Abstract
Background
Thyroid disorders are common in women of childbearing age and are associated with adverse pregnancy outcomes. Physiological changes in pregnancy and the lack of pregnancy-specific reference ranges make managing thyroid disorders in pregnancy challenging. Our aim was to establish trimester-specific thyroid function reference intervals throughout pregnancy, and to examine the prevalence of thyroid autoimmunity in otherwise euthyroid women.
Method
This was a prospective, cross-sectional study of thyroid function tests (TFTs) in pregnant women attending a large, tertiary referral maternity hospital. Patients with known thyroid disorders, autoimmune disease, recurrent miscarriage, hyperemesis gravidarum and pre-eclampsia were excluded. TFTs were analysed in the CUH biochemistry laboratory using Roche Modular E170 electrochemiluminescent immunoassay. Trimester-specific reference ranges (2.5th, 50th and 97.5th centiles) were calculated.
Results
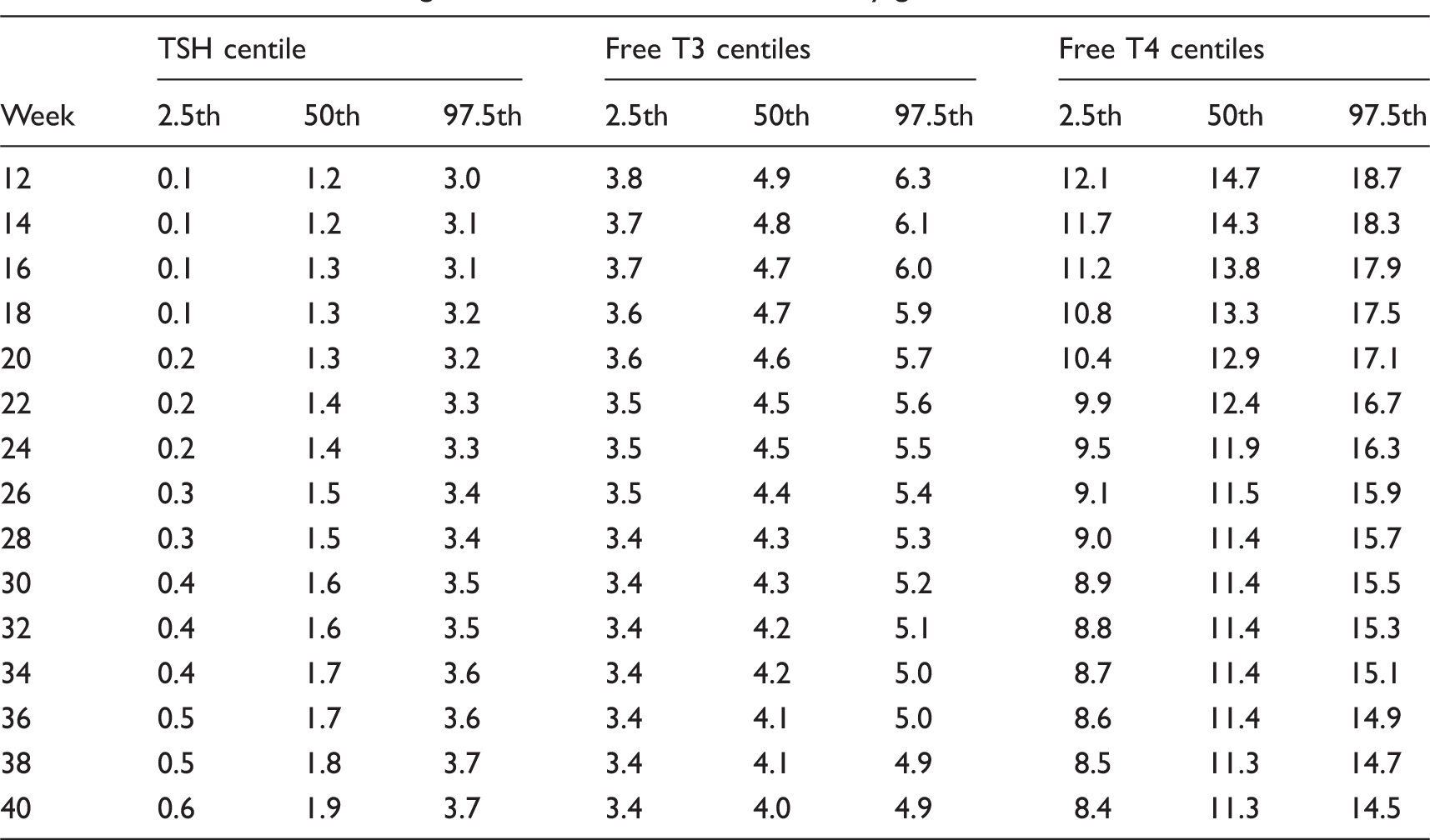
Three-hundred-and-fifty-one women were included into the analysis. Median maternal age was 30. Thyroid-stimulating hormone concentrations showed slightly increasing median centile throughout gestation. Free thyroxine (T4) and T3 decreased throughout gestation. Table 1 demonstrates the calculated percentiles according to gestational weeks.
Conclusion
We established pregnancy-specific thyroid function reference intervals for our pregnant population, for use in clinical practice.
Introduction
Thyroid disorders are common in women of childbearing age and complicate 1–2% of pregnancies. 1 Overt hyperthyroidism is reported to affect 0.1–0.4% of pregnancies, while subclinical hypothyroidism affects 2–3% of pregnancies.2,3 Untreated thyroid disorders in pregnancy are associated with adverse obstetric and fetal outcomes including fetal growth restriction, pre-eclampsia, pre-term delivery, placental abruption and fetal death.4–10 Interestingly, a few studies also showed lower average IQ levels of children born from thyroid deficient mothers.5,11,12 In women with autoimmune thyroid disorders such as Graves and Hashimoto’s disease, the passage of maternal thyroid antibodies to the fetus also results in neonatal thyroid disorders. 13 However, due to the physiological changes in pregnancy, neither the use of non-pregnant thyroid function reference intervals nor a single pregnancy reference interval is appropriate. This makes the diagnosis and management of thyroid disorders in pregnancy challenging.
A significant change in the regulation of thyroid hormone in pregnancy is attributed to the 50% increase in plasma volume, the increase in thyroid-binding globulin (TBG) caused by the elevated oestrogen concentrations in pregnancy and the relative deficiency of iodine in pregnancy. As a result, free T4 concentrations in pregnant women are about 10–15% lower than non-pregnant women.14,15 Furthermore, free T4 concentrations naturally decline across trimesters. 16 In addition to the altered regulation, the demands on the thyroid also increase in pregnancy. This is due to the adjustment of the thyroid in response to the increase in TBG, the alterations in thyroid metabolism at placental concentration in later gestation and also the demands from the fetus in the early gestational weeks.14,15 The thyroid gland in the fetus begins to develop during week 7–9 of gestation, and structurally matures around week 17. Although fetal thyroid-stimulating hormone (TSH) is detectable at week 10, it remains relatively low until week 18. Until then, the fetus depends solely on maternal thyroid hormone, which is vital for fetal brain development. 15
In recent years, understanding of thyroid physiology and its effects on pregnancy has increased. The development of sensitive biochemical assays has also improved understanding of the natural trends in pregnancy where FT4 and FT3 decrease, and TSH increases through gestation.
Several studies have previously demonstrated the changes of thyroid function in pregnancy and calculated both longitudinal and cross-sectional reference intervals. In 2001, Price et al. had constructed reference intervals and compared Caucasian women to Asian women. They concluded that there was no significant difference, however, this only included 31 pregnant Asian women and 76 pregnant Caucasian women, and was limited to the first and second trimesters of pregnancy. 17 In 2010, Vila et al. in Spain constructed reference intervals in the first and third trimesters. This, however, was a combination of cross-sectional samples as well as longitudinal samples from the same patients. 18 In 2010, Yu et al. 19 calculated longitudinal reference intervals in the Chinese population and demonstrated narrower intervals compared to other studies. Cotzias et al. in 2007 also reported cross-sectional reference intervals.1 However, the assay used in that study is not used in our laboratory.1,19–22
The validity of these earlier studies on pregnancy-specific thyroid reference ranges has been questioned due to several issues: use of small sample groups; restriction to one single trimester; use of older or different free hormone assays; differing methods of reference range calculation and studying populations with different endemic iodine status. 1 According to the guidelines by the British Thyroid Association, the reference population should not only be free of any family of personal history of thyroid disorders, but should also be negative for thyroid autoimmunity. They also recommend reference ranges to be confirmed locally, and calculated using 95% confidence limits from either log transformed data or using non-parametric methods of statistical analysis. 23 As thyroid disorders in pregnancy are not uncommon and known to affect pregnancy outcome, the determination of pregnancy-specific thyroid function reference intervals in each population for the use in clinical practice is needed.1,4,19–21 This is to ensure accurate diagnosis and management of overt hypo- and hyperthyroidism, as well as subclinical hypothyroidism in pregnancy. In current practice, we are using trimester-specific reference ranges provided by Roche, 24 which were established from healthy pregnant women in Essen and Hamburg, Germany, published in 2009. Additional reference ranges for populations in Norway, Austria and Sweden are also available. 24 We designed this study due to the lack of available pregnancy-specific thyroid function reference ranges in the Irish population.
We aimed to establish trimester-specific maternal FT4, FT3 and TSH concentrations in the low-risk pregnant population attending our hospital. Concentrations were, determined with the Roche Modular E170 electrochemiluminescent immunoassay, which is in routine use in our biochemistry laboratory. We also aimed to investigate the prevalence of thyroid autoimmunity in otherwise euthyroid women in our population.
Methods
A cross-sectional analysis of thyroid function tests (TFTs) in pregnant women attending a large, tertiary referral maternity hospital (∼9000 deliveries per year) was conducted between February 2010 and February 2011. As previously shown, 1 a sample size of 300 or more women has ample power to estimate reliable reference ranges for thyroid hormones. Thus, a sample size of 350 women was sought.
Healthy women attending the antenatal care clinic with a singleton pregnancy were randomly selected for participation in the study. Women with known thyroid disease, autoimmune diseases, diabetes mellitus, recurrent miscarriage, hyperemesis gravidarum and pre-eclampsia were excluded from the study. In women with TSH concentrations <0.1 or >10 mU/L or with FT4 > 22 pmol/L, exclusion was only made on the basis of diagnosis of thyroid disorder. As this was a cross-sectional study, each woman in this study had only one single TFT and no follow-up TFT within the study was done. Retrospective exclusion should be avoided when generating time-specific reference intervals.25,26 Therefore, exclusion was only done at the time of participation into the study.
Women were recruited between 11 and 36 weeks gestation. Each woman was sampled on only one occasion. Recruited women were provided a copy of the study protocol and written informed consent was received from all participating women. This study was approved by the Clinical Research Ethics Committee of the Cork Teaching Hospitals.
Two researchers were responsible for recruitment and collection of blood samples. A single serum sample by venepuncture was collected into a non-heparinized tube from each participant. TFTs were analysed using the Roche Modular E170 electrochemiluminescent immunoassay in the Biochemistry Laboratory at Cork University Hospital. The equipment was a Roche E170 analyser routinely used in practice.
Statistics
Descriptive statistics were derived to describe maternal characteristics in the study population. Since maternal age was skewed, the median and inter-quartile ranges are presented. To assess the association between thyroid peroxidase antibodies (TPO) with TSH and free T4, a Pearson’s correlation was performed.
The distributions of the data for TSH, free T3 and free T4 were examined using quantile–quantile plots (Q–Q plots) and box-plots. If the data exhibited skewness, the Box–Cox method was performed to identify the optimal power transformation to achieve normality of the data. For heavy-tailed data, no corresponding transformation was available.
Quantile regression was conducted to derive gestation-specific 95% reference intervals (2.5th, 50th and 97.5th centiles) for each thyroid hormone. 27 Quantile regression models the relationship between the covariate of interest (i.e. gestational week) and the conditional quantiles of the outcome of interest (i.e. thyroid hormone), and this approach is favourable in health studies where lower and upper quantiles are of clinical interest. 27 Dependent upon model fit, either parametric linear or quadratic predictors were used. Analyses were calculated using the statistical software package R. 27
Results
Overall, 354 women were recruited into the study; however, a total of three women were excluded as they did not meet the inclusion criteria. In the first trimester, one woman was excluded due to T4 concentrations above 22.00 pmol/L. In the second trimester, another one was excluded due to an outlying TSH of 7.02 mU/L. One was excluded in the third trimester due to TSH concentrations of 13.00 mU/L. These exclusions were made on the basis of the likelihood of undiagnosed overt thyroid disorder in these women. Women with TSH < 0.1 mU/L were included as no diagnosis of hyperthyroidism was subsequently made. Thus, the final sample size for the study was 351 women.
In the final study population, median maternal age was 30 years (inter-quartile range 27–33, range 17–45); all women had an uncomplicated medical history. The majority of women (78.63%;
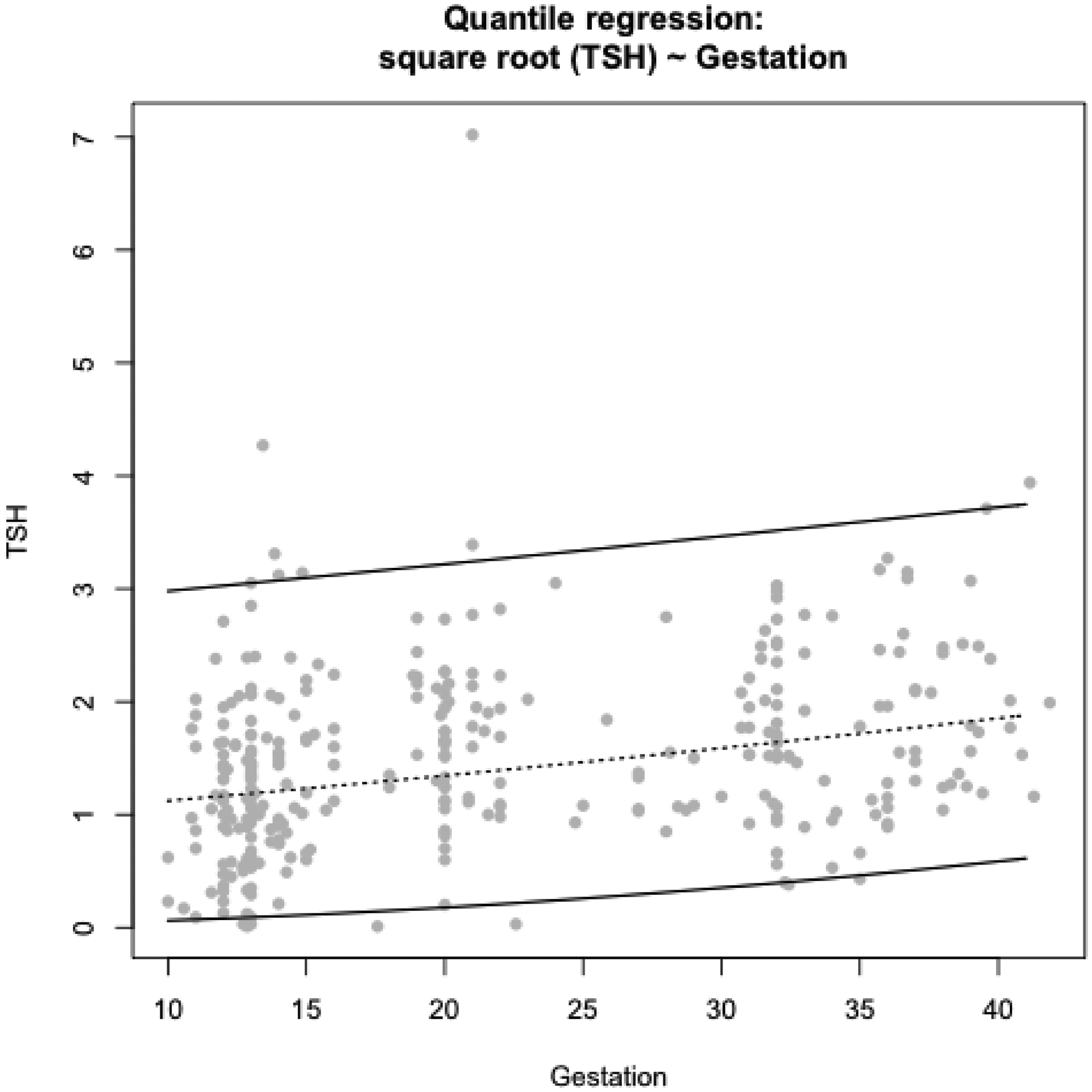
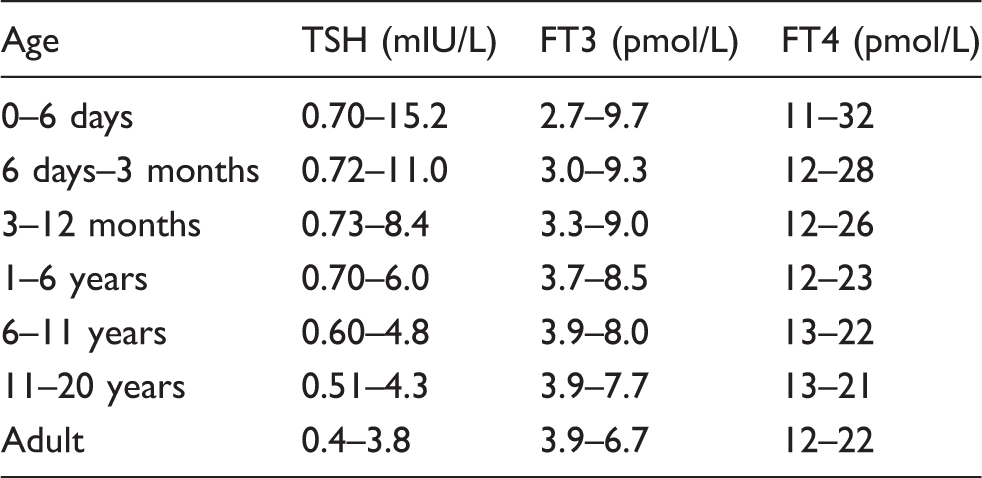
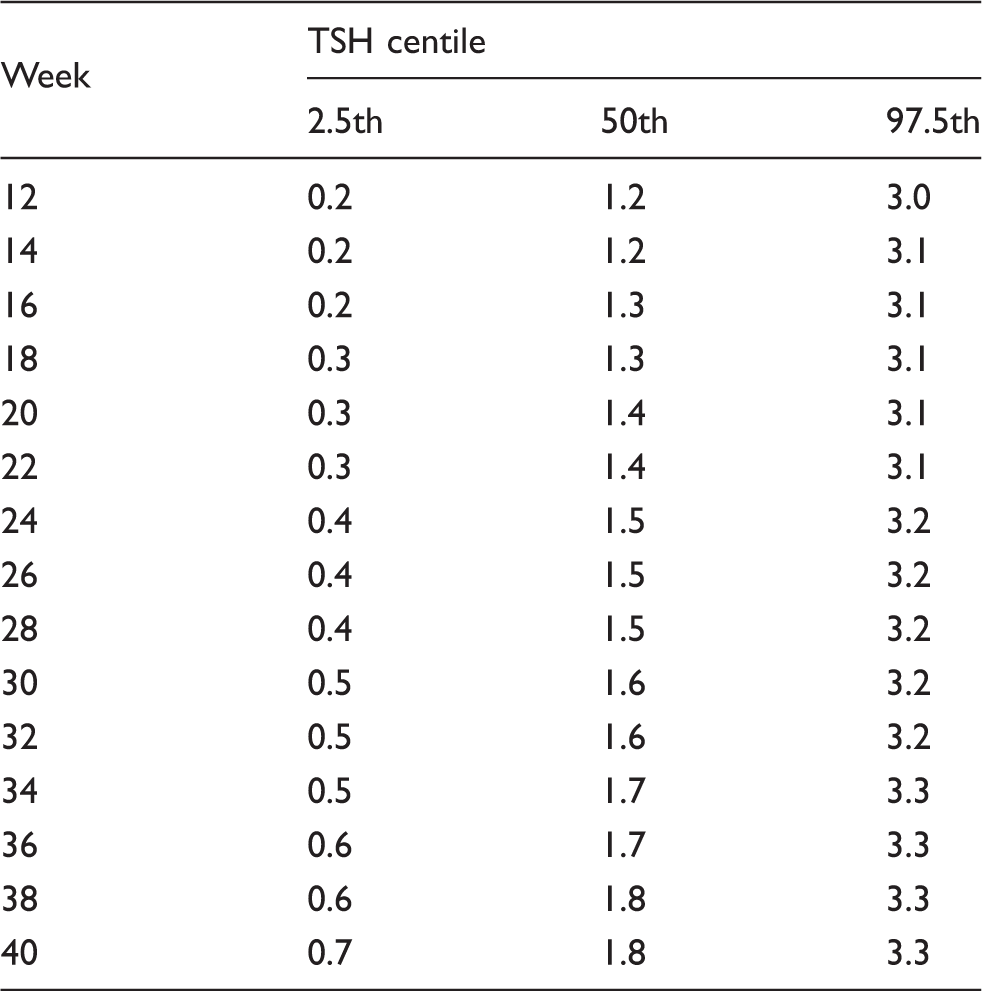
TSH concentrations were positively skewed. Using the Box–Cox method, the maximum likelihood estimate of lambda was 0.49, supporting a square-root transformation of TSH. TSH centiles, after conversion back to its original scale, were wide throughout the three trimesters (Figure 1). The median centile increased slightly throughout pregnancy.
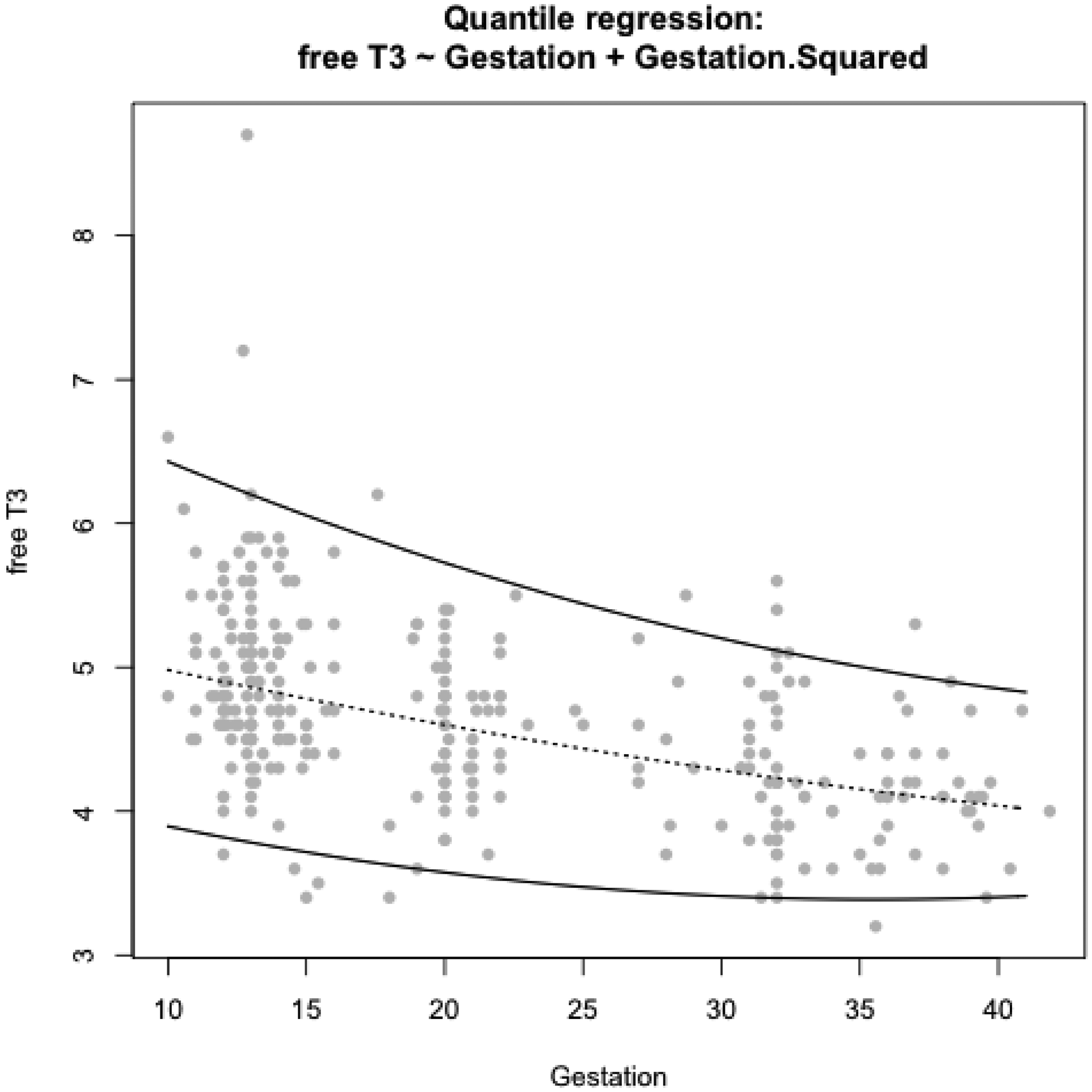
Although heavily tailed, free T3 concentrations were not significantly skewed, and thus no transformation was undertaken. The quadratic quantile model conferred an improved fit over the linear model and was consequently used to estimate centiles. Free T3 concentrations decreased throughout pregnancy (Figure 2).
Similar to T3 concentrations, free T4 concentrations were heavily tailed, though not significantly skewed. However, in this study population, a prominent decline in free T4 concentrations was observed until approximately 25 weeks gestation; after this time point, free T4 concentrations began to concentrate (Figure 3). Therefore to capture this trend and improve the fit of the model, a ‘Broken Stick’ quantile model was performed with a change point at 25 weeks gestation. Notably, reference intervals were slightly wider after this change point.
Pregnancy-specific centiles for free T4, calculated as 2.5th centile = 14.485 − 0.2136 Gest + 0.1634 [Gest − 25]if Gest > 25; 50th centile = 17.285 − 0.2321 Gest + 0.2202 [Gest − 25]if Gest > 25 and 97.5th centile = 20.941 −-0.1977 Gest + 0.0966 [Gest − 25]if Gest > 25.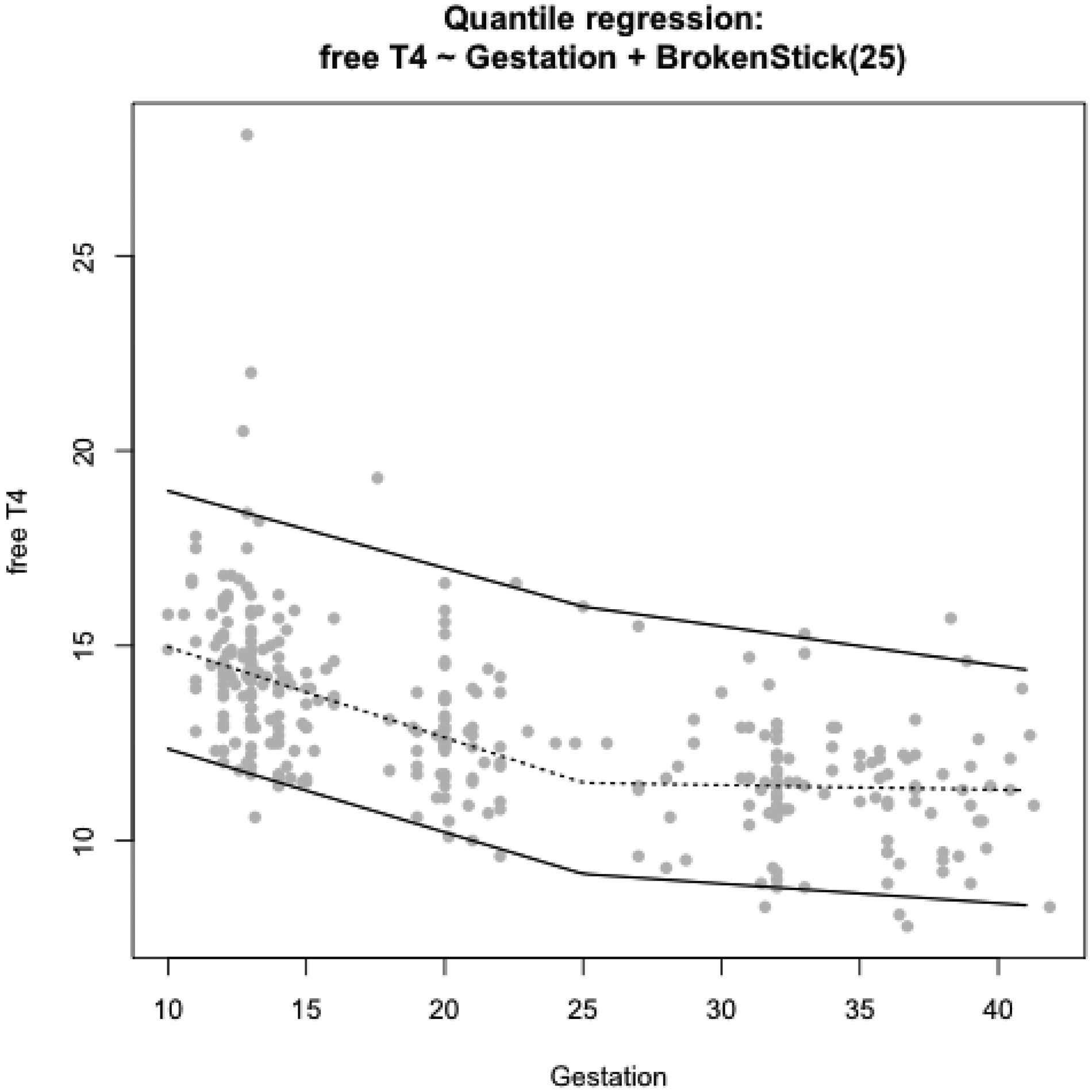
Derived reference ranges for TSH, free T3 and free T4 by gestational week.
Non-pregnant reference intervals for free T4, TSH and free T3 used in Cork University Hospital (provided by Roche 22 ).
Prevalence of TPO within this cohort were also examined. We found 7.2% (
Derived reference ranges for TSH exclusive of TPO positive women by gestational week.
Discussion
In this study, we report trimester-specific maternal FT4, FT3 and TSH concentrations in the current low-risk pregnant Irish population, determined with the Roche Modular E170 electrochemiluminescent immunoassay which is in routine use in our biochemistry laboratory. We also report the prevalence of positive thyroid autoimmunity in otherwise euthyroid women in our population.
This study demonstrates the changes in thyroid function in pregnancy as has been described in previous studies. Free T4 and T3 were found to decrease, and TSH found to increase throughout pregnancy. We also found a significant relationship between TPO and TSH, where 12% of patients with positive TPO also had TSH concentrations above the 97.5th centile.
This supports existing evidence of thyroid changes in pregnancy and the need for pregnancy-specific reference ranges. Differences in demographics, iodine intake and assays used affect the reference intervals produced. Thus, reference intervals for each population should be established to ensure accurate diagnosis and management of thyroid disorders in pregnancy.
There are several preceding studies examining thyroid function in pregnancy. In comparison to non-pregnant concentrations, free T4 and TSH were generally found to be on the hypothyroid spectrum, and T3 to be on the hyperthyroid spectrum. In contrast to previous studies, we elected the use of the ‘Broken Stick’ model for T4 concentrations as this was the best fit to our data.
Samples in this study were taken at set points of hospital contact with patients in each trimester. Being a tertiary hospital with a very busy service, it was deemed not feasible to have samples from each week of gestation. Thus, this does not reflect the changes in each week of gestation, but rather, reflects the changes across trimesters.
We compared our intervals to the pregnancy-specific reference intervals provided by Roche, 24 which are a combination of healthy pregnant women from Essen and Hamburg. The inclusion and exclusion criteria were similar to our study; healthy, pregnant women with a normal pregnancy without known complication. In the study by Roche, 24 women with a known or supposed thyroid dysfunction or history, substitution of thyroid hormones, fT3 concentrations < 3 pmol/L and fT4 concentrations > 22 pmol/L and TSH concentrations outside a range of 0.1–10 mIU/L were excluded. Whereas in our study, women with TSH concentrations of <0.1 mIU/L were only excluded if a diagnosis of hyperthyroid was made. It is unclear whether women with hyperemesis gravidarum, known autoimmune disorders, diabetes mellitus, recurrent miscarriage or pre-eclampsia were excluded in Roche’s 24 study, whilst these women were excluded from our study. We found narrower TSH intervals with lower upper limits in each trimester. TSH concentrations in our study were comparatively lower in the first trimester, despite exclusion of patients with hyperemesis gravidarum. Free T4 intervals were similar whereas free T3 were higher in our study. However, these were still lower than that of the non-pregnant population. In their study, TPO concentrations were slightly higher in the third trimester. 24
Another study in Canada using the Roche immunoassay showed similar trends with our study. This study examined FT4 in each trimester of pregnancy. Women were included only when judged to be euthyroid based on non-elevated anti-TPO and TSH concentrations that were within the reference interval between 0.1 mIU/L and thresholds of 2.5, 3.0 and 3.5 mIU/L in the first, second and third trimesters, respectively. Although the intervals for the first and second trimesters were comparable to our study, the reference intervals in our study in the third trimester were lower. This could be due to the strict inclusion criteria of the Canadian study. 20 However, it is questionable whether exclusion based on such thresholds is appropriate.
A study in the UK found that TSH concentrations were closer to non-pregnant concentrations in the first and second trimesters. In contrast with our findings, they found higher T3 concentrations compared to the non-pregnant levels. 1 Earlier work by a Chinese study found narrower intervals for free T4 and TSH. This was thought to be due to ethnic difference, but may also be due to the longitudinal design of the study, whereas most other studies were cross-sectional. 19
Differences in the TFT reference intervals could also be attributed to differences in iodine status in the study populations. A study by Nawoor et al. 29 showed that 55% of Irish pregnant women in the first trimester had urinary iodine excretion (UID) suggestive of iodine deficiency during the summer, and 23% in the winter. While in the control non-pregnant female Irish group, 53% and 11% had UID suggestive of iodine deficiency in the summer and in the winter, respectively. 29
Further research surrounding the diagnosis and management of thyroid disorders in pregnancy is needed. Thyroid disorders are known to be associated with adverse pregnancy outcome. Treatment should be aimed to achieve euthyroidism in each trimester. Future studies should focus on the positive effects and risk reduction with adequate treatment of thyroid disorders in pregnancy.
This study is one of many stepping-stones in the management of thyroid disorders in pregnancy. Although we have demonstrated similar trends to other studies in the changes of thyroid function in pregnancy, differences in assays used, ethnicity and possibly geographical location may influence the reference intervals produced. Therefore, there should be reference intervals unique to each population to ensure accurate diagnosis and management of thyroid disorders in pregnancy. We also demonstrated differences in the upper limits of TSH when TPO positive women were excluded, supporting the relationship with subclinical hypothyroidism. We have produced trimester-specific thyroid function reference intervals for the current Irish pregnant adult population.
Footnotes
Declaration of conflicting interests
None.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
This study was approved by the Clinical Research Ethics Committee of the Cork Teaching Hospitals (ECM 4 (z) 02/03/10).
Guarantor
KOD.
Contributorship
ASK was involved in protocol development, gaining ethical approval, patient recruitment, data analysis and wrote the first draft of the manuscript. ZM was involved in patient recruitment. JEL and KH were involved in statistical analysis. CJ, MS and JM approved and provided laboratory services. KOD supervised and is the appointed guarantor. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
