Abstract
Background
Ethylene glycol poisoning remains a rare but important presentation to acute toxicology units. Guidelines recommended that ethylene glycol should be available as an ‘urgent’ test within 4 h, but these are difficult to deliver in practice. This study assessed a semi-quantitative enzymatic spectrophotometric assay for ethylene glycol compatible with automated platforms.
Methods
The ethylene glycol method was assessed in 21 samples from patients with an increased anion gap and metabolic acidosis not due to ethylene glycol ingestion, and seven samples known to contain ethylene glycol. All samples were analysed in random order in a blinded manner to their origin on a laboratory spectrophotometer.
Results
In this study, seven samples were known to contain ethylene glycol at concentrations >100 mg/L. The method correctly identified all seven samples as containing ethylene glycol. No false-positives were observed. Thirteen samples gave clear negative results. Ethylene glycol was present at <20 mg/L in one sample, but this sample remained within the limits of the negative control. Passing-Bablock correlation of estimates of ethylene glycol concentration against results obtained when the samples had been analysed using the quantitative method on an automated analyser showed a good correlation (R = 0.84) but with an apparent under-recovery.
Conclusions
A semi-quantitative assay for ethylene glycol was able to discriminate well between samples containing ethylene glycol and those with other causes of acidosis. It is a practical small-scale assay for rapid identification of cases of ethylene glycol poisoning.
Ethylene glycol (EG) poisoning remains a rare but important presentation to acute toxicology units with most cases of accidental poisoning occurring in children. 1 Diagnosis is usually based on clinical symptoms allied with commonly used but unreliable indices such as osmolar gap whose sensitivity is 90% and specificity 50% at best. 2 Delays in diagnosis are serious as given the 4-h half-life, a 2-h delay results in conversion of 30% of EG to glycolic acid. Ideally, a biochemical diagnosis should be made immediately using gas chromatography-mass spectrometry (GC-MS)-based methods 1 but delivering a definitive result within the guideline-recommended 6-h time limit for initiation of treatment is challenging in the vast majority of centres. 1 Ingestion of EG is rare in the UK with only 608 enquiries to the National Poisons Information service in 2010–2011 relating to 488 individual exposure events to toxic alcohols and glycols, 1 so many laboratories find it difficult to justify maintaining this assay in their repertoires. Logistic and laboratory limitations leave clinicians with a dilemma of whether or not to treat on a presumptive basis or unable to exclude EG poisoning in their differential diagnosis.
Recently, a quantitative enzymatic spectrophotometric assay for EG compatible with common automated platforms has become available based on modification of the kinetic assay parameters of a long-established veterinary method (Nuuchem Diagnostics Ltd, Armagh BT61 9HR). 3 This method is based on the enzymatic oxidation of EG using glycerol dehydrogenase/NAD and has now been adapted for use as a manual test using a basic spectrophotometer, providing a pragmatic solution for this rare but important assay. The kits come with materials to perform three separate measurements on patient samples, have an 18-month shelf-life and include a positive control and a sample at 200 mg/L as a standard against which the unknown sample may be compared with. Results are available in 15 min. The performance characteristics of this assay have not been assessed.
Methods
To assess the suitability of this method for rapid confirmation or exclusion of EG being present in a sample, a total of 21 samples from patients with an increased anion gap metabolic acidosis not due to EG ingestion were obtained, including 7 samples known to contain EG were presented for analysis. The known EG-containing samples were distributed randomly throughout the batch. All samples were analysed in a blinded manner to their origin.
Assays were performed using Thermo Eurovision201 spectrophotometer (Paisley, UK). Following reconstitution of the sample diluent and allowing 5 min for it to dissolve completely, 750 µL of diluent was dispensed into three cuvettes. To each cuvette, 10 µL of either sample, positive control or negative control (containing 200 mg/L) is added and the cuvettes mixed by inversion. After 5 min of incubation (tests were performed at room temperature – 18℃, but the manufacturer indicates that the incubations may be performed at 37℃ if preferred), the initial OD at 340 nm for each cuvette is recorded. Hundred and fifty microliters of activation reagent are then added to each cuvette and the cuvettes are incubated for a further 5 min, after which time the OD is again recorded. The increase in OD of the sample is compared with the negative control, and if greater than that of the negative control and the result for the positive control is also greater than that of the negative control, then there is a strong likelihood that the patient has ingested EG. Using the data obtained for the negative control, estimates of the concentration of EG can be made and compared with values obtained using the quantitative enzymatic automated analyser assay.
Results
In this study, seven samples were known to contain EG at concentrations >100 mg/L. The method correctly identified all seven samples as containing EG. EG was present at <200 mg/L in one sample, but with the spectrophotometer operated in kinetic mode, a linear reaction rate could be observed after approximately 2 min which continued until the end of the incubation. This was not seen in other samples that were classified as negative. No false-positives were observed. There was a clear difference in the reaction plot between samples not containing EG and the sample with low EG concentration. It was also evident that with increasing EG concentration in a sample, the observed OD at 340 nm also increased.
A Passing-Bablock correlation of estimates of EG concentration against results obtained when the samples had been analysed using the quantitative method on a Roche Cobas 8000 analyser showed a good correlation (0.84) but with an apparent under recovery (Figure 1).
Passing-Bablock regression plot of results derived from a semi-quantitative spectrophotometer assay compared with the same method run on an automated Cobas Mira analyser.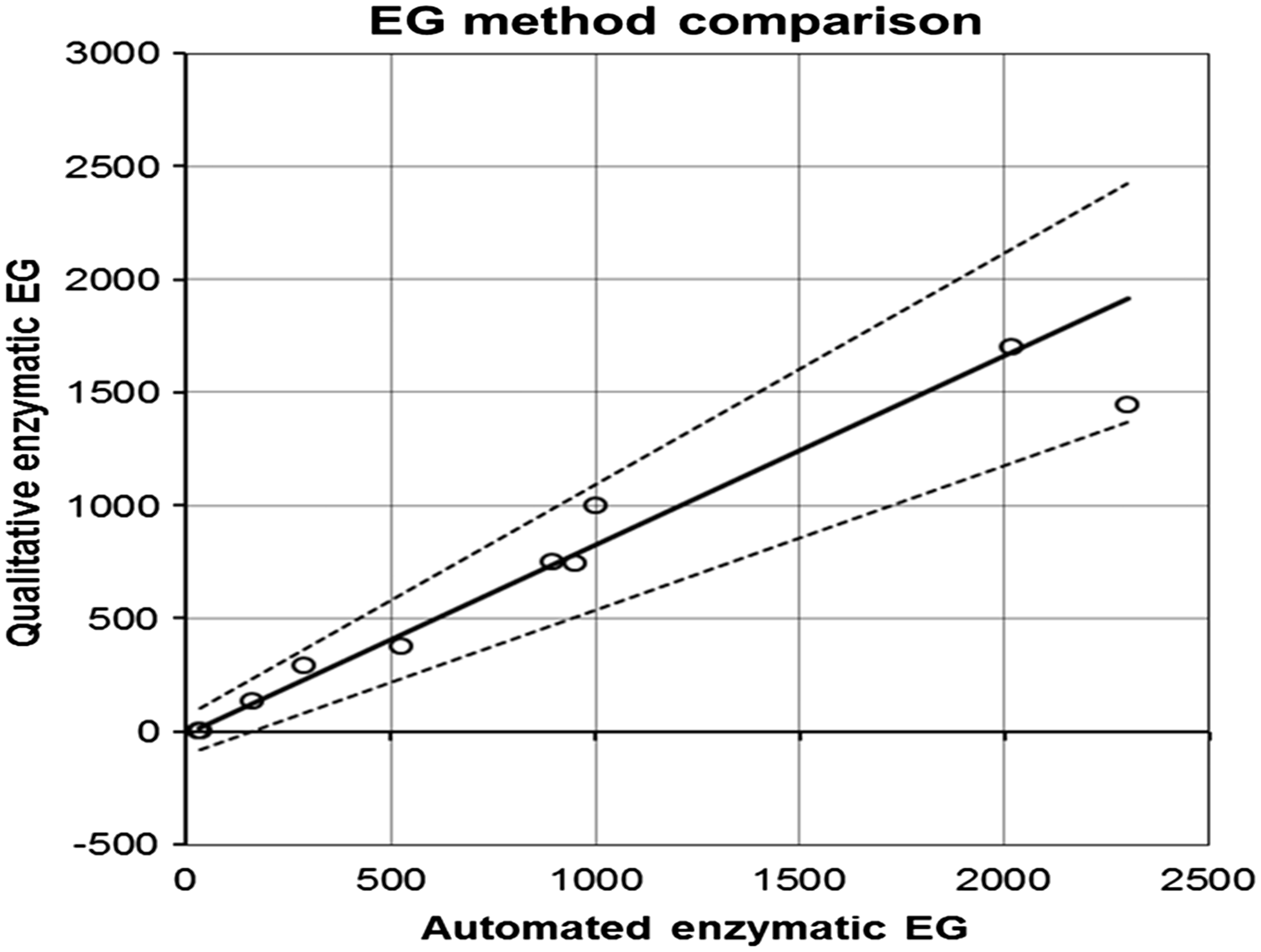
Discussion
The qualitative method performed well in this study, allowing for rapid and correct identification of all EG-containing samples in a mixture of samples from patients with other causes of acidosis. The method is a generalized variation of the quantitative assay designed for auto-analysers. Although the method as used appeared to under recover EG, it does seem to have the potential for semi-quantitative use, with a relationship between OD increase or reaction rate and the EG concentration. It would appear to be an effective method for rapid evaluation of samples in suspected EG ingestion.
In the original evaluation, 3 the enzymatic method was found to be very specific, with only di-, tri-, tetra- and propylene glycol and 2,3-butanediol causing falsely elevated measurements at concentrations in excess of 1000 mg/L and 22 other potential interfering compounds having no effect.
Although enzymatic methods will not match the performance of GC-based methods, they do allow a rapid assessment for the presence of EG. Series of cases of EG poisoning are rare. US data from one centre for 2010–2014 based on 106 patient encounters and 222 analyses including 10 cases of known EG poisoning suggested that 80% could be managed using the enzymatic assay alone. 4 The other two cases required GC-MS confirmation due to the presence of propylene glycol. In total, 25 samples contained propylene glycol and 8 cases required GC-MS confirmation to exclude the presence of EG. UK experience based on 811 requests showed 33.5% contained EG with diethylene glycol present in 14% of positive samples and never alone. 5 However, the laboratory clinician will still need to be aware of the limitations of the assay in interpreting the results and advising colleagues in the Emergency Department.
It is likely that limitations in the availability of diagnostics lead to under-reporting of cases of EG poisoning in the UK. The availability of a semi-quantitative screening kit may help close this gap and can easily and cost-effectively be incorporated into routine UK laboratories.
Conclusions
A semi-quantitative assay for EG was able to discriminate well between samples containing EG and those with other causes of acidosis. It is a practical small-scale assay to allow routine laboratories to identify cases of EG poisoning quickly and cost-effectively and easily be added to diagnostic algorithms for this disorder.
Footnotes
Declaration of conflicting interests
Dr James is Medical Director of NuuChem, supplier of the kits and helped locate samples for this analysis.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
ASW.
Contributorship
AS: performance of assays; sample location; data analysis.
AT: Original assay assessment. Supply of patient samples from WEQAS.
DJ: supply of kits and QC materials. Manuscript review.
ASW: Writing and review of manuscript.
