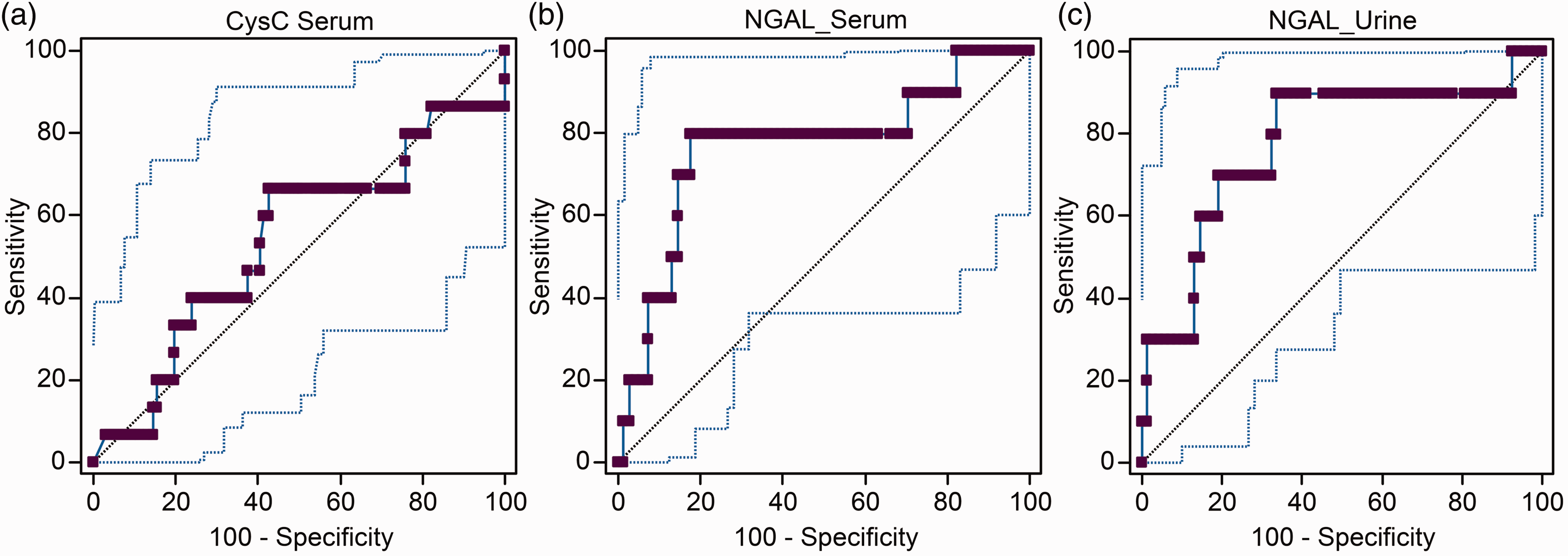
Linear regression analysis was performed to evaluate the factors that affected the NGAL and CysC concentrations. Based on this assessment, patient age, haemoglobin (Hb) concentration, creatinine concentration and coronary artery disease affected the sCysC concentration (P = 0.028, 0.028, 0.041 and 0.050, respectively). However, only the APACHE II severity score and serum Hb concentration influenced the sNGAL concentration(P = 0.004 and 0.003). The mortality rates of AKI and non-AKI patients were 65.1% and 32.5%, respectively. Patients who developed AKI during the first two days in the ICU and recovered had a mortality rate of 44%, but those who exhibited progression of AKI for seven days (group 3) had a higher mortality rate of 70%.
Discussion
Early recognition of AKI in patients admitted to the ICU is critical and affects the mortality rate. The prevention of kidney damage in critically ill patients can reduce the risk of developing end-stage renal disease and significantly decreases mortality. Therefore, research on biomarkers for the identification of patients before they develop AKI is critical.2,15,22
A small change in the GFR may affect the filtration of CysC, but not that of creatinine.16,23 Therefore, many investigators proposed that CysC may be more sensitive for the prediction of early AKI development and small changes in the GFR than conventional markers, such as creatinine. 24 A recent study of ICU patients suggested that a 50% increase in the CysC concentration predicts the development of AKI within 24 h (AUC-ROC: 0.97), indicating that CysC is a very reliable marker of small changes in the GFR that may be useful for the detection of early-stage AKI. 24 However, a large multicenter study revealed that CysC is less sensitive than creatinine for the early diagnosis of AKI. 25 In this study, we demonstrated that the sCysC concentration during the first 48 h was not reliable for the prediction of subsequent AKI development in ICU patients.
This study demonstrated that the sNGAL and uNGAL concentrations are better markers than sCysC for the prediction of the development of AKI. NGAL, similar to CysC, was not significantly predictive in patients diagnosed with AKI during the first two days (AUC-ROC: 0.61; P = 0.259; and AUC-ROC: 0.54; P = 0.319, respectively). However, unlike the sCysC concentration, the uNGAL and sNGAL concentrations were strong predictive factors for AKI development over the following days. Constantin et al. 26 found that the plasma NGAL concentration in ICU patients with a RIFLE score of 0 during the first 48 h after admission is predictive of the development of AKI during the following week in the ICU (AUC-ROC: 0.97; sensitivity: 93%; and specificity: 97%).
The sNGAL and uNGAL concentrations are increased by 10- to 100-fold in tubular epithelial cells at 2 h after insult leading to AKI, but the serum creatinine concentration does not increase until 24 to 72 h after kidney injury.11,27 These findings indicate that NGAL is a better biomarker than conventional biomarkers. However, previous studies and the present study have demonstrated that the NGAL concentration is highly variable and exhibits a very wide standard deviation range, possibly because sNGAL can be derived from systemic sources other than renal tubular cells. 28 When the sNGAL concentration in the systemic circulation increases, although tubular epithelial cells are intact, the reabsorption capacity of these cells is overwhelmed and the uNGAL concentration increases. The NGAL discussed here is not specific to the kidney and does not necessarily reflect tubular injury. Many events occurring in critically ill patients may affect the sNGAL concentration, and predicting AKI development in these patients is thus difficult. The NGAL concentration may increase in response to acute or chronic infection, sepsis, cardiopulmonary surgery, chronic kidney disease, urinary infection, chronic obstructive pulmonary disease and many other situations encountered in the ICU. These conditions make the identification of causes of acute tubular injury difficult to distinguish; however, early and more specific diagnosis of AKI in ICU patients may become possible with the development of a kidney-specific NGAL (monomer) assay. 29
In conclusion, the results of this study have demonstrated that although sCysC is a sensitive marker for the early detection of the deterioration of kidney function in steady-state patients, it is more useful for diagnosing the current rather than future AKI. The findings of this study support the results of our previous study showing that uNGAL and sNGAL but not sCysC concentrations in ICU patients are useful for the prediction of the subsequent development of AKI.
Footnotes
Acknowledgements
We are grateful to the nurses and technicians in the ICU who helped us with our data collection.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the Kocaeli University Ethical Committee (KOU KAEK) (2011/39-14/3) (2015/219).
Guarantor
IY.
Contributorship
IY: Researched literature and conceived the study and wrote the first draft of the manuscript.
FK, KC, NE: Gained ethical approval, patient recruitment and data analysis.
CB: Statistical and data analysis.
EE: Laboratory work and data interpretation.
NB: Researched literature and conceived the study, patient recruitment and data analysis.
All authors reviewed, edited and approved the final version of the manuscript.
