Abstract
Introduction
The thyroid hormones T4 (thyroxine) and T3 (tri-iodothyronine) are secreted by the thyroid gland in response to pituitary thyroid-stimulating hormone (TSH) and are responsible for maintaining the basal metabolic rate of the body. Hypothyroidism is a deficiency of thyroid hormones, which can be congenital or acquired. It leads to a diverse array of clinical signs and symptoms including fatigue, cold intolerance, weight gain and constipation. 1 First-line testing for hypothyroidism is based on measuring TSH, which will identify cases of primary hypothyroidism, that is, defect in thyroid hormone synthesis or secretion. Treatment of hypothyroidism with thyroxine replacement leads to symptomatic relief in the majority of patients, but life-long monitoring of TSH is required to ensure the correct dose is being administered.2,3 The measurement of TSH is therefore essential to both the diagnosis and management of hypothyroidism.
Congenital hypothyroidism (CHT) has an incidence of 0.53 per 1000 live births in the United Kingdom, with approximately 90% of cases caused by primary thyroid disorders.4,5 Screening for CHT was first introduced in England and Wales in 1981 and is currently offered to all newborn babies in the United Kingdom on day 5 of life (with samples collected up to day 8 being acceptable). 6 The screening strategy is based on measuring TSH in a dried blood spot (DBS) sample, with all suspected cases referred for further diagnostic investigations. DBS are formed by collecting blood from a heel prick onto a filter paper collection device (Guthrie card). To allow correct interpretation of results, strict sampling guidelines are in place; key requirements are a single drop of blood that evenly saturates the guide circle and soaks through to the back of the card. 7 In South West Thames Newborn Screening laboratory, DBS TSH is measured using the PerkinElmer Neonatal hTSH kit on the Genetic Screening Processor (GSP) instrument. As this is a screening test, ‘cut-offs’ are used rather than reference intervals, with a day 5 result of ≥20 mU/L, or a borderline repeat result of ≥8 mU/L, referred for diagnostic investigations. 6 The identification and timely treatment of CHT mitigates the intellectual disability that can otherwise be caused by the disorder. 8
Acquired primary hypothyroidism is a common disorder that increases in prevalence with age, especially after the middle-age period, and occurs more frequently in women than men. 3 The incidence of acquired hypothyroidism in Tayside, Scotland, was 4.75 per 1000 for women and 1.09 per 1000 for men based on data from 2001. 9 Primary hypothyroidism is far more common than secondary, with a ratio of approximately 1000–1. 1 Acquired hypothyroidism is investigated by measuring serum TSH, which requires venepuncture for sample collection. The local upper limit of normal for TSH is 5.0 mU/L, but current guidelines from NICE only recommend treating primary hypothyroidism with levothyroxine when serum TSH is consistently above 10 mU/L. 2
There are therefore differences in the samples used for screening for CHT and investigating acquired hypothyroidism when measuring TSH. Although not offered as a routine service, the South West Thames Newborn Screening laboratory and other newborn screening laboratories frequently receive DBS samples for TSH on adults who are needle phobic and cannot tolerate venepuncture. If the DBS fulfils the acceptance criteria, the TSH concentration can be measured and reported. However, to our knowledge, there is no established method-specific reference interval for DBS TSH in adults, so the results are difficult to interpret. Additionally, comparison of DBS TSH with serum results in adults when using the GSP analyser and routine laboratory platforms, for example, Abbott Architect, has not been reported before. An established reference range will also benefit patients who have problems in accessing local phlebotomy services due to personal or public health reasons, such as mobility issues or self-isolation.
The aim of this study was therefore to establish an adult DBS TSH reference interval for the PerkinElmer GSP analyser using the Neonatal hTSH kit and compare the DBS results to serum TSH. An additional aim of this study was to determine the reference interval for day 5–8 DBS TSH as this has also not been published using the GSP analyser before.
Methods
Ethical approval
This study performed experiments on redundant samples previously received by the laboratory. No tests were performed that had not already been requested and all confidential data was anonymised. For these reasons, approval from a Research Ethics Committee was not required.
Selection of lithium heparin samples for DBS TSH analysis
The Blood Sciences department frequently receives serum, EDTA and lithium heparin samples collected from the same patient, at the same time. This most often occur with requests made from GP practices. The PerkinElmer GSP Instrument used for DBS TSH analysis is compatible with whole blood collected directly from a patient onto filter paper (i.e. newborn screening) or whole blood collected into EDTA or lithium heparin tubes, then spotted onto filter paper. Although EDTA samples can be used for TSH analysis, we chose to use lithium heparin samples as EDTA is known to cause interference in the immunoreactive trypsinogen assay, which is also run on the instrument. We collected all extra lithium heparin tubes (LH BD vacutainer) that had been received on adult patients (≥18 years old) on a daily basis over a 2-month period (August–September 2020). Using our laboratory information management system (LIMS), Telepath, the following criteria were applied to leave only those samples that were suitable for DBS preparation. 1. The lithium heparin sample had to have been received with a paired serum sample that had TFTs requested and a paired EDTA sample that had a haematocrit requested. 2. TFT results on the serum sample had to be within the reference interval. The laboratory currently uses TSH and FT4 as first-line TFTs with reference intervals of 0.35–5.00 mU/L and 9–22 pmol/L, respectively. 3. Based on the information provided on the electronic request and that stored in our LIMS, the patients were not known to be taking levothyroxine, carbimazole or propylthiouracyl (PTU). 4. The haematocrit on the EDTA whole blood sample had to be within the sex-specific reference interval; male: 0.40–0.50 L/L and female: 0.36–0.46 L/L.
Preparation and analysis of lithium heparin DBS TSH
Bloods spots were prepared from the lithium heparin samples on the same day of sample collection. The tubes were inverted several times to ensure that the whole blood was homogenous before 50 µL of the sample was spotted onto PerkinElmer 226 filter paper (Lot 0,120,201/1,001,268) with a 200 µL calibrated pipette; this was performed consistently by a single individual. We chose 50 µL as this volume has been shown to produce fully penetrating DBS with an approximate diameter of 12 mm, which fills the printed guide circles (∼11 mm in diameter) on UK newborn screening DBS cards. 10 PerkinElmer 226 filter paper was chosen as it is used for newborn screening by all four UK countries and is compliant with Clinical and Laboratory Standards Institute (CLSI) NBS01-A6 specifications. 10 The DBS were left at room temperature overnight to dry and the following morning placed in a sealed bag with desiccant and stored in a fridge at 4°C for up to 7 days before analysis. A previous study has shown that TSH is stable in heparinised DBS for at least 60 days when stored at 4°C. 11
A single, peripheral, 3.2 mm diameter punch was made using the PerkinElmer Panthera-Puncher 9 Instrument. Thyroid-stimulating hormone was measured in the punches using the PerkinElmer time-resolved fluoroimmunoassay Neonatal hTSH kit on the PerkinElmer GSP instrument, according to the manufacturer’s instructions. A single kit lot (680,993) was used for the entirety of the study. Six DBS calibrators, with a TSH range of 0.9–297 mU/L and a haematocrit of 50%–55%, were provided in the kit and used for calibration. The DBS calibrators are standardised to the WHO 3rd International Standard (81/565) for TSH. The assay has an inter-assay imprecision coefficient of variation (CV) of 7.4% at a TSH concentration of 15 mU/L. An in-house internal quality control (IQC) DBS sample and two levels of kit IQC material (lot 679,963) were run in duplicate with every 96-well plate, with mean TSH concentrations of 6.1 mU/L (CV 7.5%), 17.1 mU/L (CV 7.2%) and 72.3 mU/L (CV 8.5%), respectively. The manufacturer reported limit of detection (LoD) and limit of quantification (LoQ; lowest TSH concentration with a CV of <20%) for the assay are both 1.31 mU/L, but the analyser is capable of reporting below this value.
Imprecision around the lower limit of reporting for the DBS TSH assay
Imprecision around the lower detection limit of the hTSH kit on the PerkinElmer GSP instrument was investigated by running 2 DBS samples with TSH concentrations of approximately 0.5 and 3.0 mU/L. These DBS were prepared from lithium heparin whole blood samples in the same way as described above. Thirty replicates were generated over 8 separate runs for each sample to determine the imprecision of the assay.
Analysis of serum TFTs and whole blood haematocrit
Blood samples for serum TSH and FT4 collected into BD vacutainer SST II Advance tubes were centrifuged at 2671 RCF for 7 min at room temperature using a Hettich Rotanta 460 R centrifuge to isolate the serum. Thyroid-stimulating hormone and FT4 concentrations were measured on Abbott Architect i2000 immunoassay analysers. Thyroid-stimulating hormone was measured using a sandwich immunoassay with an inter-assay imprecision CV of 2.0% at 2.0 mU/L. Two calibrators with TSH concentrations of 0 and 40 IU/L were used to calibrate the assay. The calibrators are traceable to the WHO 2nd International Standard (80/558) for TSH. FT4 was measured by a competitive immunoassay with an inter-assay imprecision CV of 4.4% at 16.1 pmol/L. Blood samples for haematocrit analysis were collected into BD vacutainer K2E tubes containing K2-EDTA. Measurement of the haematocrit was performed using a Sysmex XE-5000 analyser with an inter-assay imprecision CV of 1.5%.
Determination of day 5–8 DBS TSH reference interval
UK screening standards state that ≥90% of samples should be collected on day 5, but samples taken up to day 8, are acceptable. 12 We frequently receive DBS samples from day 6 to 8, so to reflect this reality, we included day 5–8 DBS samples in our reference interval. We used our LIMS to collate all day 5–8 (including preterm samples) newborn DBS screening samples that had a TSH measured using the Neonatal hTSH kit lot 680,993, the same used for the adult experiments. This kit lot was in use for routine screening from the 19/08/20 to the 22/10/20. This data was filtered to remove screened positive results (≥20 mU/L on a first card, or a borderline result (≥8 to <20 mU/L) that was subsequently found to be positive (≥8 mU/L) on a follow-up sample), so only ‘euthyroid’ samples were included. The reference range was derived from this trimmed data. Unlike the adult DBS TSH reference interval, we could not fully control the size of the spots that were included in the day 5–8 reference interval. The minimum acceptance criteria for a newborn DBS is ≥7 mm, 10 so a range of sizes above this would have been included.
Statistics
Statistical analysis and graphing was performed using Microsoft Excel and the Analyse-it software add-on (Version 2.30, Analyse-it software Ltd., Leeds, UK). Normal distribution of data was assessed using the Shapiro–Wilk test. Data with a normal distribution is described as the mean and standard deviation (SD) and non-normal as the median and interquartile range (IQR). When data with a normal distribution is compared to non-normal data, the median and IQR are used. The parametric Student’s t-test was used to determine if two groups with a normal distribution were from the same population. If one or both groups had a non-normal distribution, the non-parametric Mann–Whitney U test was used. Passing–Bablok regression was used to compare the serum and DBS TSH results. Reference intervals were calculated from normally distributed data using the parametric (mean ± 1.96 × SD) method to encompass the central 95% of the distribution. Non-normally distributed data was transformed (log10 or square root) and if the data was normally distributed the parametric reference interval was determined then back transformed. 13 If normality was not achieved after transformation, the non-parametric 2.5th – 97.5th percentile method for reference intervals was used. 13 For all statistical tests, p < 0.05 was used to reject the null hypothesis.
Results
Adult sample demographics
Age distribution and lab results for male and female samples.
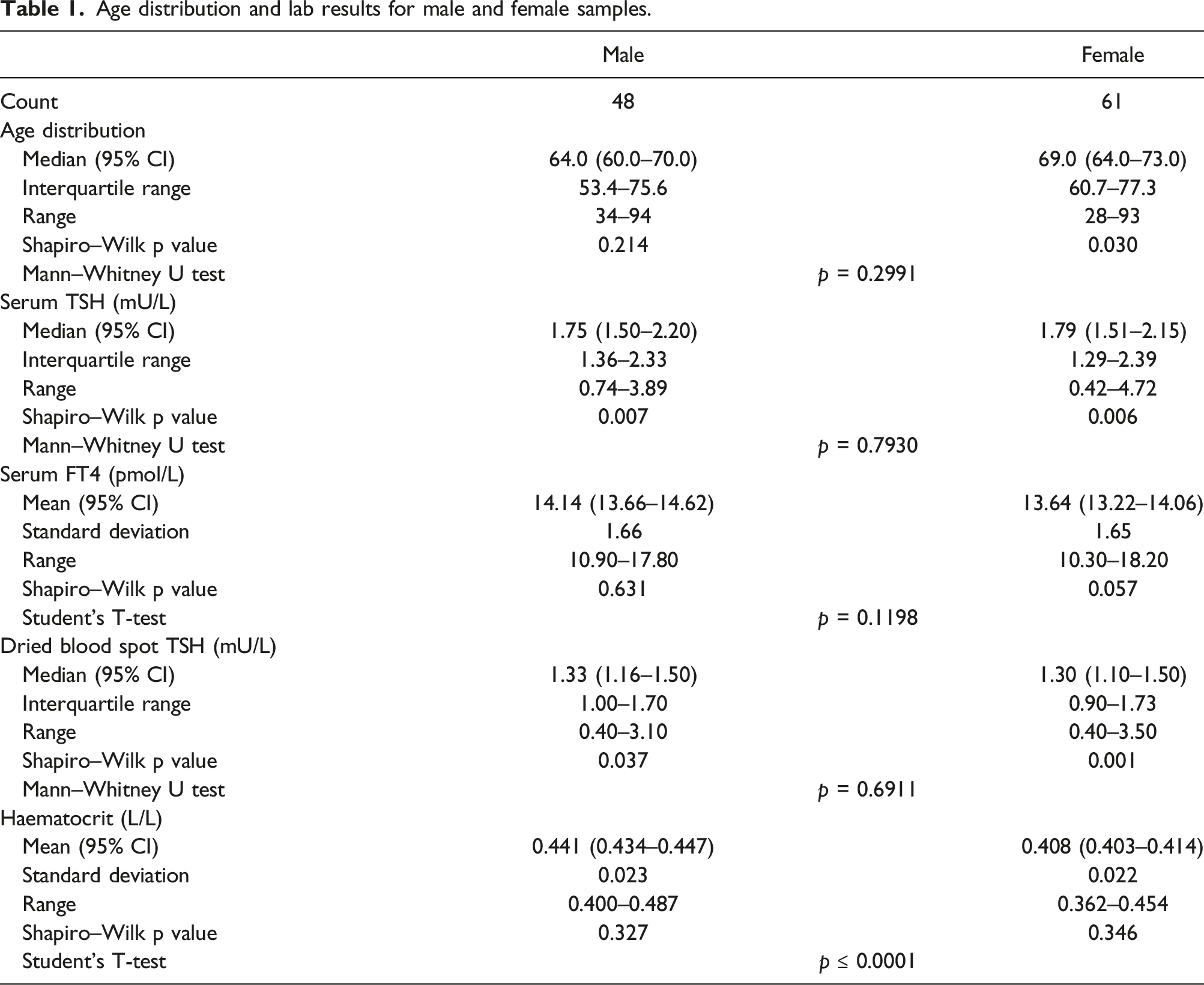
Comparison of serum and DBS TSH
When the male and female samples were combined the Passing–Bablok linear equation was: DBS TSH = 0.68 × serum TSH + 0.07 (Figure 1(a)). As the haematocrit is known to influence DBS TSH concentrations, the male and female samples were also analysed separately. Passing–Bablok regression for the male samples showed a linear equation of: DBS TSH = 0.70 × serum TSH + 0.02 (Figure 1(b)), and the female: DBS TSH = 0.68 × serum TSH + 0.09 (Figure 1(c)). Passing–Bablok regression for serum versus dried blood spot thyroid-stimulating hormone for (a) male and female samples combined, (b) male samples and (c) female samples.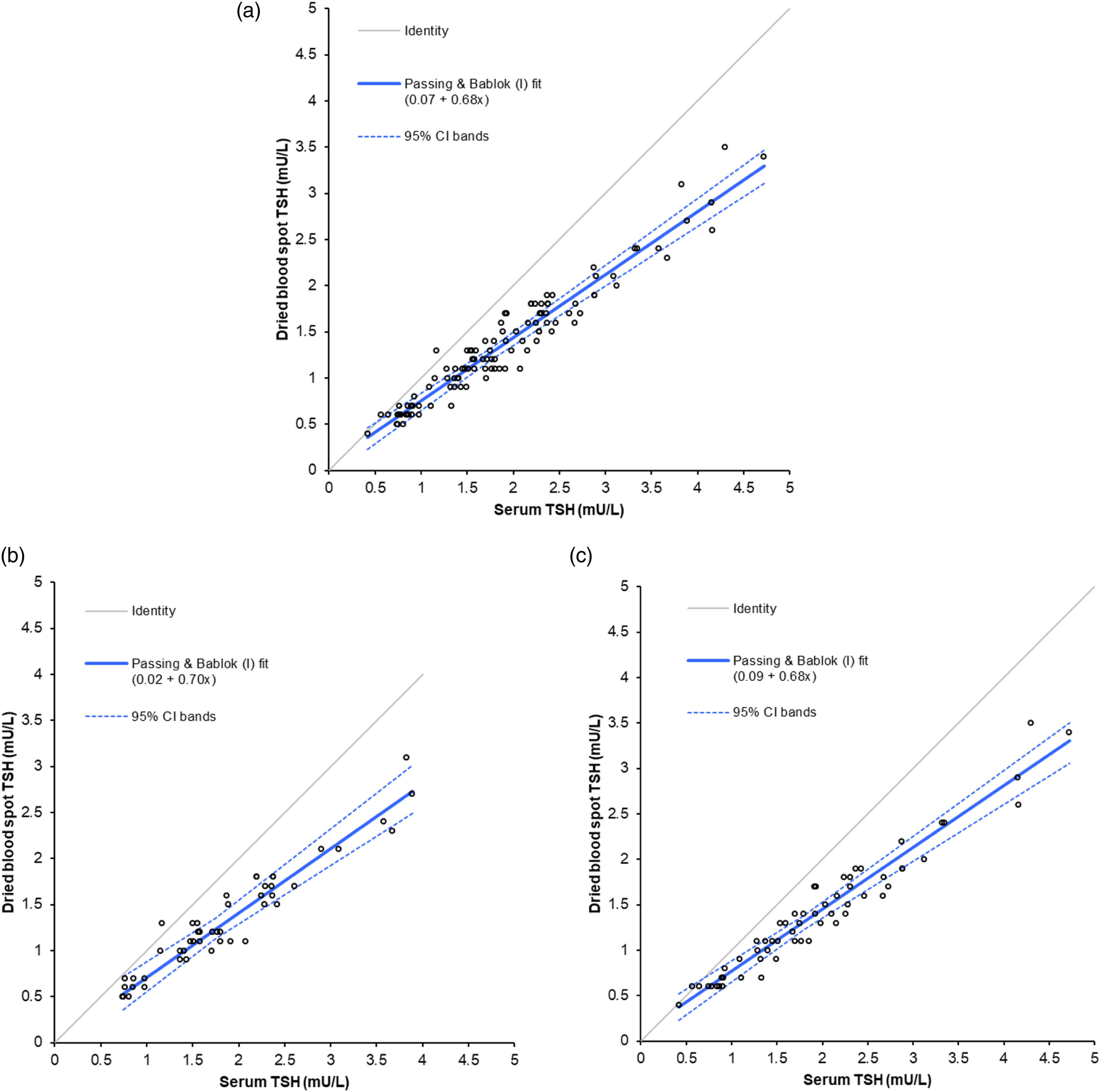
Adult DBS TSH reference interval
The combined male and female DBS TSH data was non-normally distributed (p = < 0.0001) but had a normal distribution when log10 transformed (p = 0.089); the parametric reference interval of the back transformed data was 0.49 (95% CI: 0.43–0.57) to 3.07 (95% CI: 2.64–3.57) mU/L (Figure 2(a)). Both the male and female separated DBS TSH data were also non-normally distributed (p = 0.037 and p = 0.001, respectively) but had normal distributions when log10 transformed (p = 0.4175 and p = 0.2398, respectively). The parametric reference interval of the male back transformed data was 0.49 (95% CI: 0.39–0.61) to 2.97 (95% CI: 2.38–3.71) mU/L (Figure 2(b)) and the female data was 0.48 (95% CI: 0.39–0.60) to 3.21 (95% CI: 2.61–3.95) mU/L (Figure 2(c)). Dried blood spot thyroid-stimulating hormone reference intervals derived from (a) male and female samples combined, (b) male samples and (c) female samples.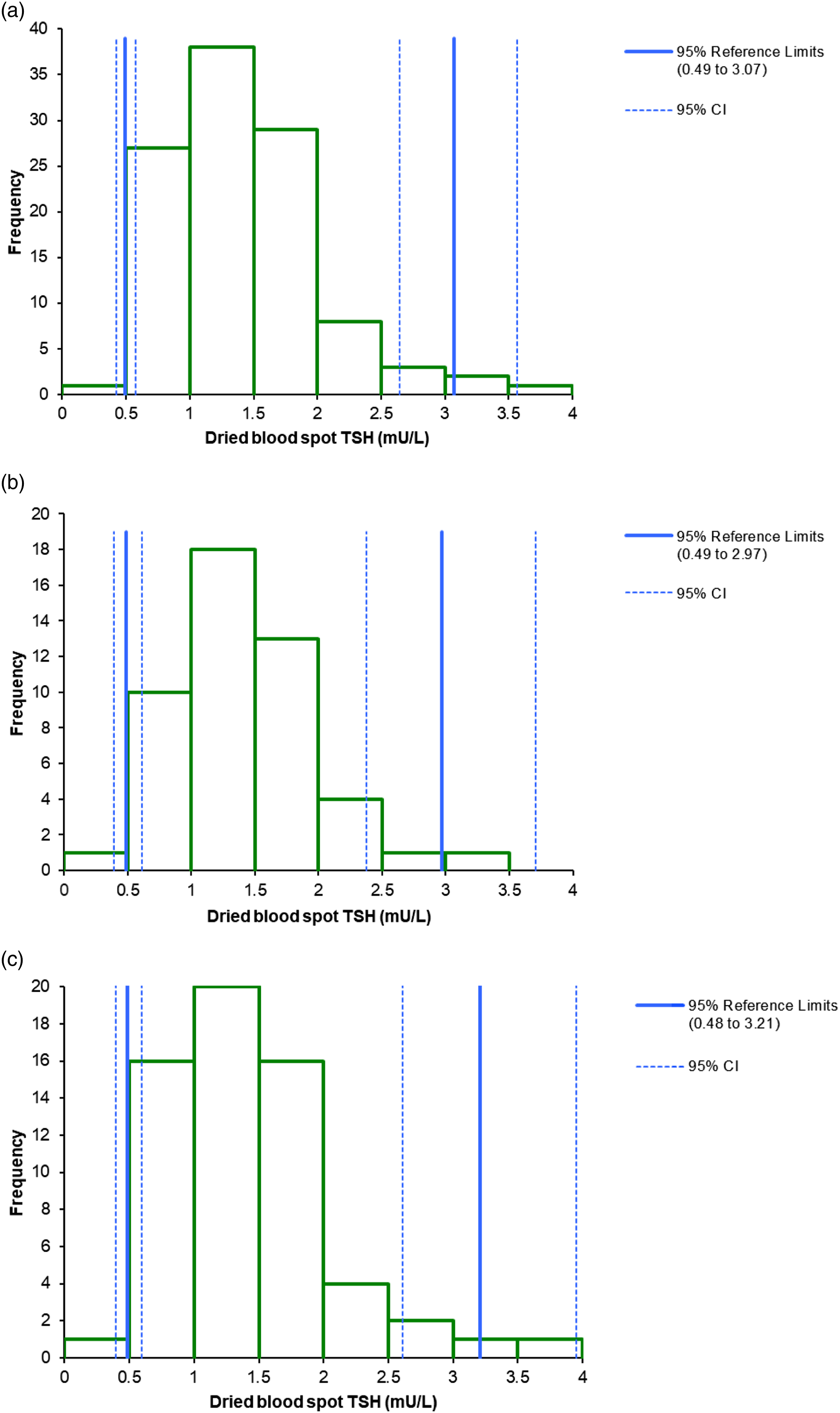
Day 5–8 DBS TSH reference interval
A total of 8356 day 5–8 DBS samples were measured for TSH. After screened positive results were removed, 8351 samples remained, 7212 from day 5, 839 from day 6, 200 from day 7 and 100 from day 8. The distribution of TSH results in the sample was non-normally distributed (p = <0.0001) and stayed that way after both log10 and square root transformations. The non-parametric 2.5th – 97.5th percentile reference interval for the data was 0.40 (95% CI: 0.40–0.50) to 4.10 (95% CI: 4.00–4.30) mU/L (Figure 3). Dried bloodspot thyroid-stimulating hormone reference interval derived from day 5–8 samples.
Imprecision of the DBS hTSH assay at the lower limit of reporting
The PerkinElmer hTSH assay had a CV of 20.9% at a mean TSH concentration of 0.6 mU/L and a 10.1% CV at 2.8 mU/L.
Discussion
The purpose of this study was to establish method-specific TSH reference intervals for adults and day 5–8 neonates using the Neonatal hTSH kit on the GSP analyser.
Our findings are important as they permit healthcare professionals to better understand how to interpret DBS TSH results in both adults and neonates by providing a method-specific ‘normal’ range. Knowing the method-specific relationship between DBS and serum TSH is also valuable as it provides a means of comparing the two sample types. This is particularly useful for referred cases of CHT where initially only a DBS TSH result is known.
TSH reference intervals are fundamental to interpreting TSH results and there has been a long history in establishing the serum TSH interval. 14 To our knowledge, this is the first study to directly determine a DBS TSH reference interval in adults using any method. Fundamental to any reference interval is the establishment of a reference population, who must be healthy and free from disease that may influence the analyte of interest. The British Thyroid Association recommend (expert opinion) that the reference population should have no personal or family history of thyroid dysfunction, be on no medication known to alter TSH and have no thyroid antibodies (TPO and TSH receptor) detectable. 15 All the adults that were included in our reference population were euthyroid, as judged by serum TSH and FT4 results, and were not on any medication that directly influences thyroid function (levothyroxine, carbimazole or PTU). However, we did not screen our patients further for family history or for other medications known to alter TSH (e.g. dopamine and glucocorticoids). Additionally, we did not add on tests that had not already been requested so we were unable to check thyroid antibody status. According to the CLSI guidelines on reference intervals, a sample size of 120 is ideal for establishing a reference interval. 16 Unfortunately, this was not possible for our study due to the limited number of appropriate samples received in the experimental time-frame. We therefore recognise that there are limitations in the establishment of our reference intervals, but due to the reason explained above, they could not be overcome.
Due to the above mentioned limitations in the reference population, we verified the reference interval using the relationship between serum and DBS TSH. Our established serum TSH reference interval is 0.35–5.00 mU/L and when this is converted to DBS TSH using our sex-specific linear regression fits, the following DBS reference intervals are obtained: male 0.27–3.52 mU/L, female 0.33–3.49 mU/L and male and female combined 0.31–3.47 mU/L. These ranges are similar to the intervals obtained with our adult reference population and show that, although our population was not perfect, it does provide a reasonably robust interval.
We determined DBS reference intervals in adult males and females, both individually and combined. The reason we generated sex-specific intervals is that males and females have different (albeit overlapping) reference intervals for haematocrits and the haematocrit is known to affect the size of a DBS and how it spreads across filter paper. 17 We chose to only include patients with a haematocrit in the normal range as Lawson et al. 2016 has demonstrated that when the haematocrit changes from a normal value of 0.500 to an abnormal value of 0.300, the DBS TSH concentration increases by ∼15%. 18 The reference intervals we have derived for DBS TSH therefore only apply to adults that have a haematocrit within the normal interval. This may be seen as a downside as the haematocrit of a DBS is normally unknown, but a simple method has been published recently that describes how the haematocrit can be measured in a DBS. 19 Only including samples with a normal haematocrit and providing sex-specific intervals is a strength of our study as it will reduce the effect that the haematocrit has on the TSH value. However, this may also be seen as a weakness as it limits the use of the reference interval only to those with normal haematocrits.
We found that DBS TSH concentrations were approximately 30% lower than serum. This is in contrast to two similar studies that used the PerkinElmer AutoDelfia instrument (the predecessor to the GSP instrument). The first study by Hofman et al. 2003 found DBS TSH to be approximately 40% lower than serum TSH when measured using the Abbott AxSYM instrument. 20 The study by Evans et al. 2011 showed that DBS TSH is approximately 50% lower than plasma TSH measured using the same Abbott Architect assay we used in our study. 21 This difference in results may be explained by a number of factors and highlights the challenges when trying to establish a DBS reference interval for TSH. Firstly, the AutoDelfia and GSP instruments are not directly comparable when measuring DBS TSH, as previous studies have shown that the GSP assay has a negative bias of around 10%–20%, when compared to the AutoDelfia assay.22,23 Secondly, serum/plasma TSH assays are not always comparable across different platforms and the level of comparability also changes over time with the release of new reagent and calibrator lots. Thirdly, the haematocrit of the blood (as discussed above), the volume of blood used and the punch location, all influence the final DBS TSH result. As explained in the methods section, we chose to use 50 µL of whole blood to make the DBS in our study as this fills the 11 mm guide circles on UK newborn screening DBS cards. 10 Hofman et al. 2003 also used this volume, 20 whereas Evans et al. 2011 used 25 µL. 21 A 25 µL DBS has a diameter of <11 mm 10 and it has been shown previously that the smaller the blood spot, the lower the TSH concentration,10,18,24 which may account for some of the difference between our reference intervals. Peripheral punches, as opposed to central, have also been shown to produce higher TSH results, ranging from around 5% to 30%.10,18,24 For this reason, punches were consistently taken from peripheral locations in our current study as it has been recommended that the initial punch should be from the peripheral and then repeats from central location. 24 Neither the Hofman et al. 2003 nor the Evans et al. 2011 studies specified the punch locations or checked the haematocrit of the samples they included.20,21 All of these factors combined will therefore have led to differences between the studies. We believe that our consistent approach to producing DBS is a strength of our study and has helped to minimise many of the variables that are associated with this sample type.
Our study has therefore produced an adult DBS reference interval but a number of caveats must be applied when interpreting patient results. Firstly, the DBS must fill the guide circle on the filter paper and be collected as recommended by the newborn DBS screening programme. 7 This is dependent on the person taking the sample and is therefore outside the control of the laboratory. Secondly, it only applies to patients with a normal sex-specific reference interval for their haematocrit, but it is possible for this to be checked by the lab (see above). Thirdly, the punch must be taken from a peripheral location, which can be easily controlled by the lab. Finally, we have established an ‘adult’ TSH reference interval, but evidence shows that the upper limit of normal for TSH increases with ageing, 25 so a single reference interval may not be appropriate, but this is an issue with all adult TSH reference intervals in use. Another consideration is that the lower end of our derived reference intervals are below the lowest calibrator value (0.9 mU/L) and the LoD/LoQ (1.31 mU/L) of the assay. Imprecision around these limits were investigated using in-house IQC samples with mean TSH DBS concentrations of 0.6 and 2.8 mU/L and showed CVs of 20.9% and 10.1%, respectively. This shows that the upper reference interval of the assay has acceptable precision but the lower limit of the reference interval is just below what is deemed acceptable for the assay (a CV of <20%). Therefore the assay is only suitable to detect abnormally high concentrations of TSH, rather than low, so should only be used to investigate possible cases of primary hypothyroidism. Additionally, no IQC samples with values around the adult reference interval are routinely run, so if this assay is to be used for the purpose of detecting primary hypothyroidism in adults, appropriate control material will need to be prepared.
The concept of using DBS TSH to investigate hypothyroidism outside of the newborn screening field has been investigated and published previously. Unlike our study, these publications did not use the GSP instrument and did not derive a DBS reference range, but rather came up with positive cut-off values. The first study from 2000 used a TSH cut-off of 10 mU/L as this was the LoD of their immunoradiometric assay. 26 They screened a population of school children with Down’s syndrome aged 4–19 years of age and identified 15 children with raised DBS TSH, with all cases being confirmed by serum TSH analysis using an Abbott AxSYM analyser. A second study published in 2003 using the AutoDELFIA instrument derived a cut-off of 4.6 mU/L based on the serum TSH upper limit of normal when using the Bayer Technicon assay and the linear regression between serum and DBS TSH. 20 They identified 19 adult patients with elevated DBS TSH, 16 of whom also had a raised serum TSH. It should be noted that the follow-up serum sample was taken up to a month after the DBS sample, so this might explain why three patients did not have concordant elevated serum TSH concentrations. Both of these studies therefore support the use of DBS TSH for detecting primary hypothyroidism in adults and children. However, a study by Takáts et al. 2000 reported that a high diagnostic sensitivity of 94% for DBS TSH could only be achieved at the expense of a poor specificity of 72%. 27 Future work for us would therefore be to perform a study evaluating the diagnostic potential of the DBS TSH reference interval.
As well as establishing adult DBS TSH reference intervals, we derived a reference interval for day 5–8 samples. This has been done previously using the AutoDelfia instrument, 21 but not with the GSP instrument. We found our 97.5th percentile to be 4.10 mU/L for day 5–8 samples, which is higher than the AutoDelfia study which found it to be 3.04 mU/L. The cause of this difference is likely related to those explained above including the size of the spots, punch location and haematocrit of the blood. Knowing the 97.5th percentile for day 5 samples is useful as it provides a means of knowing what a ‘normal’ DBS result for TSH is and provides a way of gauging how abnormal an elevated day 5 DBS TSH result is.
In conclusion, our study derived sex-specific, and combined, reference intervals for DBS TSH in adults, which is essential information when interpreting results. For some patients, taking a serum sample for TSH determination is not always possible, so DBS TSH may be the only alternative. The reference ranges established in this study should be used as estimates of the normal range and not taken as absolute, due to the limitations and caveats of this study, as mentioned above. We would therefore suggest that if the DBS result is abnormal, it should be followed up by a serum sample to confirm the result, as we have yet to perform a diagnostic evaluation of the derived DBS TSH reference intervals.
Footnotes
Acknowledgements
The authors would like to thank Nawal Mohamed and the other members of the South West Thames Newborn Screening specimen reception team for all their help with sample handling and organisation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
MM.
Contributorship
All authors contributed to the design and execution of the study. All authors participated in manuscript drafting and critical revision.
