Abstract
Background
Macrocomplexes can be the cause of elevated serum hormone concentrations and may cause diagnostic confusion. This is well recognized for prolactin and commonly screened for using polyethylene glycol (PEG) precipitation. The phenomenon and a suitable screening method is less familiar with respect to thyroid-stimulating hormone (TSH).
Method
Samples sent to the laboratory for routine analysis of thyroid function and found to have a TSH >10 mU/L were evaluated to determine the prevalence of macro-TSH in the Roche Elecsys assay, using PEG precipitation with confirmation by gel filtration chromatography.
Results
Of 495 samples tested, 3 (0.6%) were found to have macro-TSH. From the distribution of recoveries, a cut-off <25% was determined for identifying samples requiring further investigation for the presence of macro-TSH.
Conclusion
The prevalence of elevated TSH due to macro-TSH was found to be 0.6%. Laboratories should be aware of this cause of assay interference.
Introduction
Requests for the measurement of thyroid-stimulating hormone (TSH) are frequently made to laboratories, in order to screen for elevated concentrations consistent with primary hypothyroidism. In primary hypothyroidism, a raised TSH would be expected to be associated with a lowered concentration of free thyroxine (FT4), both parameters returning to within the reference range once the patient is established on appropriate levothyroxine replacement therapy. In borderline or subclinical hypothyroidism, or if replacement therapy is suboptimal, TSH may be elevated with a FT4 concentration within the reference range.
Discordance between TSH and FT4 results may indicate less common causes of thyroid dysfunction, for example a normal or high TSH with a raised FT4 concentration is found in TSH secreting tumours, but an analytical cause for unusual or unexpected results should always be considered.
Analyte-antibody complexes are a well-known cause of clinical confusion in the interpretation of endocrine results and have been particularly well delineated with respect to prolactin (macroprolactin). 1 We report investigations in a patient sample found to have elevated serum TSH due to an immunoglobulin-TSH complex and a study of the prevalence of the phenomenon in routine requests for serum TSH made to the laboratory.
Samples and methods
Serum was obtained from patients’ samples on which thyroid function testing had been requested as part of routine clinical management. To determine the prevalence of immunoglobulin-TSH complexes, 495 samples from 463 patients found to have a TSH concentration >10 mU/L were investigated by polyethylene glycol (PEG) precipitation. Samples were analysed on the day of collection or stored frozen prior to analysis.
PEG precipitation was used as a simple method of precipitating immunoglobulins G, A and M. A solution of 25% PEG 6000 (VWR, Lutterworth, UK) was prepared in water. A serum aliquot of 0.2 mL of was added to 0.2 mL of the 25% PEG 6000 solution (TSH·PEG) and to 0.2 mL of deionized water (TSH·H2O) as a control. The samples were vortex mixed, incubated for 10 min at room temperature and then centrifuged for 5 min at 14,000 rotations per minute. The TSH concentration of the supernatant was assayed using the Roche Modular E170 (Roche Diagnostics, West Sussex, UK). The percentage recovery of TSH following PEG precipitation was calculated as [(TSH·PEG)/(TSH·H2O)] × 100.
Samples with a recovery less than 25% were further investigated by gel filtration chromatography. This was performed using a 60 × 1.6 cm HiLoad 16/60 Superdex 75 prep grade column (GE Healthcare, Chalfont St Giles, UK) with an AKTA prime protein purification system (GE Healthcare Life Sciences, Chalfont St Giles, UK). Serum applied directly to the column was eluted using Tris buffer. Double strength Tris buffer was made by dissolving Trizma-Pre-set crystals (6.084 g) (Sigma Aldrich, Dorset, UK) in deionized water (1800 mL) using a magnetic stirrer. Sodium chloride (30.389 g) (BDH, Poole, UK) and sodium azide (0.78 g) (BDH) were added while stirring continuously. The buffer was adjusted to pH 7.40 using 1 mol/L sodium hydroxide (VWR, Lutterworth, UK) and 1 mol/L hydrochloric acid (VWR). The solution was diluted to 2 L using deionized water. The sodium concentration was checked on the Roche Modular System using a 1:2 dilution with deionized water (target value: Sodium = 137 mmol/L, range 132–142 mmol/L). At a flow rate of 1 mL/min, the first 30 min is a void volume followed by collection of 40 fractions (1.4 mL each) over 90 min, the final 30 min is a further void volume to wash the column. The concentration of TSH was measured in the fractions collected using the Roche Modular E170 (Roche Diagnostics, West Sussex, UK) and the contribution of macro-TSH to the total TSH concentration was calculated.
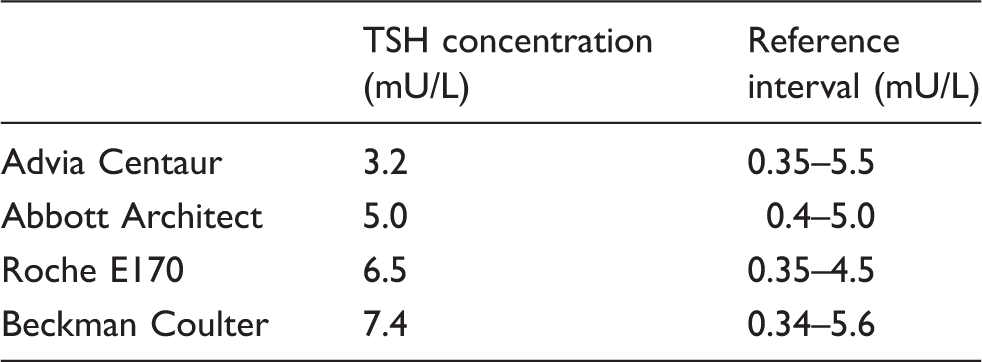
A sample found to have macro-TSH was investigated further by measurement of TSH on additional analytical platforms using the Abbott Architect (Abbott Diagnostics, Maidenhead, UK), Advia Centaur (Siemens, Erlangen, Germany) and Beckman Coulter (Beckman Coulter, High Wycombe, UK).
Results
Percentage recovery of TSH following PEG precipitation
All 495 samples with a TSH concentration >10 mU/L were treated with 25% PEG solution. The frequency distribution for percentage recovery of TSH in these samples is shown (Figure 1). The data were normally distributed using the Kolmogorov–Smirnov statistical test (P = 0.059) with a mean recovery of 47% (SD 11.17%) and 95% reference limits (mean ± 2SD) of 25.6–69.4%. A recovery of <25% was taken as indicative of requiring further investigation for the possible presence of immunoglobulin-TSH complexes.
Histogram of percentage recovery of TSH after polyethylene glycol precipitation.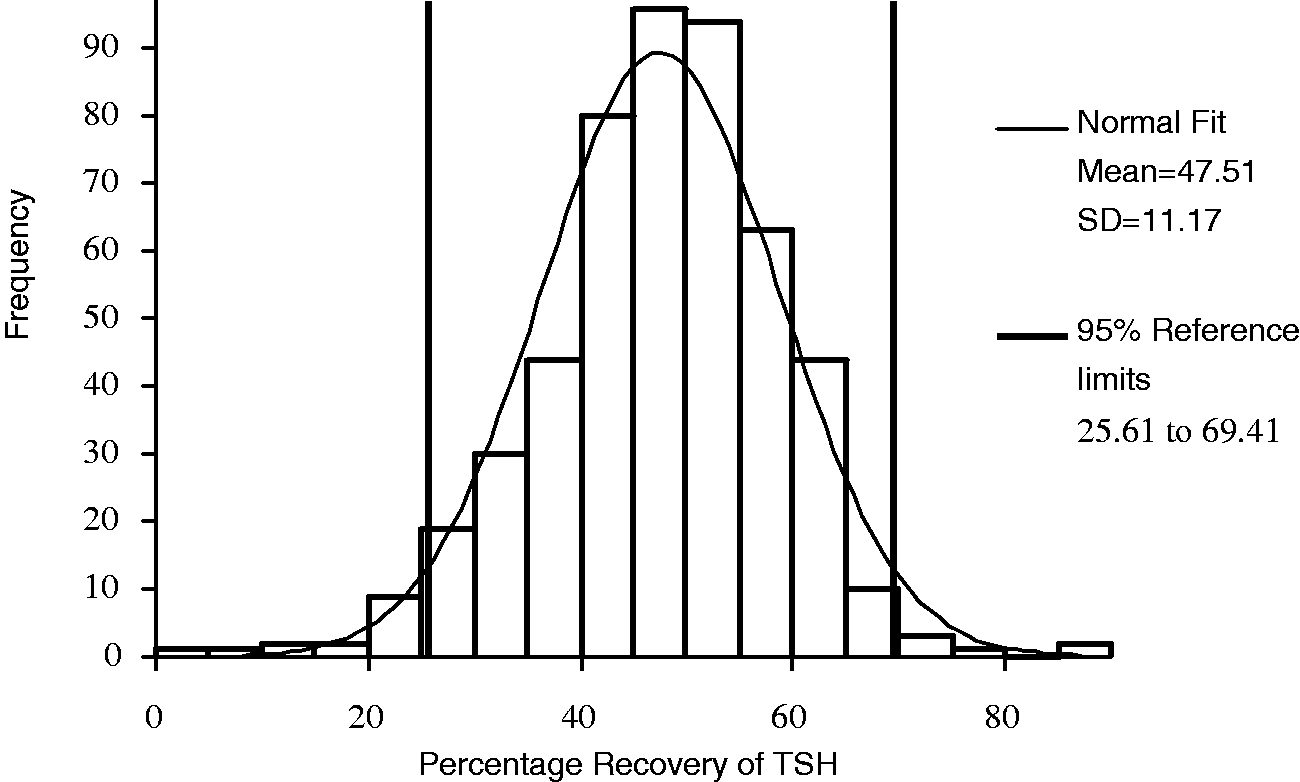
Gel filtration chromatography
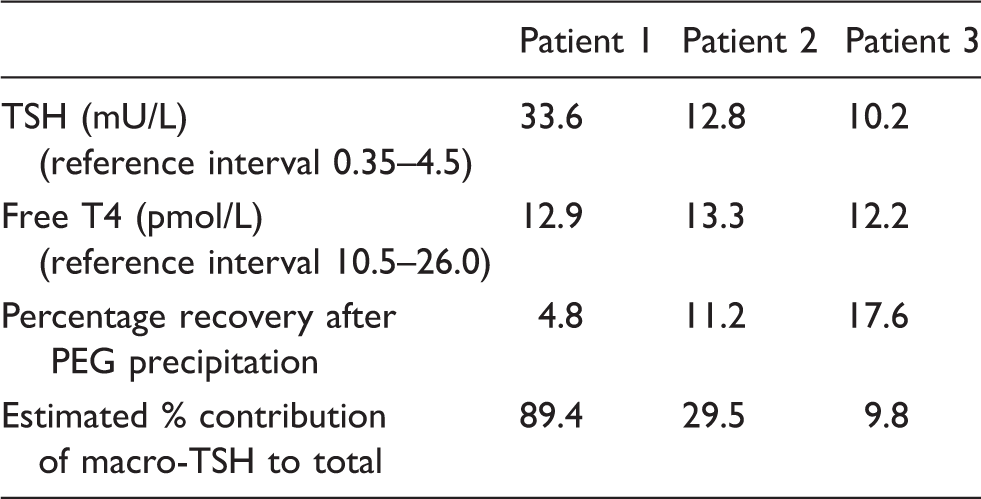
In three of the 15 patients with recovery <25%, gel filtration chromatography confirmed the presence of immunoglobulin-TSH complexes (Figure 2). The biochemical findings in these patients are summarized in Table 1.
Gel filtration chromatogram (separated on Superdex S75 column) of (a) monomeric TSH (albumin is shown as a molecular weight marker) and (b) serum from patient 1 showing the presence of macro-TSH. Biochemical findings in the three patients’ samples in whom macro-TSH was identified.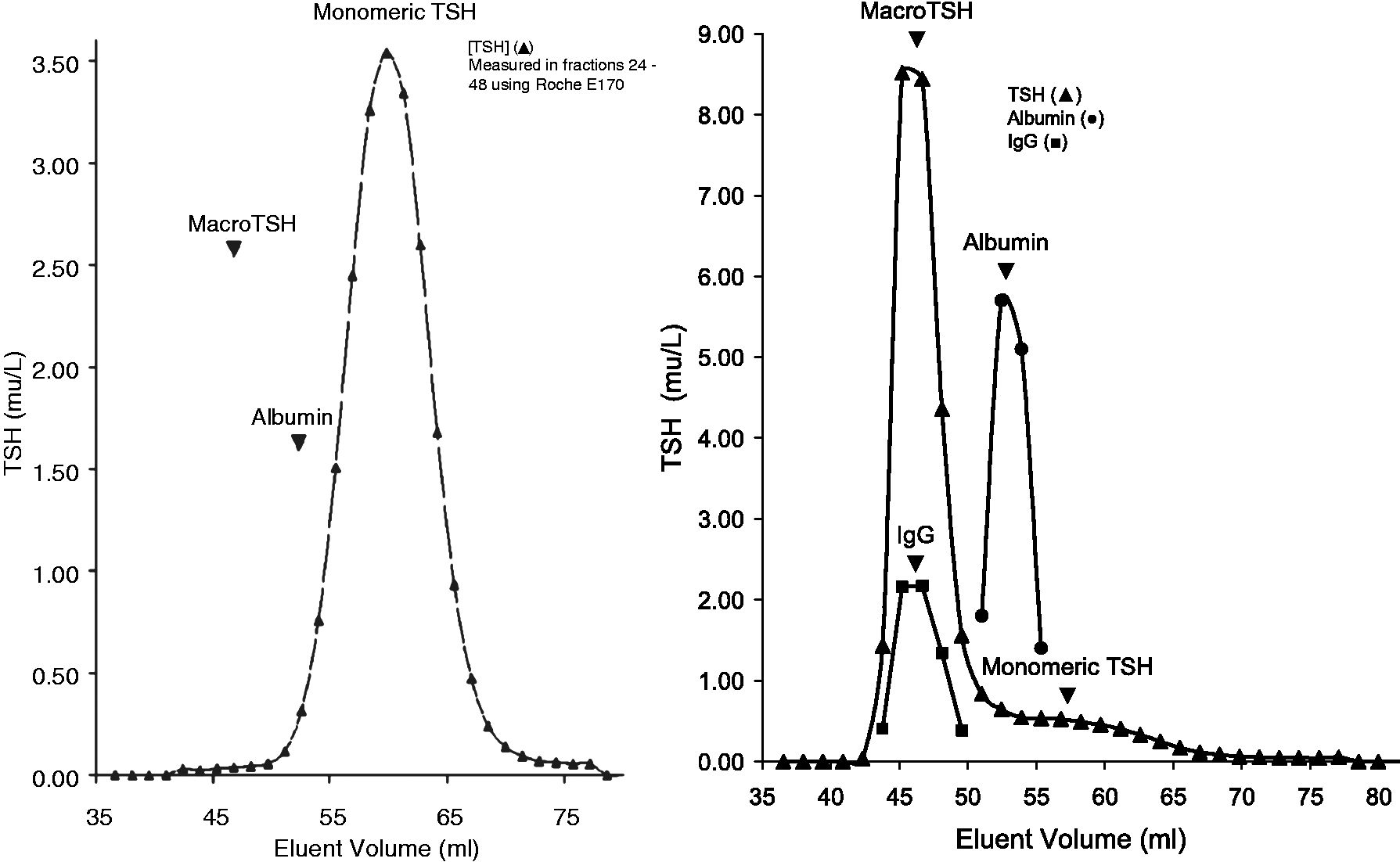
Measurement of TSH concentration in a sample containing immunoglobulin-bound TSH using different analytical platforms
Comparison of results of TSH measurement by different methods using a serum sample from Patient 1.
Discussion
Interference in immunoassays is relatively common. One large study of 5130 sets of results found analytically incorrect results in 28 (0.53%) which, in 23 patients, was considered of significant magnitude to have had a potentially adverse effect on cost and/or clinical care. 2 Our laboratory performs over 50,000 thyroid function tests a year so, if this estimate is correct we should be alert to the possibility of at least five analytically incorrect results with potentially significant consequences every week. There are a number of causes of interference in thyroid hormone assays. Cross-reactivity has been virtually eliminated from modern assays but other causes of interference include drugs, 3 antibodies 4 and macrocomplexes.
Macrocomplexes as a cause of interference have been best described with respect to prolactin; it is recommended that all laboratories screen for the presence of macroprolactin and PEG precipitation has been proposed as a way to achieve this. 1 Macrocomplexes have also been well documented as a cause of spurious elevation of various other hormones, 5 enzyme activities 6 and, in one study, at least 8% of samples with elevated vitamin B12 concentrations. 7
Macro-TSH was described in a mother and two neonates who had a raised TSH detected as part of screening programmes for congenital hypothyroidism. 8 A further case in a neonate was reported with the intention of raising awareness of the phenomenon and illustrating the use of gel filtration chromatography to differentiate it from interference caused by heterophile antibodies. 9
Macrocomplexes are a form of autoimmune response and as many factors influence the immune response the patient makes to the antigen, the concentration of an analyte may change. This is highlighted in the difference in TSH concentration reported for two samples from patient 1.
PEG tends to precipitate a proportion of free analyte in addition to immunoglobulin and immunoglobulin complexes and the proportion precipitated varies between analytes making it necessary to determine an analyte and method-specific reference range when using the technique. 1 This study has examined a wide range of samples and determined an analyte and method-specific reference range for percentage recovery of TSH after PEG precipitation, a cut-off of <25% could be used to direct samples for further investigations into the possible presence of macro-TSH. In a population of 495 patients having routine measurement of thyroid function and found to have a TSH >10 mU/L, three (0.6%, rising to 20% for those samples with a recovery <25%) were found to have IgG complexed TSH.
PEG precipitation is a simple, although relatively crude, technique which may be used as a screening test for the presence of immunoglobulin-complexed (macro) TSH. However, the technique is subject to certain limitations. It has been shown that there may be under-recovery of IgA macrohormones leading to misdiagnosis when PEG precipitation is used. 10 In addition, the presence of an increased serum globulin concentration may increase the amount of monomeric hormone precipitated with PEG leading to an erroneous presence of a macrohormone. 11 A further potential limitation is that PEG can interfere with some immunoassays. 12
After PEG precipitation, confirmation of the presence of a macrohormone using gel filtration is recommended. However, gel filtration may, on occasions, fail to differentiate between interference from a cross-linking mechanism (e.g. heterophile antibodies) and a macrohormone. 13 Additional work to prove definitively the presence of macro-TSH is required and could involve further gel filtration chromatography studies using acid elution buffer, 8 preincubation with urea 14 or selective precipitation with antihuman IgA Agarose or protein A or G Sepharose. 15
It is important that clinical biochemists are aware of possible causes of immunoglobulin interference in TSH assays, including the presence of macro-TSH, to prevent inappropriate interpretation of thyroid function test results from influencing clinical care.
Footnotes
Acknowledgements
The authors acknowledge the Staff at University Hospital Southampton, East Kent Hospitals University NHS Foundation Trust and Oxford University Hospitals NHS Trust for measurement of TSH on alternative platforms.
Declaration of conflicting interests
None declared.
Funding
This study was funded by Clinical Biochemistry, Derriford Hospital.
Ethical approval
This service development project was discussed with the hospital research and development department and ethical approval was not required.
Guarantor
RMA.
Contributorship
RMA conceived the study. FM, JJ, PM and AC performed the laboratory work. All authors were involved in writing of the manuscript.
