Abstract
A 52-year-old lady presented with a history of occasional, severe abdominal cramps, postprandial diarrhoea and weight loss. After routine gastrointestinal investigations, she was diagnosed with irritable bowel syndrome. Over six months, she developed occasional facial flushing prompting assessment of neuroendocrine tumour markers. Urinary 5HIAA, 5HT, chromogranin A and neurokinin A were significantly elevated. Scans showed extensive hepatic metastases but did not show the location of a primary tumour. Somatostatin analogue therapy was commenced but despite increasing doses, symptoms increased and biomarkers rose dramatically. Interferon alpha was introduced concomitant with somatostatin analogue therapy. Biomarkers were monitored regularly. Within six months, symptoms abated and biomarkers reduced, continuing to fall over the next year, close to reference range. To manage side-effects of interferon alpha, dose was reduced from time to time. During these short periods, neurokinin A showed significant transient increases (75–150 ng/L) and carcinoid symptoms returned. For more than seven years, and with the co-operation of the patient, a balance was achieved between interferon alpha side-effects and disease control. Scans showed tumour load to be stable. The patient survived for 10 years post diagnosis. She chose to discontinue interferon alpha and received peptide receptor radiation therapy in her final year. Throughout, neurokinin A remained the most sensitive monitor of her disease progression.
Introduction
Small bowel neuroendocrine tumours (SB-NETs) are uncommon, slow growing, low-grade malignant neoplasms. They may present early as an incidental finding or at a later stage with intermittent or acute bowel obstruction due to a desmoplastic reaction in the mesentery adjacent to the tumour. In about one quarter of patients, presentation is delayed and carcinoid syndrome develops typically characterized by diarrhoea, flushing and abdominal pain. As these are frequent symptoms in much more common conditions, the diagnosis of SB-NETs may be delayed for years. When diagnosis is established at a later stage, treatment options are fewer. Somatostatin analogue therapy (SST) is used in patients with SB-NETs to control symptoms before and during other interventions. However, in patients who have advanced disease, SST may fail to control symptoms.
Three circulating biomarkers are used for laboratory diagnosis of SB-NETs and are useful for monitoring disease activity thereafter. These include circulating chromogranin A (CGA), which is the only general marker for metastatic neuroendocrine tumours (NETs) (but is raised in many other conditions and situations), 24 h urinary 5HIAA, the breakdown product of serotonin, 5-hydroxy tryptamine (5HT) secreted by the majority of SB-NETs, some lung NETs and a small number of pancreatic NETs and neurokinin A (NKA) which is specific for SB-NETs. 1 NKA is a useful prognostic marker for SB-NETs and for monitoring response to treatment. 2 The NKA assay used in this study was ‘RIA in house’. The same antibody has been used over a 20-year period. All specimens measuring greater than 50 ng/L were serially diluted. Inter-assay coefficient of variation remained <10%. Details of this assay compared with an assay developed in the USA are published. 3
Interferon has been known to be an effective antiviral and antitumour agent since the 1960s, although many solid tumours do not respond to interferon alpha (IFA). Öberg et al. 4 have championed the use of IFA in NETs since the early 1980s, when IFA was introduced in the management of SB-NETs with promising results. IFA is generally used concomitant with SST for the treatment of NETs. However, many centres avoid IFA treatment, and there remains much debate surrounding its usefulness. IFA affects tumour cells by inhibiting cell proliferation via cell cycle block at the G1-S phase and so reducing tumour growth. In addition, IFA exhibits an immunomodulatory effect that stimulates killer cells and macrophages. Furthermore, IFA has antiangiogenic effects. The combination of these effects results in stabilization of disease in many patients.
Case report
A 52-year-old lady presented at a gastrointestinal (GI) clinic in June 2001 with a history of occasional abdominal cramps which could be severe and could last for several hours. These symptoms had been present intermittently, for more than three years. Her symptoms were not associated with either diarrhoea or constipation. She reported a recent weight loss of 2 kg. She had a medical history of migraine. There was nothing of note on examination, routine biochemistry was normal and a coeliac screen negative. At review eight weeks later, she had developed postprandial diarrhoea with a further weight loss of 2.5 kg. Upper and lower GI endoscopies were performed with normal findings. A diagnosis of irritable bowel syndrome was made.
Six months later, she returned to the clinic with further weight loss (8 kg, since the onset of symptoms). She now reported cyclical symptoms of diarrhoea which persisted for three to five days. She also complained of occasional facial flushing prompting assessment of neuroendocrine tumour markers. Urinary 5HIAA and 5HT were elevated at 637.1 µmol/24 h (reference range (RR) = 10–47 µmol/24 h) and 12.05 µmol/24 h (RR = 0.3–1.3µmol/24 h), respectively, CGA was 1050 U/L (RR = 0–50 U/L) and NKA 350 ng/L (RR =0–20 ng/L). A computerized tomography (CT) scan and somatostatin scintigraphy showed extensive hepatic metastases with para-aortic and iliac lymphadenopathy. However, no abnormal findings were identified in the ileum or colon suggestive of a primary lesion. The patient declined to have a liver biopsy. However, the histology grade of these tumours is usually grade 1 and only occasionally grade 2.
Disease was considered to be surgically incurable and the patient declined any surgical intervention. Treatment with SST (10 mg IM every four weeks) was commenced and the dose later increased twice (to 20 mg and then to 30 mg every four weeks) in an attempt to relieve symptoms which remained uncontrolled. After four months of SST, circulating tumour markers had continued to rise: CGA 3400 U/L and NKA 1500 ng/L (the latter indicating a prognosis of less than six months). 2 Following multidisciplinary discussion, IFA was added to SST in an initial dose of 1.5 M units three times weekly which was increased incrementally to 9 M units three times weekly.
Circulating biomarkers continued to rise for four weeks of IFA treatment, CGA to 8000 U/L and NKA to 4500 ng/L. However, within two months, her symptoms eased, and the combination of SST and IFA was continued. By six months symptoms had abated. Routine monthly haematological monitoring (required due to the possibility of leukopenia and thrombocytopenia during IFA therapy) remained satisfactory. Over the following year, tumour markers gradually fell. Urinary 5HIAA settled around the upper limit of normal and 5HT returned within the RR after 18 months of interferon treatment. Circulating CGA remained between 1000 and 2000 U/L and NKA was secured between 40 and 75 ng/L within 18 months (Figure 1).
Circulating NKA pretreatment, response to SST and then SST concomitant with IFA across 30 months from diagnosis.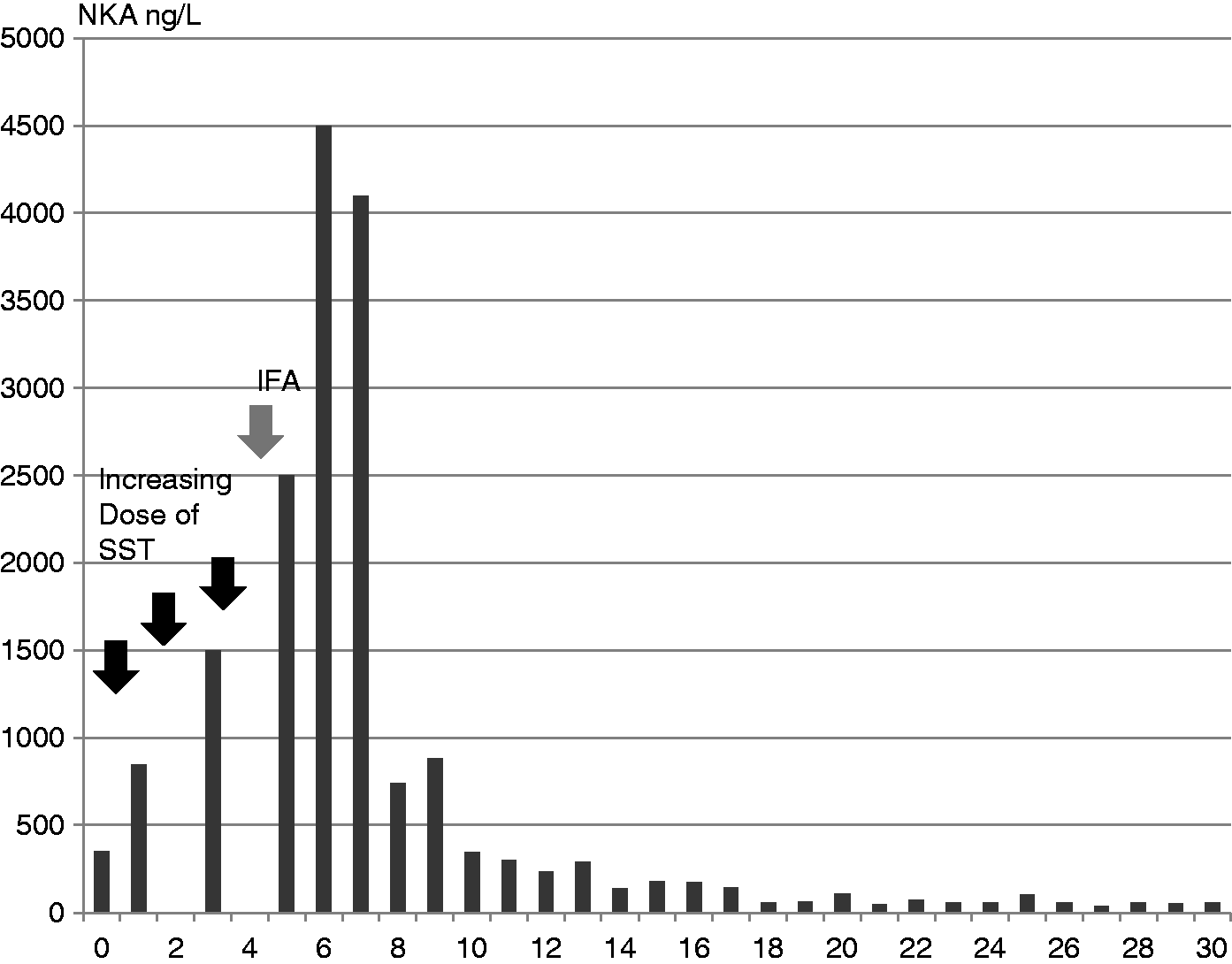
IFA is associated with unpleasant side-effects including malaise, nausea and headache, and some patients find these problems sufficiently intolerable to discontinue treatment. After starting IFA, this patient experienced marked fatigue and suffered frequent migraine, initially two to three times weekly. The dose of IFA was reduced from time to time from 9 M units to 3 or 1.5 M units three times weekly, and occasionally the drug was withdrawn for a week. Side-effects associated with IFA diminished. The frequency of migraines reduced to about once a month. For more than seven years, and with the co-operation of the patient, a balance was achieved between side-effects associated with IFA and disease control. During the short periods of IFA reduction or withdrawal, NKA showed significant transient increases (75–150 ng/L) when carcinoid symptoms would return (Figure 2).
Variation in circulating NKA across 60 months of treatment with IFA. The scale is expanded from Figure 1 to highlight the immediate response of NKA to IFA change in dose. Arrows indicate times when IFA was temporarily reduced or withdrawn.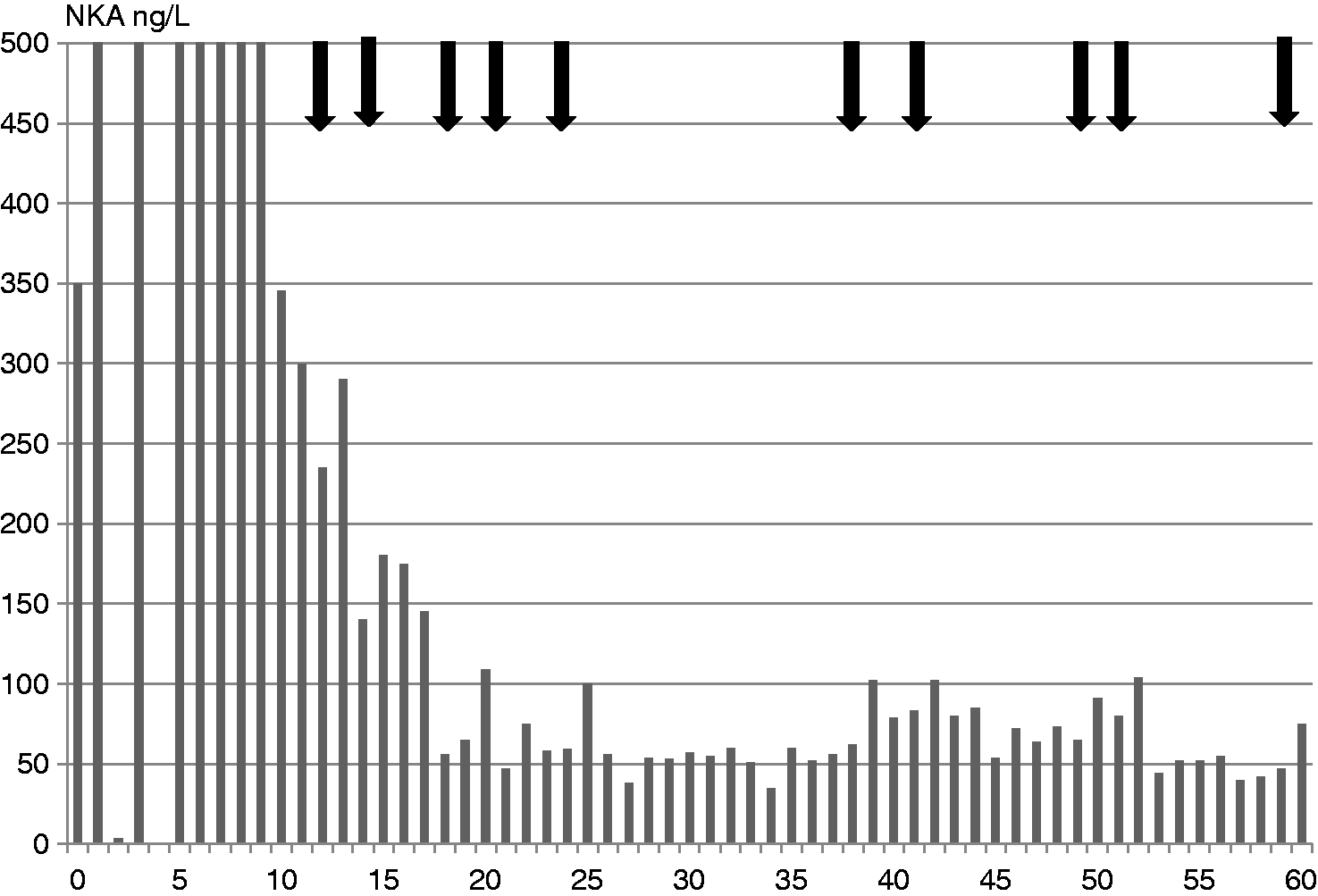
Serial CT scanning during this period showed stable disease. Multiple hepatic metastases in these studies, appeared unchanged. A mass anterior to the right common iliac vessels in 2005 measured 3.4 cm and in 2006 measured 2.8 cm.
As time progressed, the patient became less tolerant of IFA and continued on a lower dose, 1.5 M units three times weekly, finally preferring to discontinue IFA treatment altogether in November 2009. Following this, urinary 5HIAA increased to 790.2 µmol/24 h, CGA to 4620 U/L and NKA to 322 ng/L, with return of symptoms. SST was increased to 50 mg every four weeks with supplementary injections of short-acting analogue as required. Biomarkers continued to increase with urinary 5HIAA rising to 1985.4 µmol/24 h, CGA to 10,480 U/L and NKA to NKA 917 ng/L. A weight loss of 7.4 kg was recorded over three months. Other treatment options were considered at multidisciplinary discussion, including hepatic artery chemoembolization, peptide receptor radiation therapy (PRRT) and selective internal radiation therapy. Following careful assessment, this patient received PRRT, three cycles of Yttrium 90 DODATATE (at 12- and 16-week intervals) over a six-month period. After the first treatment, she gained 2 kg in weight and enjoyed improved energy levels. Symptoms continued to improve for approximately six months following her third treatment and during this time radiological examinations showed stable disease. However, circulating biomarkers reduced only temporarily, her symptoms returned and her health declined. She died nine months after her last treatment with Yttrium 90 and a total of 120 months after her original presentation.
Discussion
Although more than 50% of patients with SB-NETs may be stabilized using SST, the management of SB-NETs that present with high-volume liver disease and carcinoid syndrome remains challenging. Over the last two decades, several new modalities of treatment have been shown to have clinical utility including extensive hepatic resection, hepatic artery embolization or chemoembolization and the use of PRRT and selective internal radiation therapy. Most recently, it has also been reported that in some types of NET, the tyrosine kinase inhibitor sunitinib 5 and the mechanistic target of rapamycin inhibitor everolimus 6 have been shown to improve outcomes. There are, however, no trials evaluating the comparative effectiveness or the optimal scheduling of these treatment options.
Since the earliest reports of IFA use in the treatment of NETs in 1982, further studies showing favourable outcomes for patients treated with IFA long term have been published. 7 Tumour reduction was reported by Arnold 8 in 50% of patients treated with a combination of IFA and SST. In a review, Öberg et al. discussed published data from more than 500 patients treated with IFA worldwide. Median dose of IFA used was 5 million units subcutaneously three to five times per week. The subjective response rate was 60%, biochemical response 44% and tumour response 11% in these patients. 9 Several reports have shown a benefit of IFA therapy in combination with somatostatin analogues in patients resistant to SSTs alone, where 77% showed biochemical response with 18% showing complete biochemical remission. 10 However, in a randomized trial enrolling only therapy-naive patients, there was no reported difference in outcome between patients treated with SST alone IFA alone or a combination of SST and IFA, although this report has been criticized on the basis of small subgroup numbers and mixed tumour locations. 11
Of those who do receive and tolerate IFA long term, approximately half show a good symptomatic response, with disease stabilization and reduction in circulation tumour markers. Chemical response has been clearly illustrated with IFA; however, tumour response is recorded in only about 20% of patients. Andersson et al. 12 have reported that tumour cells content in metastases is reduced, but lesion size may remain unchanged. They suggest that microscopic assessment of tumours may be a better measure of change with IFA treatment as they recorded a decrease in tumour tissue content of 2.5%/month with 95% confidence without measurable change in tumours radiologically.
We have found that 66% of patients tolerate IFA long term with all showing a biochemical response within one month of starting treatment. 13 The regular dose we use is 3–5 MU three times weekly. This case report illustrates that the dose may be reduced for short periods in order to improve quality of life, yet with maintenance of overall disease control. For patients who tolerate IFA, other treatment options, not previously available, can now be considered.
Biochemical response has been clearly illustrated with IFA. However, tumour response is recorded in only about 20% of patients. We show that circulating NKA is a sensitive monitoring tool for disease change in patients treated with IFA. In the light of the publication by Andersson et al., 12 who showed tumour cell reduction without change in metastases size, the measurement of NKA offers an excellent means of response assessment to IFA that is non-invasive.
Conclusion
In a 52-year-old lady who presented with an aggressive, advanced, metastatic SB-NET with poor prognosis and limited treatment options, 10-year survival was achieved using SST concomitant with IFA. Her clinical response to treatment was accompanied by a dramatic and sustained fall in circulating NKA and radiological evidence of disease stability. A good quality of life was maintained by careful adjustment of IFA dosage to achieve a balance of effective treatment and tolerability. Measurement of circulating NKA permitted careful monitoring of disease during reduction of IFA. IFA has a useful place in the therapeutic armamentarium for advanced midgut NETs and its effectiveness as a treatment can be monitored accurately using circulating NKA.
Footnotes
Acknowledgement
The authors wish to thank Mrs Claire Erwin for her expert technical assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Joy Ardill is in receipt of an unrestricted grant from Inter Science Institute, USA. This work was funded by Belfast Health and Social Care Trust and an unrestricted Grant from Ipsen UK.
Ethical approval
Ethical approval was obtained from the Office for Research Ethics Committees Northern Ireland (ORECNI) (12/NI/0098). Written, informed consent was obtained from the patient for publication of her clinical data.
Guarantor
JESA.
Contributorship
JA Collected the data from the patient and from her clinical notes and wrote the manuscript.
BJ, DMcC and ME managed the patient treatment and edited the manuscript.
