Abstract
Background
Urinary excretion of alpha-1-microglobulin and beta-2-microglobulin reflects tubular damage and predicts outcome in patients with idiopathic membranous nephropathy with reasonable accuracy. Urinary kidney injury molecule-1 and neutrophil gelatinase-associated lipocalin are novel biomarkers of tubular damage. We investigated if these markers could improve prediction of outcome in idiopathic membranous nephropathy.
Methods
We measured kidney injury molecule-1 and neutrophil gelatinase-associated lipocalin in urine samples from patients with idiopathic membranous nephropathy, who had nephrotic proteinuria and normal renal function. Excretion of alpha-1-microglobulin and beta-2-microglobulin had been measured previously. Progression was defined as a serum creatinine rise >30%, a rise in serum creatinine to an absolute value of ≥135 µmol/L, or a clinical decision to start immunosuppressive therapy. Remission was defined as proteinuria <3.5 g/day and >50% reduction from baseline.
Results
Sixty-nine patients were included. Median follow-up was 35 months (interquartile range 18–63 months). Progression occurred in 30 patients (44%), and spontaneous remission in 36 (52%). Kidney injury molecule-1 and neutrophil gelatinase-associated lipocalin excretion rates were significantly correlated with each other, and with alpha-1-microglobulin and beta-2-microglobulin. The areas under the receiver operating characteristic curves for progression were 0.75 (0.62–0.87) for kidney injury molecule-1 and 0.74 (0.62–0.87) for neutrophil gelatinase-associated lipocalin. In multivariate analysis with either alpha-1-microglobulin and beta-2-microglobulin, kidney injury molecule-1 and neutrophil gelatinase-associated lipocalin did not independently predict outcome.
Conclusion
Kidney injury molecule-1 and neutrophil gelatinase-associated lipocalin excretion rates correlated with excretion rates of other tubular damage markers and predicted outcome in patients with idiopathic membranous nephropathy. They did not add prognostic value compared to measurement of either alpha-1-microglobulin or beta-2-microglobulin.
Keywords
Background
Idiopathic membranous nephropathy (iMN) is a leading cause of nephrotic syndrome in Caucasian adults. 1 Natural history studies with follow-up of at least three years indicate that approximately 50% of patients with nephrotic iMN develop renal failure.2–4 It has been demonstrated that a restrictive treatment strategy which limits immunosuppressive treatment to patients with established renal insufficiency results in good long-term renal survival.5,6 Although this strategy assures that patients who will develop spontaneous remission do not unnecessarily receive toxic therapy, it delays its use in others. 7 Preferably, immunosuppressive treatment should be initiated at an early stage and only in patients who will otherwise develop progressive disease. This requires a highly accurate method to identify high-risk patients.
High urinary concentrations of proteins that reflect tubular or glomerular damage precede a decline in estimated glomerular filtration rate (eGFR) in patients with iMN.8–11 We have previously validated a prognostic model to predict disease progression in patients with iMN and preserved renal function using urinary beta-2-microglobulin (β2m) and IgG excretion rates. 12 However, a recent evaluation of the data indicated that a substantial proportion of patients are still misclassified when urinary β2 m or the closely resembling biomarker alpha-1-microglobulin (α1m), or IgG are used as predictors. 3
Kidney injury molecule-1 (KIM-1) and neutrophil gelatinase-associated lipocalin (NGAL) are novel and early markers of tubular damage, which have mainly been studied in the context of acute kidney injury.13–16 There is increasing evidence for a prognostic value of KIM-1 and NGAL in patients with glomerular disease.17–20 Neither were strongly correlated with β2 m excretion in proteinuric patients with IgA nephropathy. 17 The aim of the study was to evaluate the prognostic value of urinary KIM-1 and NGAL excretion in patients with iMN.
Methods
Subjects
The present study cohort represents a subgroup from a previously described cohort of 129 patients with biopsy-proven iMN designed to re-evaluate the prognostic value of urinary low-molecular weight proteins α1m and β2m. 3 Patients who were referred to our clinic for a timed urinary protein measurement between 1995 and 2009 had been included. Other inclusion criteria were preserved renal function (defined by a serum creatinine <135 µmol/L), proteinuria ≥3.0 g/10 mmol creatinine, and absence of prior immunosuppressive therapy. Patients were excluded if follow-up was <1 year, or if time between renal biopsy and measurement was >3 years. In the present study, we included all patients from this cohort who had available stored urine samples for measurement of KIM-1 and NGAL. Patients were followed until a predefined end point (see below) was reached or until June 2011.
Methods
Patients with iMN were evaluated using a standardized protocol, which included a timed measurement of urinary proteins including β2m and α1m. 12 Patients were instructed to take sodium bicarbonate on the night before urinary measurement to obtain a urinary pH >6.0, which is mandatory for reliable measurement of β2 m. Timed urine samples were collected, and blood samples were obtained. Urinary β2 m was measured by an in-house developed ELISA (detection limit of 0.002 mg/L, interassay variation coefficient of 5.7%), and urinary α1 m was measured by immunonephelometry on a BNII nephelometer (Behring, Marburg, Germany; detection limit for urinary α1m was 2 mg/L, and interassay variation coefficient was 4.7%).12,21 A portion of the timed urine sample was immediately centrifuged (10 min, 3000 r/min) and stored at −80℃. Urinary KIM-1 and NGAL concentrations were measured after overnight thawing of frozen stored samples using human-specific ELISA (intra-assay and inter-assay variation coefficients for KIM-1 7.4 and 14.5%; for NGAL 6.8 and 19.6%). Antibodies were obtained from R&D systems (Minneapolis, MN). Detection limit for KIM-1 was 0.02 µg/L, for NGAL 0.66 µg/L. All measurements were performed in duplicate.
Outcome
Progression was defined as (a) a ≥30% rise in serum creatinine, (b) a rise in serum creatinine to an absolute value ≥135 µmol/L, or (c) other clinical indications for starting immunosuppressive therapy established by the treating nephrologist. Complete remission was defined as proteinuria reduction to <0.2 g/10 mmol creatinine. Partial remission was defined as proteinuria <3.5 g/10 mmol creatinine with reduction of >50% from baseline in the presence of stable renal function. 22
Statistical analyses
Median values and interquartile range are reported for non-normally distributed values. Statistical differences between groups were assessed with χ2 or Mann–Whitney U test. Correlations between parameters were calculated with Spearman’s correlation coefficient. Areas under the receiver operating characteristic curves (ROC-AUC) were used to assess prognostic performance of respective biomarkers. For all biomarkers, the optimal cut-off level was determined where the most patients were correctly classified. The discriminatory capacity for progression and remission of respective biomarkers at optimal cut-off levels was compared using the net reclassification improvement (NRI). Calibration of biomarkers was plotted using the Hosmer–Lemeshow goodness-of-fit test, which compares expected versus observed progression across deciles of risk. We used logistic regression models to evaluate combinations of biomarkers. Statistical analyses were performed with SPSS (IBM SPSS Statistics for Windows, Version 20.0. Armonk, NY: IBM Corp.) and GraphPad Prism (GraphPad Prism version 5.03 for Windows, GraphPad Software, San Diego California USA, www.graphpad.com). P values <0.05 were considered to reflect statistical significance.
Results
Clinical characteristics of patients with idiopathic membranous nephropathy included in the analysis (urine available for KIM-1/NGAL measurement).
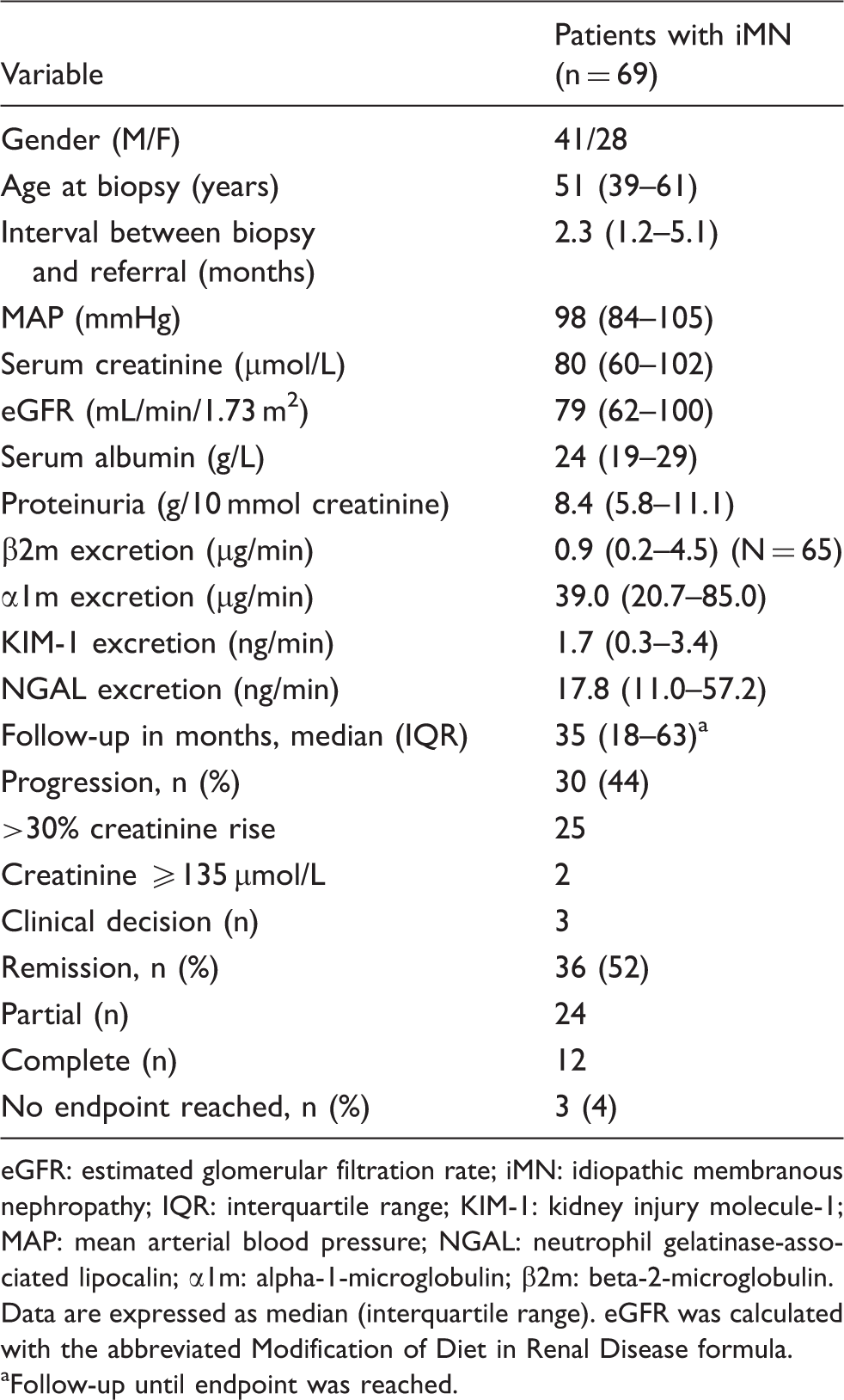
eGFR: estimated glomerular filtration rate; iMN: idiopathic membranous nephropathy; IQR: interquartile range; KIM-1: kidney injury molecule-1; MAP: mean arterial blood pressure; NGAL: neutrophil gelatinase-associated lipocalin; α1m: alpha-1-microglobulin; β2m: beta-2-microglobulin.
Data are expressed as median (interquartile range). eGFR was calculated with the abbreviated Modification of Diet in Renal Disease formula.
Follow-up until endpoint was reached.
Excretion of KIM-1 and NGAL
Correlations of KIM-1 and NGAL urinary excretion with other parameters.
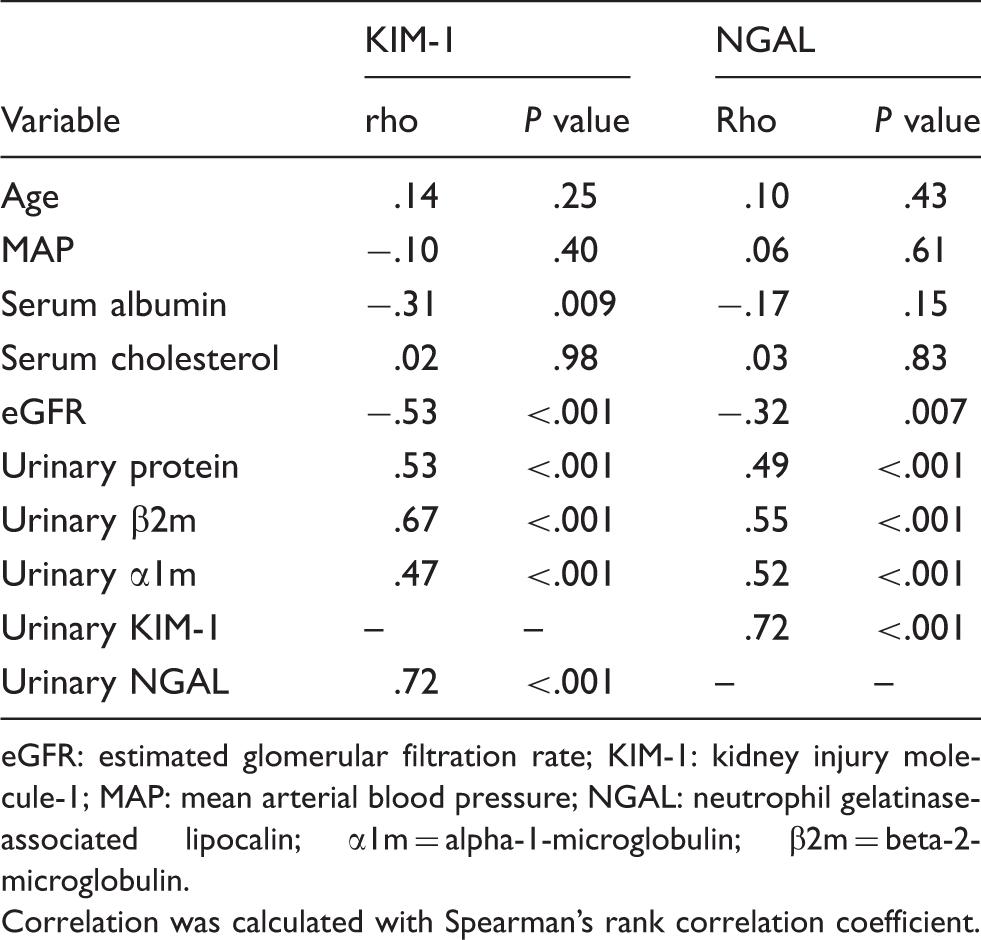
eGFR: estimated glomerular filtration rate; KIM-1: kidney injury molecule-1; MAP: mean arterial blood pressure; NGAL: neutrophil gelatinase-associated lipocalin; α1m = alpha-1-microglobulin; β2m = beta-2-microglobulin.
Correlation was calculated with Spearman’s rank correlation coefficient.
Prediction of outcome
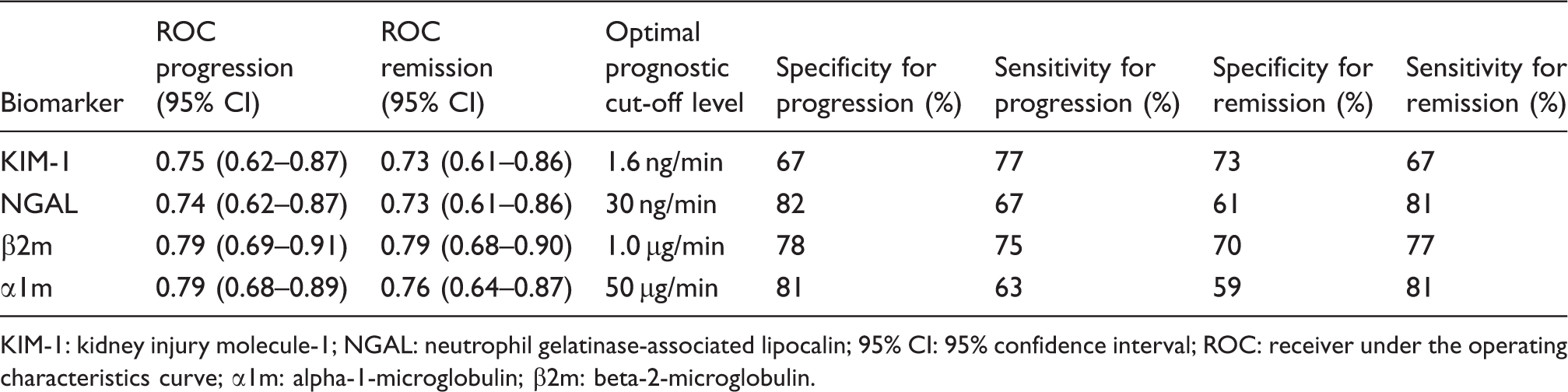
ROC curves for progression and remission, and optimal cut-off values of respective biomarkers are shown in Figure 1 and Table 3. The NRI values show that there is no significant improvement in outcome prediction of KIM-1 or NGAL compared to α1m or β2m (Figure 2). Hosmer–Lemeshow goodness-of-fit tests were non-significant for all biomarkers, indicating good model calibration (Supplementary Figure). In the backward stepwise logistic regression models, KIM-1 and NGAL did not reach statistical significance as independent predictors.
Receiver operating characteristic curves for prognostic accuracy of kidney injury molecule-1 (KIM-1; AUC 0.75 [95% CI 0.62–0.87]), neutrophil gelatinase-associated lipocalin (NGAL; AUC 0.74 [95% CI 0.62–0.87]), β-2-microglobulin (β2m; AUC 0.79 [95% CI 0.69–0.91]), and α-1-microglobulin (α1m; AUC 0.79 [AUC 0.68–0.89]) in the prediction of disease progression. Reclassifications using prognostic cut-off values for urinary neutrophil gelatinase-associated lipocalin (NGAL; 30 ng/min) or kidney injury molecule-1 excretion (KIM-1; 1.6 ng/min) versus beta-2-microglobulin (β2m; 1.0 µg/min) or alpha-1-microglobulin (α1m; 50 µg/min), respectively. Urinary β2m was measured in 65 patients. (a) KIM-1 versus β2m. NRI progression 3.5% (95% CI −12.1 to 19.5%); NRI remission −8.6% (95% CI −23.4 to 6.2%); overall NRI −5.0% (95% CI −26.7 to 16.6%). (b) NGAL versus β2m. NRI progression −10.7% (95% CI −31 to 9.1%); NRI remission 8.6% (95% CI −4.0 to 21.1%); overall NRI −2.1% (95% CI −24.5 to 20.2%). (c) KIM-1 versus α1m. NRI progression 10.0% (95% CI −7.3 to 27.3%); NRI remission −13.9% (95% CI −31.9 to 4.2%); overall NRI −3.9% (95% CI −28.9 to 21.1%). (d) NGAL versus α1m. NRI progression 3.3% (95% CI −11.2 to 17.9%); NRI remission 0 (−16.0 to 16.0%); overall NRI 3.3% (95% CI −17.5 to 24.1%). Receiver under the operating characteristic curves of urinary biomarkers for progression and remission of idiopathic membranous nephropathy and optimal prognostic cut-off levels. KIM-1: kidney injury molecule-1; NGAL: neutrophil gelatinase-associated lipocalin; 95% CI: 95% confidence interval; ROC: receiver under the operating characteristics curve; α1m: alpha-1-microglobulin; β2m: beta-2-microglobulin.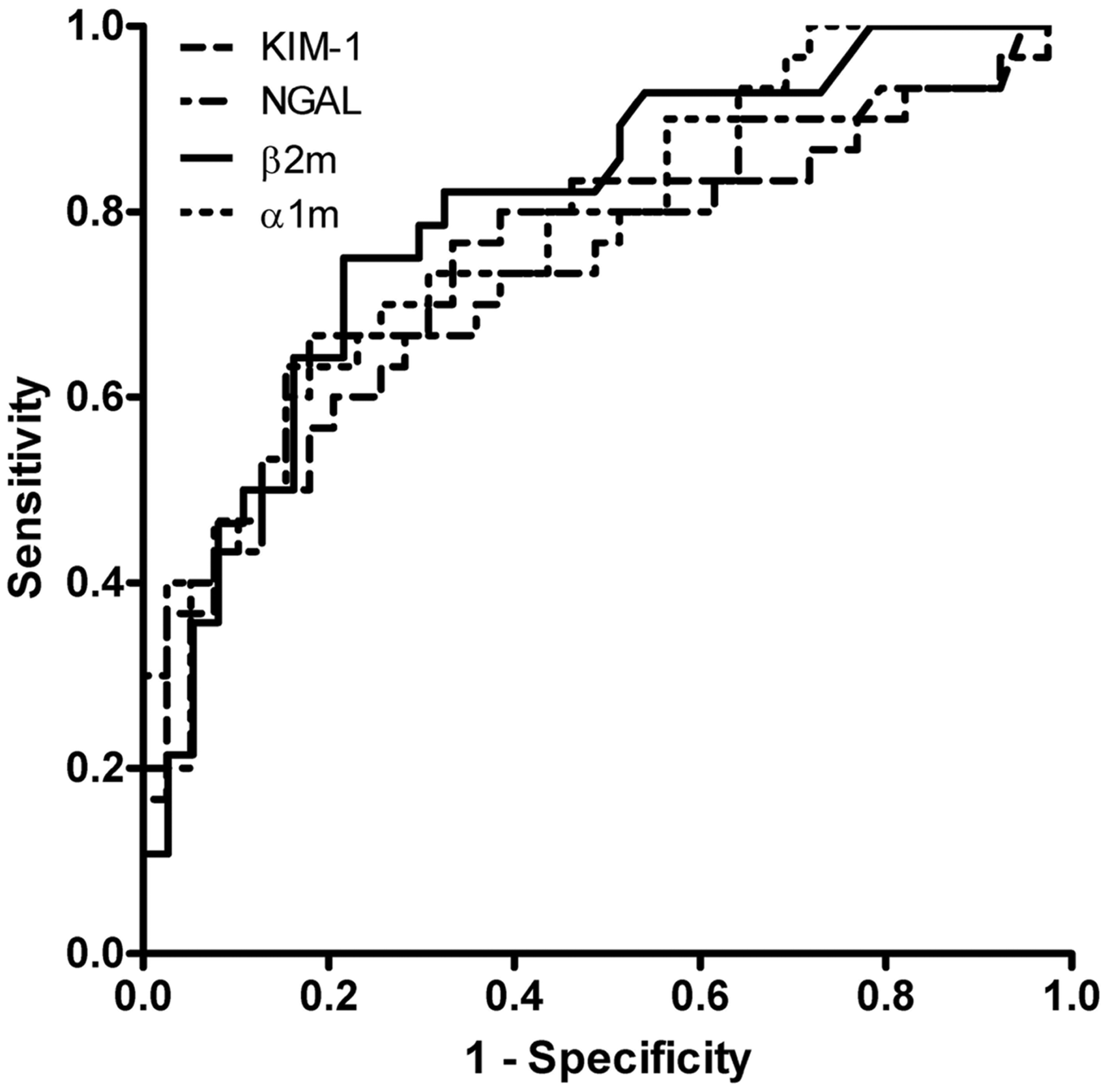
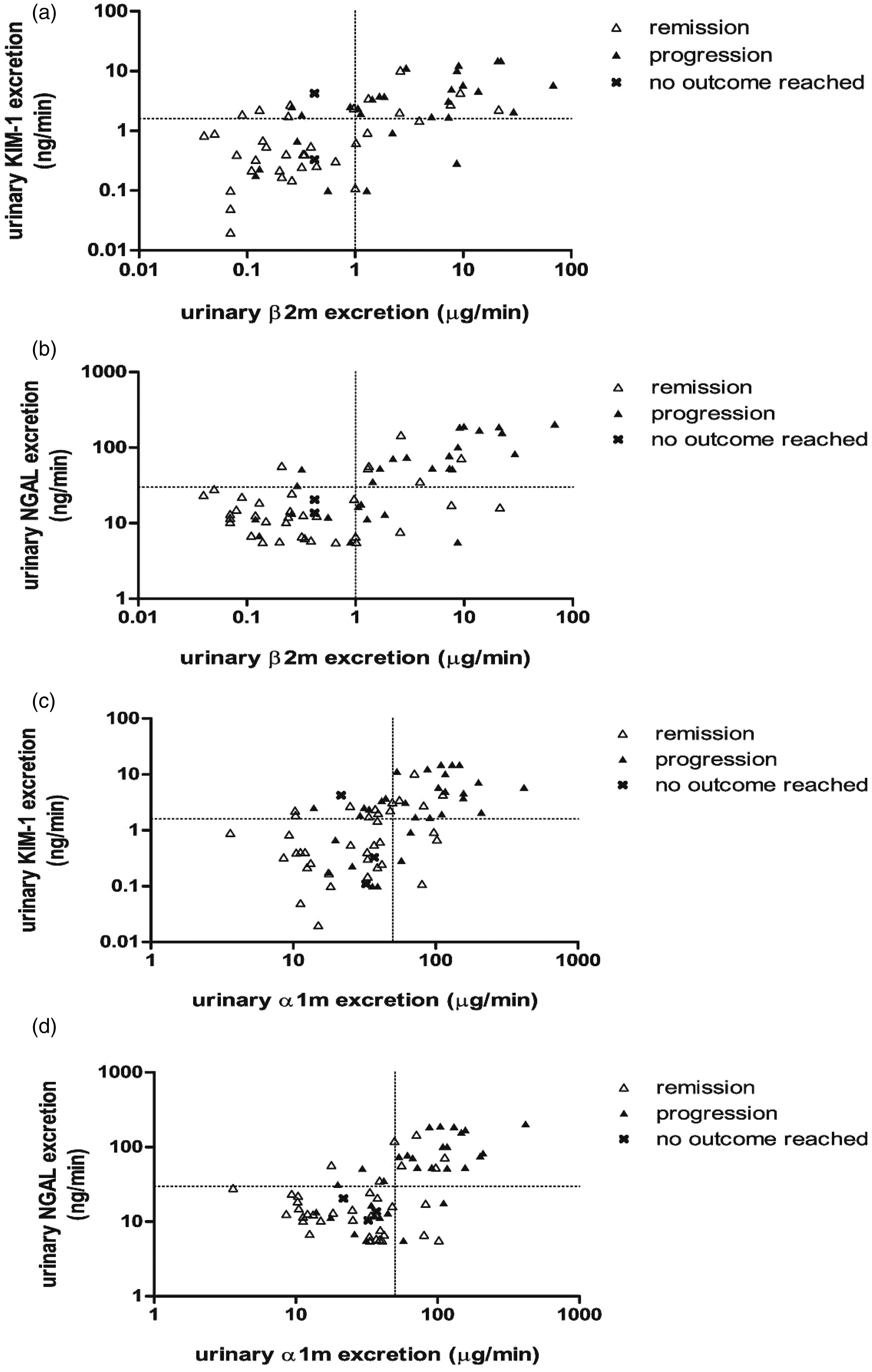
Reclassifications using KIM-1 and NGAL in patients misclassified by β2m
Prognostic misclassifications by β2m cut-off levels were studied in detail in order to detect a potential benefit of KIM-1 and NGAL in certain subgroups. Seven patients with β2m < 1.0 µg/min progressed (Supplementary Table). A common characteristic in most of these patients was performance of urinary measurement relatively early after biopsy. Three patients were reclassified using KIM-1 and two by NGAL prognostic cut-off levels as described above. Nine patients with β2m ≥ 1.0 µg/min did not progress (Supplementary Table), and all reached remissions. Four of them were correctly reclassified with NGAL and three by KIM-1 using the described cut-off values.
Discussion
Our study clearly demonstrates that the recently identified urinary biomarkers NGAL and KIM-1 predict outcome in patients with iMN and preserved renal function. However, overall they did not have additional prognostic value beyond established markers α1m and β2m.
NGAL is a low-molecular weight protein of ∼25 kDa. It is expressed on neutrophils and also on inflamed epithelia including kidney tubules.23,24 In experimental acute kidney injury, tubular NGAL expression and urinary NGAL excretion occur within 2 h, before β2m is observed in the urine. 24 Increased urinary NGAL excretion is also observed in glomerular disease. A strong correlation between proximal tubular NGAL expression and urinary NGAL has been found in patients with IgA nephropathy, suggesting that tubular cells actively produce NGAL. 25
KIM-1 is a transmembrane glycoprotein, which is not expressed in the normal kidney. It is strongly upregulated by proximal tubular epithelial cells after toxic or ischemic acute kidney injury. 26 Cleavage of KIM-1 by MAP-kinases results in urinary excretion of its 90 kDa soluble ectodomain. 27 There is a strong relationship between tubular expression and urinary excretion of KIM-1. 28 It is not produced by other cells to an extent that can account for urinary levels that are observed in patients with kidney disease. 29
Thus, urinary NGAL and KIM-1 are specific markers of tubular cell injury. In contrast, the low-molecular weight proteins α1m and β2m only reflect a reduction in tubular reabsorptive capacity. We hypothesized that KIM-1 and NGAL may be more specific markers of tubular injury and thus better predictors of outcome than α1m and β2m. Evidently, neither KIM-1 nor NGAL showed improved overall prognostic accuracy. By contrast, we have identified KIM-1 as an independent predictor of ESRD in IgA nephropathy, where α1m and β2m did not predict outcome. 17 There were strong significant correlations between the excretion rates of respective proteins in iMN, which were absent in IgA nephropathy. Tubular dysfunction may progress faster in the heavily nephrotic patients with iMN. We hypothesize that elevated NGAL and/or KIM-1 may have preceded elevated α1m and β2m in patients with progressive iMN. The data of patients with progression in the presence of β2m below prognostic cut-off support this hypothesis. Indeed, urinary measurement was performed shortly after biopsy in most of these patients. Thus, KIM-1 and NGAL may have added prognostic value in the earliest stages of iMN.
The definition for progression used in our study may be criticized. Assuming an initial normal kidney function, a rise to a serum creatinine ≥135 µmol/L indicates a serious deterioration which is associated with progression to ESRD.5,30 The other criteria for progression are in conformity with current guidelines for treatment of iMN. 31
The present cohort comprises a subgroup from a larger study that was conducted to evaluate prognostic accuracy of urinary α1m and β2m in iMN. 3 Although optimal prognostic cut-off values of urinary α1m and β2m were unchanged in this subgroup, the smaller number of patients limits the accuracy of the estimated predictive values of KIM-1 and NGAL excretion.
In conclusion, we have shown that urinary KIM-1 and NGAL are elevated in many patients with nephrotic iMN and preserved renal function. However, KIM-1 and NGAL measurement did not improve prognostic accuracy when compared or combined with established markers. Our data suggest that these markers may have added value in the earliest stages of the disease. Additional studies with longitudinal follow-up of these urinary markers are warranted to investigate their potential benefit in iMN.
Footnotes
Declaration of conflicting interests
None declared.
Funding
RJHM is supported by a grant of the Dutch Kidney Foundation (Nierstichting Nederland OW08).
Ethical approval
The study was performed under approval of the Radboud UMC Ethical Committee for the study of prognostic factors in proteinuria (CEOM:9701-0016).
Guarantor
RJHM.
Contributorship
RJHM analysed data and was the primary author of the manuscript, JAJGB collected and analysed data, FW and EM performed assays, HPP collected data and samples, JMH collected data, HG and JFMW conceived the study. All authors were involved in writing the paper and had final approval of the submitted version.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
