Abstract
Background
Thyroid-stimulating hormone (TSH) is used as the first-line test of thyroid function. Siemens Healthcare Diagnostics recommend that Siemens Centaur reagents must be protected from light in the assay information and on reagent packaging. We have compared the effect of light exposure on results using Siemens TSH-3Ultra and follicle-stimulating hormone reagents. The thyroid-stimulating hormone reagent includes fluoroscein thiocyanate whereas the follicle-stimulating hormone reagent does not.
Methods
Three levels of quality controls were analysed using SiemensTSH-3Ultra and follicle-stimulating hormone reagent packs that had been kept protected from light or exposed to light at 6-h intervals for 48 h and then at 96 h.
Results
Thyroid-stimulating hormone results were significantly lower after exposure of TSH-3Ultra reagent packs to light. Results were >15% lower at all three levels of quality control following 18 h of light exposure and continued to decrease until 96 h. There was no significant difference in follicle-stimulating hormone results whether reagents had been exposed to or protected from light.
Conclusions
Thyroid-stimulating hormone results but not follicle-stimulating hormone results are lowered after exposure of reagent packs to light. Laboratories must ensure that TSH-3Ultra reagents are not exposed to light and analyse quality control samples on every reagent pack to check that there has not been light exposure prior to delivery. The labelling on TSH-3Ultra reagent packs should reflect the significant effect of light exposure compared with the follicle-stimulating hormone reagent. We propose that the effect of light exposure on binding of fluoroscein thiocyanate to the solid phase antibody causes the falsely low results.
Keywords
Introduction
Thyroid-stimulating hormone (TSH) is used to assess thyroid function and is the first-line test of thyroid function in many laboratories. The provision of accurate and precise results is required for the assessment of patients with suspected thyroid dysfunction and for monitoring patients on treatment for thyroid disorders. The prevalence of hypothyroidism is 1–2% and it is 10 times more common in females than in males. The prevalence of hyperthyroidism is 0.5–2%. 1
The Siemens Advia Centaur TSH3-Ultra (TSH-3Ultra) assay is a third-generation non-competitive chemiluminescent immunoassay incorporating three antibodies (anti-TSH antibody labelled with acridinium ester, anti-TSH antibody labelled with fluorescein isothiocyanate [FITC] and anti-FITC antibody covalently linked to paramagnetic particles). The FITC component is used to link the TSH antibody to the solid phase. A full evaluation of the TSH3-Ultra assay has been previously performed. 2
The TSH3-Ultra assay information advises users to protect reagent packs from all heat and light sources. This is also indicated by the ‘Keep away from sunlight’  symbol on all reagent packs. All reagents are stored in a refrigerated room within our laboratory. The room is permanently lit with standard fluorescent tube lighting. As part of the investigation of an internal quality control failure for the TSH reagent (all three levels >3 standard deviations below the target TSH concentration) on two separate occasions, we determined the impact of light exposure on the TSH3-Ultra assay. For comparison, we have also investigated the impact of light exposure on the Siemens Advia Centaur non-competitive chemiluminescent immunoassay for follicle-stimulating hormone (FSH). In this assay, an anti-FSH antibody is bound to the solid phase. The FSH assay information about protection from light is the same as for the TSH-3Ultra assay.
symbol on all reagent packs. All reagents are stored in a refrigerated room within our laboratory. The room is permanently lit with standard fluorescent tube lighting. As part of the investigation of an internal quality control failure for the TSH reagent (all three levels >3 standard deviations below the target TSH concentration) on two separate occasions, we determined the impact of light exposure on the TSH3-Ultra assay. For comparison, we have also investigated the impact of light exposure on the Siemens Advia Centaur non-competitive chemiluminescent immunoassay for follicle-stimulating hormone (FSH). In this assay, an anti-FSH antibody is bound to the solid phase. The FSH assay information about protection from light is the same as for the TSH-3Ultra assay.
Materials and methods
Two TSH3-Ultra reagent packs were opened for use and put on-board the Siemens Centaur XP analyser (Siemens Healthcare Diagnostics, Munich, Germany) at 6 hourly intervals for 48 h and then at 96 h (n = 10). Both reagent packs were stored in the refrigerated room next to each other on the same shelf. Reagent pack 1 (R1-T) was kept inside the manufacturer’s packaging with the box lid closed to ensure that there was no light exposure. Reagent pack 2 (R2-T) was kept on the shelf with no packaging so was exposed to fluorescent light at all times. At each time interval, R1-T and R2-T were used to analyse the three levels of Bio-Rad Liquichek Immunoassay Quality Controls (Bio-Rad Laboratories Ltd, Hemel Hempstead, UK).
This was repeated for FSH reagent packs (Siemens Healthcare Diagnostics, Munich, Germany) defined as R1-F (protected from light) and R2-F (exposed to light). Both assays were performed according to manufacturers’ instructions.
The quality control (QC) results at each time interval for TSH and FSH at all three levels were compared with the results at baseline. The percentage differences in results compared with the baseline result were calculated (percentage difference = [(current result − baseline result)/baseline result] × 100). The CV at each level of QC was determined for R1-T, R2-T, R1-F and R2-F. Repeated measures analysis of variance (ANOVA) in Microsoft Excel was used to compare the results at each time interval using reagent packs after protection from or exposure to light for both TSH and FSH. A P value < 0.05 was used to determine statistical significance.
Results
The average TSH result (CV) for the three levels of QC using the unexposed reagent (R1-T) were 0.73 (1.71%), 4.94 (1.81%) and 26.05 (2.22%) mU/L, respectively. The maximum percentage difference from the baseline result during the 96 h was −4.05%, 3.22% and 5.02% for levels 1, 2 and 3, respectively. In contrast, the TSH CVs for the three levels of QC analysed using the exposed reagent (R2-T) were 19.90%, 20.71% and 20.03%, respectively. Following 18 h of light exposure, the results at all three QC levels were >15% lower than the baseline result and by 42 h, the results at all three QC levels were >30% lower than the baseline result (see Figure 1(a)). The results after light exposure were significantly different for all three levels of QC (P value ≤ 0.001 for all).
(a) Percentage difference in TSH results on Quality Control levels 1, 2 and 3 using reagent protected from light (R1-T, solid line) and reagent not protected from light (R2-T, dashed line). (b) Percentage difference in FSH results on quality control levels 1, 2 and 3 using reagent protected from light (R1-F, solid line) and reagent not protected from light (R2-F, dashed line).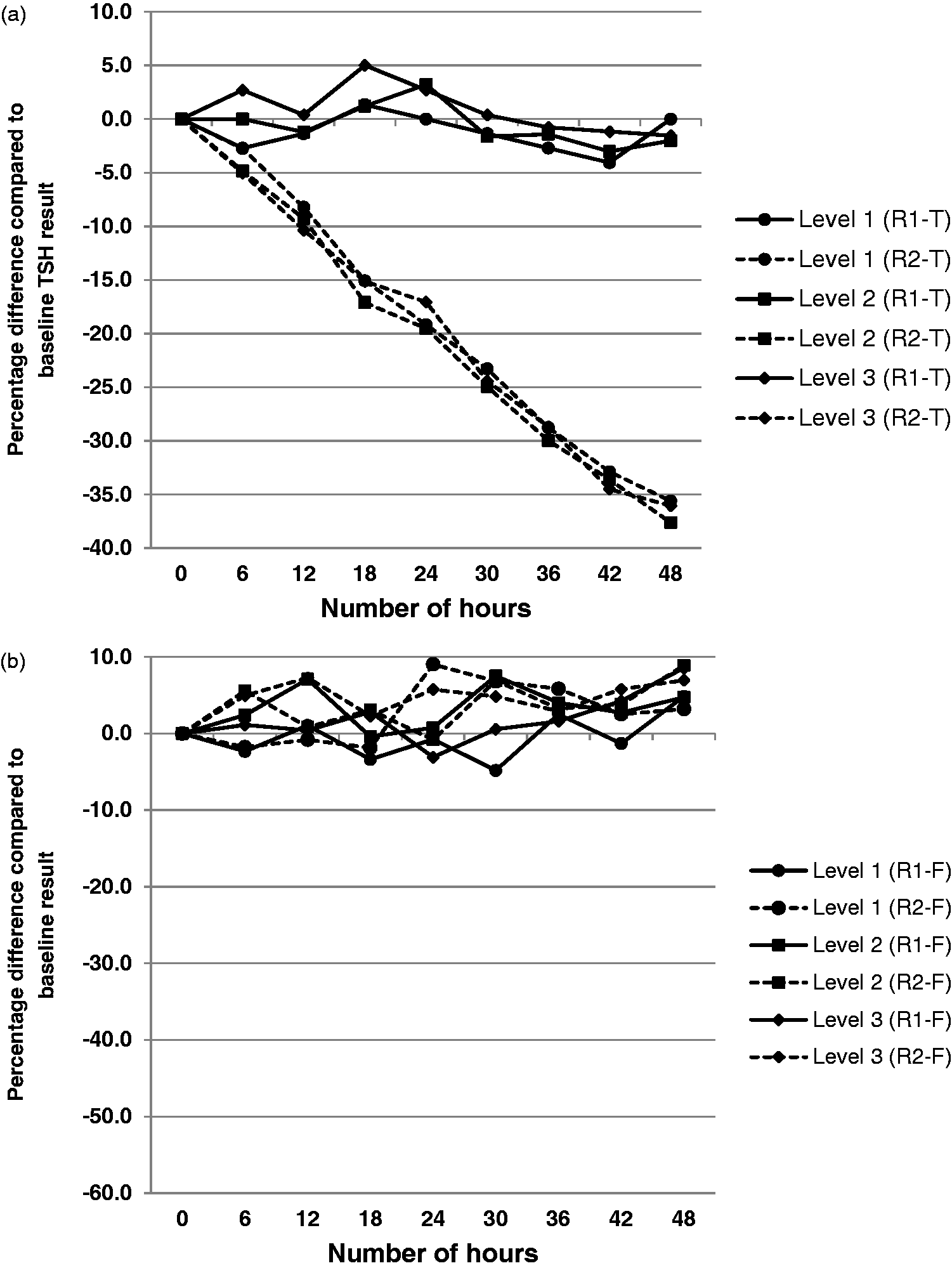
The average FSH results (CV) for the three levels of QC using the unexposed reagent (R1-F) were 7.20 (3.96%), 17.25 (2.65%) and 50.21 (3.02%) IU/L, respectively. The maximum percentage difference from the baseline result during the 96 h was −4.81%, 7.51% and 8.55% for levels 1, 2 and 3, respectively. The FSH CVs for the three levels of exposed reagent (R2-F) were 3.67%, 3.18% and 2.15%, respectively. The maximum percentage difference from the baseline result during the 96 h was 9.04%, 8.88% and 6.97% for levels 1, 2 and 3, respectively (see Figure 1(b)). The results were not statistically different following light exposure (P value = 0.37, 0.29 and 0.80, respectively).
Discussion
The CVs of all three levels of QC using the TSH-3Ultra reagent not exposed to light were similar to those reported by Reix et al. 2 (<4.4%). We have found a significant decrease in TSH results following exposure to fluorescent light. This decrease was apparent after 6 h of fluorescent light exposure and continued for the 96 h by which time the decrease in results was >50% at all three levels of QC.
The FSH QC results were not significantly different whether the reagent had been protected from light or exposed to light. However, the symbol on the FSH packaging and the information about protecting from heat and light sources in the FSH assay information are identical to the TSH-3Ultra reagent. Additional labelling on the TSH-3Ultra reagent about the significant effect of light exposure on results would be helpful. We would also suggest that the manufacturer should consider storing the FITC component of the reagent pack in an opaque section to ensure that there is no light exposure from manufacture through to routine laboratory use.
A key difference between the TSH-3Ultra and FSH reagents is the FITC component in the TSH-3Ultra reagent. In both assays, the chemiluminescent reaction rather than FITC is generating the light signal. Therefore, the lower TSH results after exposure to fluorescent light are not caused directly by photo bleaching of FITC. Instead, the effect of light exposure on FITC is indirectly causing a decrease in the chemiluminscent signal in the TSH assay. A possible mechanism is that the exposure of FITC to light alters the binding affinity of FITC to the anti-FITC antibody that is bound to the solid phase. After washing, TSH bound to FITC that is not attached to the solid phase (via anti-FITC) will be removed, leading to reduced chemiluminescent signal and lower results. Further work would be required to determine if the FITC component is the cause of the lower results following light exposure and if this is related to loss of antigenicity; in the first instance, a TSH assay where the FITC/anti-FITC solid phase has been replaced with biotin-streptavidin could be used to demonstrate that it is the FITC component causing this decrease in TSH results following light exposure. 3
The significantly lower TSH results reported on patients following exposure of TSH-3Ultra reagent to light could lead to altered diagnosis or management of thyroid dysfunction. Many laboratories keep their reagents (often out of the original packaging) in refrigerated rooms where they may be exposed to light for long time periods. Following this work, we changed the lighting in our refrigerated room so it has to be switched on/off and we ensure that our reagent packs are kept in the closed manufacturer’s packaging. We would advise other laboratories using TSH-3Ultra reagent to review their storage procedures. In addition, we now routinely perform IQC on every TSH3-Ultra reagent pack to check for light exposure prior to delivery. The purpose of this report is to highlight to users the dramatic effect of light on TSH results.
Footnotes
Acknowledgments
The authors would like to thank Blood Sciences staff at the Royal Blackburn Hospital who assisted with the analysis of samples overnight.
Declaration of competing interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Ethical approval
None applicable.
Guarantor
JA.
Contributorship
IL and JA designed the study and analysed the data. IL and DG were involved in sample analysis. JA wrote the first draft of the manuscript. All authors edited, reviewed and approved the final manuscript.
