Abstract
Background
Urokinase plasminogen activator (uPA) and its inhibitor (PAI-1) have been shown to be of merit as biomarkers for a variety of cancers. Prostate tissue resections from patients with prostate cancer (PCa) and benign prostatic hyperplasia were analysed to determine the influence of freeze-drying on the recovery of uPA and PAI-1 and their predictive performance.
Methods
Prostate tissue was frozen in liquid nitrogen and homogenised into a fine powder in a precooled stainless steel punch homogeniser. One aliquot of the powder was extracted directly, and a second aliquot was freeze-dried overnight and then extracted. The extracts were analysed by FEMTELLE assay to determine the concentrations of uPA and PAI-1. uPA/PAI-1 ratios were calculated for each sample, and the mean ratios for the frozen and the lyophilised tissue were compared.
Results
The concentrations of uPA measured for the frozen and lyophilised samples are strongly correlated (R = 0.90 ± 0.05). The same applies to the PAI-1 measured (R = 0.89 ± 0.03). The uPA/PAI-1 ratios for the lyophilised and frozen samples were strongly correlated. The uPA/PAI-1 ratios for frozen and lyophilised samples were found to be essentially the same with values of 0.0344 ± 0.0066 and 0.0340 ± 0.0068, respectively (P = 0.9633).
Conclusion
The recovery of uPA and PAI-1 from a deep frozen prostate tissue homogenate followed by freeze-drying proceeds with a loss of 10 and 11%, respectively, with no influence on the uPA/PAI-1 ratio. Lyophilisation is a safe procedure for the preservation of frozen prostate tissue samples as it permits recovery of the markers without compromising their use for diagnostic purposes.
Introduction
Tissue samples often have to undergo long distance transport to reach specialised laboratories for the purpose of analysis. The transport of biological material is strictly regulated to avoid leakage, dissipation or infection to third parties. Specific regulations exist for each mode of transport, with air transport being the most rigorous. In view of the stringent regulations especially for fresh and frozen biological material, methods by which samples can be preserved for transport without compromising the performance of the assay in question are of great interest.
One avenue of facilitating the transport of frozen biological material is lyophilisation (freeze-drying). Freeze-drying removes water from frozen samples by sublimation and desorption. It can be viewed as a three-step process consisting of freezing, primary drying and secondary drying. 1 This technique thus combines the benefits of freezing and drying to provide a shelf-stable and readily soluble product.2,3 Lyophilisation is under renewed discussion even for the preservation of vaccines to remote areas. 4 Biological material processed in this manner may be readily transported across administrative boundaries with little or no restrictions. A recurring major concern being that the integrity of biological samples remains intact. 1 Due to the long duration of the process, freeze-drying can lead to some irreversible adhesions in the tissue matrix and small losses of the target molecules, be it steroids, oligosaccharides or proteins. Therefore, a detailed elucidation of the impact of lyophilisation on sample stability and marker recovery is warranted.
In a recent study on prostate cancer diagnosis, we have demonstrated that the relative abundance of urokinase plasminogen activator (uPA) and its inhibitor type 1 (PAI-1) has the potential of distinguishing between prostate cancer and benign prostatic hyperplasia (BPH). 5 The current study was motivated by the problem that prostate tissue resections and biopsies flash frozen in liquid nitrogen and homogenised in a freeze punch process must be stored at –80℃ to ensure stability of the uPA and PAI-1 marker components. In the present work, we sought to determine whether freeze-drying affects the uPA/PAI-1 ratio which was identified as a candidate marker. 5 Accordingly, prostate tissue was collected by transurethral resection (TURP), precooled in liquid nitrogen, crushed into fine powder and then freeze-dried for assessment of uPA and PAI-1 concentrations. The data show that the process of freeze-drying may not only facilitate transport of biological samples at normal temperature, but that the diagnostic merits of the two marker components are only marginally affected. The implications of these findings for the stability of freeze-dried prostate tissue, and the specific problem of uPA and PAI-1 preservation, are further discussed.
Materials and methods
Patients and samples
Patient data and diagnosis.
Sample preparation and measurement of uPA and PAI-1 content
The first step in sample preparation was performed as described previously.
5
The punch type tissue homogeniser consists of a cylinder which accommodates a piston. The polished instrument (Suppl Figure 1) was precision machined from stainless steel and had the following dimensions: cylinder height: 40 mm, outside diameter: 50 mm, inside diameter: 24 mm, inside depth: 30 mm; piston length: 90 mm and piston diameter: 23 mm. Pincher type handles were flexibly attached to both piston and cylinder to permit immersion into liquid nitrogen. For homogenisation, 100–300 mg of fresh prostate tissue was cut into small pieces to give a volume equivalent of 2–3 peas, placed on aluminium foil, blotted dry with paper towel and wrapped to be cooled in liquid nitrogen. The solid tissue pieces were then entered into the cylinder which was precooled in liquid nitrogen. The precooled piston was then inserted and 10–12 firm hammer blows were applied to the piston with a few lifts and turns. The resulting tissue powder was removed with precooled spatulas and transferred into screw cap vials for storage in liquid nitrogen. Approximately one half of each powdered sample was set aside for freeze-drying. Marker extraction by the piston type punch homogeniser was also compared with a FastPrep-24 tissue and cell homogeniser (MP Biomedicals, Europe). For the latter, a small measure of 2-mm glass beads and the deep frozen prostate tissue were placed in an impact-resistant 2-mL screw-capped microtube. The tube was shaken for two periods of 30 s at a homogenisation speed of 6 m/s, before being stored in liquid nitrogen for subsequent measurement and comparison of uPA and PAI-1 content. For freeze-drying, the samples placed in cryovials were arranged on a rack. The caps of the cryovials were replaced with parafilm and pierced with a syringe needle to permit the escape of vapour. Samples were then freeze-dried overnight at a vacuum of less than 0.1 mbar and a temperature of less than –40℃, using a 6 -L Labconco freeze drier (Kansas, MO, USA).
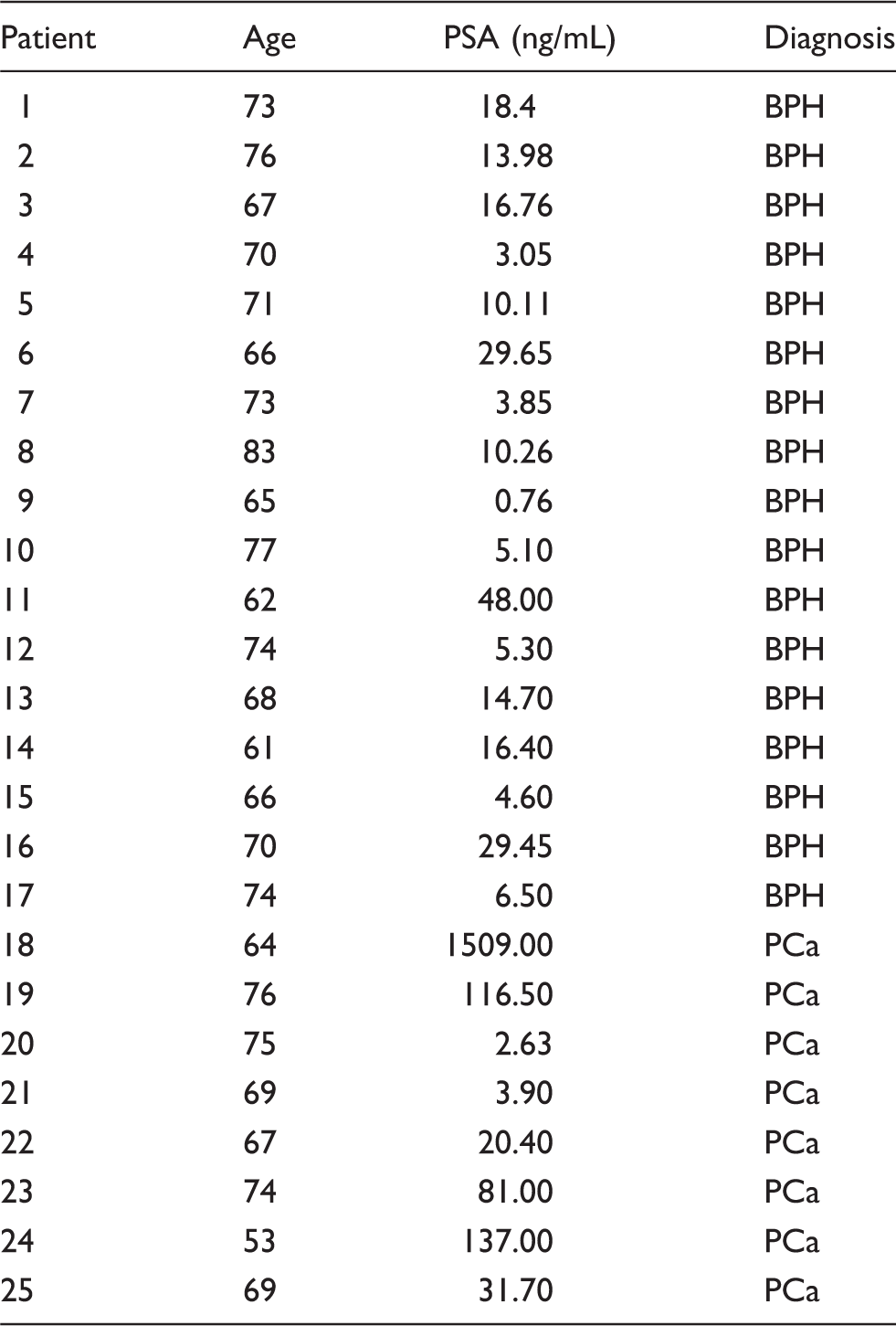
Plot of (a) uPA content, (b) PAI-1 content and (c) uPA/PAI-1 ratio in freeze-dried tissue samples as a function of the frozen aliquots from 25 patients. Solid line represents the linear regression fit of all data points for BPH (open symbols) and PCa (solid symbols) patients. Dashed line represents the 1:1 scenario where the freeze-drying process does not result in any change in marker concentration or ratio.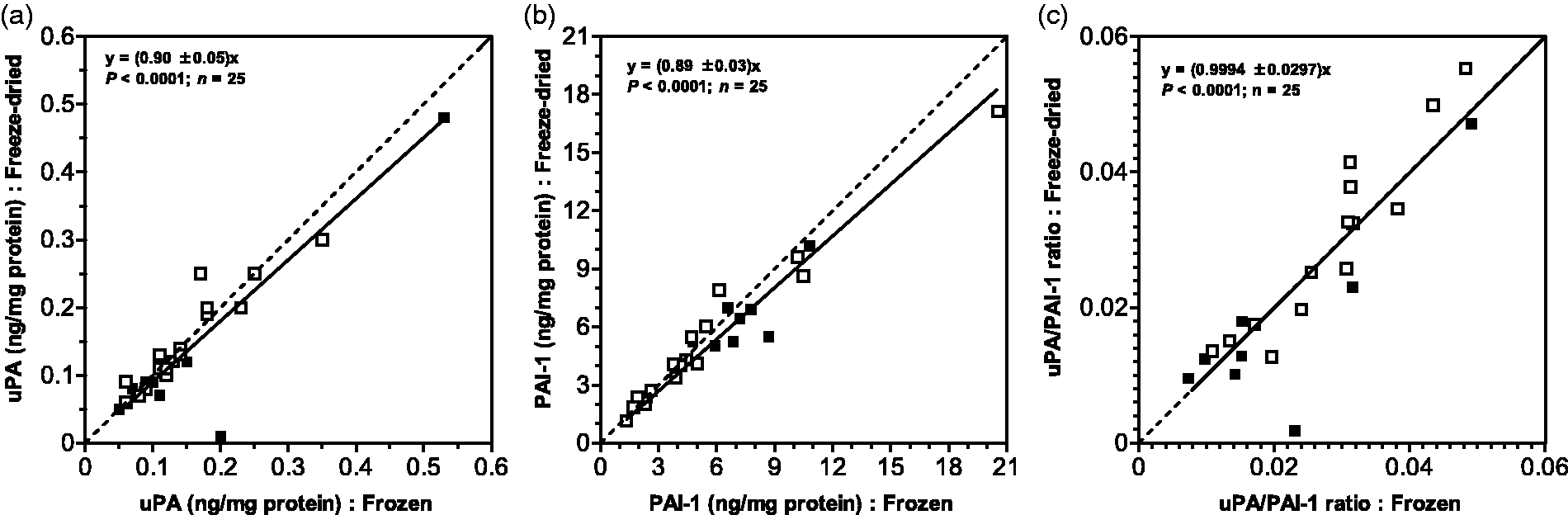
The content of uPA and PAI-1 in the frozen and freeze-dried tissue samples was determined as described previously. 5 Briefly, both sets of samples were weighed and 300 µL of extraction buffer/20 mg tissue was added. Protein was then extracted at 4℃ for 12 h on a rotating roller. The extraction buffer was composed of 240 mL pH 8.5 Tris-buffered saline and 10 mL of 25% Triton X-100 (Sekisui Diagnostics Product R22). Aliquots of the extracts were then subjected to the Pierce BCA protein assay (Thermo Scientific, Rockford, USA). Determination of the uPA and PAI-1 content was by the FEMTELLE assay (Sekisui Diagnostics, LLC, Lexington, MA, USA), as described elsewhere.6,7 Total protein was expressed as mg/mL, while the content of uPA and PAI-1 was expressed in ng/mg total protein. To test the effect of freeze-drying on the capacity of the uPA-PAI-1 pair to predict disease state, uPA/PAI-1 ratios were calculated for each patient sample and for each processing technique for further analysis.
Stability of lyophilised prostate tissue samples
To verify the stability of the lyophilised tissue samples used in this study, a cohort of samples was re-analysed for uPA and PAI-1, 30 months after storage at 4℃, using the Sekisui Diagnostics FEMTELLE kit.
Investigating the effect of detergent
The powdered prostate tissue samples were split into two aliquots. To produce tumour tissue extract (Tx), one aliquot received Tris-buffered saline (pH 8.5) and 1% Triton X-100. For the production of tumour tissue cytosol (Cyt), the other aliquot received Tris-buffered saline (pH 8.5) only. The protein was extracted overnight at 4℃ and the uPA/PAI-1 assays were performed, and the data from the two arms of the experiments compared. Aliquots of the samples described earlier were subsequently freeze-dried. After reconstitution of the samples, the protein and uPA/PAI-1 assays were repeated and the data compared.
Standard addition
To assess the recovery rate of endogenous uPA and PAI-1 contained in prostate tumour tissue powder, the markers were assayed in extracts with and without the addition of recombinant uPA and PAI-1. For this, one multiwell set received 100 µL of extract and the other received 90 µL of extract and 10 µL of uPA (0.25 ng/mL) or PAI-1 (2.5 ng/mL) standards. Samples were then assayed for uPA and PAI-1 by ELISA, adjusted for the difference in extract sample volume. Recovery rate was expressed as the ratio of marker concentration in cocktail to that in plain extract.
Data analysis
Statistical analyses were performed using the GraphPad Prism (GraphPad Software, San Diego, CA, USA) computer program. For associations, linear regression analyses were used. To compare two data-sets, the unpaired t test was used, and P values were calculated from two-sided tests. A P value of < 0.05 indicates a statistically significant difference between the data-sets.
Results and discussion
The influence of the homogenisation method was evaluated. Comparison of the punch type homogenisation with the dismembrator type of mechanical shaker using glass beads undertaken on 10 prostate tissue samples (6 BPH, 4 PCa) showed uPA values of 0.2090 ± 0.0637 and 0.3050 ± 0.1163 ng/mg protein, respectively (P = 0.4784), while the corresponding mean PAI-1 values were found to be 4.869 ± 0.9429 and 4.9080 ± 1.0430 ng/mg protein, respectively (P = 0.9782). The resulting uPA/PAI-1 ratios for the punch homogeniser and the mechanical bead shaker method were 0.0487 ± 0.0102 and 0.0606 ± 0.0123, respectively (P = 0.4680) indicating no significant difference.
The aim of freeze-drying is to ensure stability of the target molecule for storage and transport and accomplish this without compromises, i.e. losses of the target molecule. 1 In the case of the two prostate markers, losses ascertained by analysis may be identical, in which case the correlation with disease (e.g. cut-off values) may be different but will still apply. A more serious situation arises from differential losses of components. In our experiments we find the amounts of uPA and PAI-1 in the freeze-dried batches to be marginally smaller than in the frozen controls (Figure 1(a) and (b)) as indicated by slopes of 0.90 and 0.89 for uPA and PAI-1, respectively. Both uPA and PAI-1 in the frozen tissue are shown to be strongly correlated with the corresponding abundance in the freeze-dried samples with P < 0.0001 (Figure 1(a) and (b)).
Patient age, diagnosis and PSA status did not appear to have an influence on this correlation (Table 1). The data presented in Figure 1 show that freeze-drying results in a 10% reduction in the content of uPA when compared with the frozen samples. From the slope of 0.9 it is clear that the uPA content in the freeze-dried samples represents a recovery of 90% as compared to the frozen samples. For PAI-1 the recovery is shown to be 89%, indicating a loss of 11% (Figure 1(b)). It therefore follows that freeze-drying affects the operational stability of uPA and PAI-1 albeit minimally, and that the relative losses are essentially the same. A recovery of 90% is consistent with results reported elsewhere for β-galactosidase. 8 However, the recovery of lactate dehydrogenase, glucose 6-phosphate dehydrogenase and C-reactive protein has been shown to be lower and in the region of 80%.9,10 Differences in the recovery between target molecules may be attributable to differences in the handling of samples before freeze-drying, in particular temperature which promotes protein denaturation and irreversible adhesions. It is, therefore, imperative to ensure that sample processing is carried out in the mildest possible conditions.
Interestingly, when the uPA/PAI-1 ratio for the lyophilised samples was plotted against that for the frozen samples, the resultant regression closely approached a 1:1 relationship with P = 0.0001 (Figure 1(c)). This is in line with the observations that the losses of uPA and PAI-1 are essentially the same and that operational errors
1
are of no concern here. The absence of any operational bias is further demonstrated by the observation that the uPA/PAI-1 ratios for the frozen and for the lyophilised samples were found to be 0.0344 ± 0.0066 and 0.0340 ± 0.0068, respectively, further demonstrating that the lyophilisation does not affect the marker ratio (Figure 2). This is consistent with our suggestion that the uPA/PAI-1 ratio may be a suitable expression to separate aggressive and indolent prostate pathologies.
5
The utilisation of these two markers has been singularly successful in breast cancer, where the two markers are used linearly with definite cut-off points to identify early relapse and poor prognosis.6,7,11–15
Comparison of uPA/PAI-1 ratios for freeze-dried prostate tissue samples and the corresponding frozen samples from 25 patients presenting with PCa and BPH prostate abnormalities. Horizontal lines represent the mean uPA/PAI-1 ratio in each group of samples.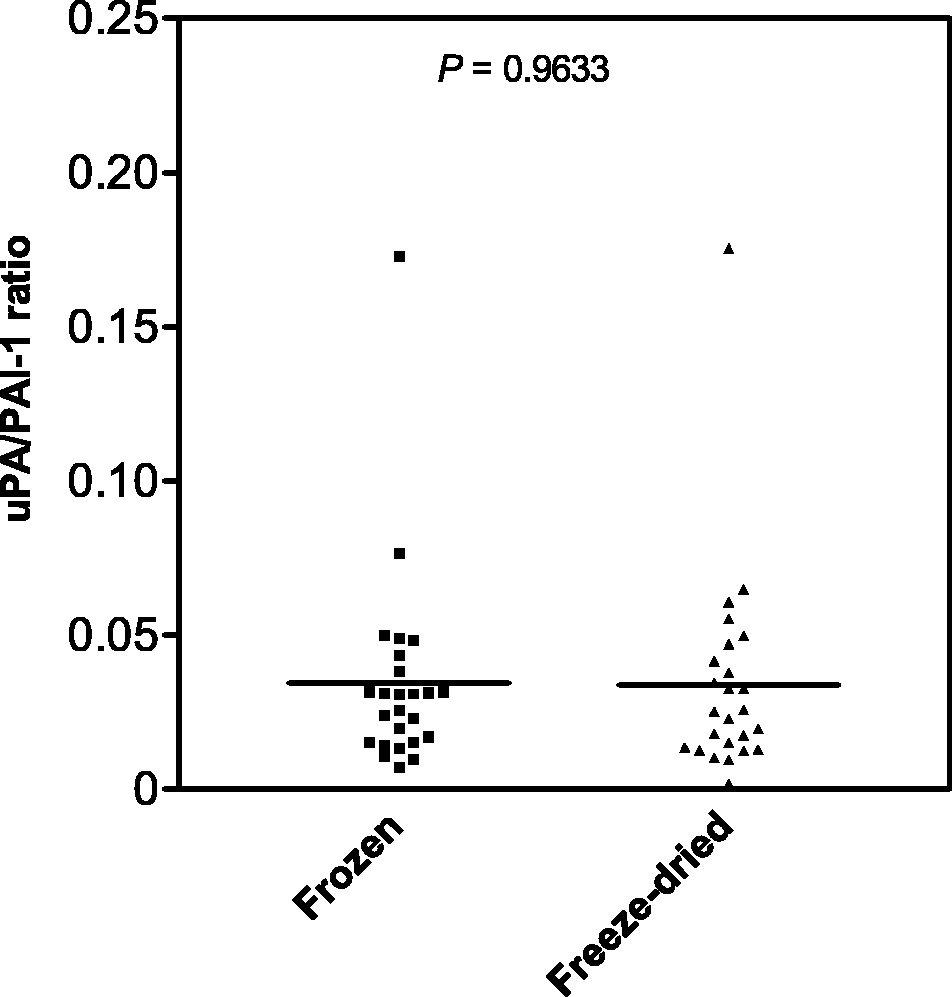
In view of the fact that the utilisation of uPA and PAI-1 titres as biomarkers for prostate cancer is still in its infancy, parameters which influence the procurement of large data collections, e.g. sample shelf life, storage conditions, extraction efficiency and marker stability will be of great interest. To assess the influence of freeze-drying on shelf life and marker recovery, a range of lyophilised prostate samples was first measured in February 2012 and again after 30 months of storage at 4℃. Figure 3 shows that the individual concentrations of uPA and PAI-1 are not significantly different and that the same applies to the uPA/PAI-1 ratio. That lyophilisation preserves uPA and PAI-1 recovery for well over two years is in agreement with the outcome of three European trials aimed at an external quality assessment of marker assays by the ELISA route, in different institutions. In this work, two lyophilised control preparations of human breast cancer tissue were found to be stable at 4℃ for up to 27 months. It also emerged that lyophilised uPA and PAI-1 standards and identical ELISA kits were key factors in minimising interlaboratory variations.
16
Much of the credit for devising successful interlaboratory comparisons in this field undoubtedly accrue to Gaffney and Heath and Gaffney and Edgell, who first demonstrated the excellent performance of lyophilised uPA and PAI-1 standards in assays on clot analysis and the implications of PAI-1 for the onset of myocardial infarction.17,18 The clinical utility of uPA and PAI-1 as prognostic markers is now part of the American Society of Clinical Oncology (ASCO) recommendations for breast cancer.
19
Influence of time on the recovery of uPA and PAI-1 from freeze-dried prostate tissue stored at 4℃.
To further investigate the behaviour of prostate tissue with regard to the release of the two markers, we also performed tissue extraction in the presence and absence of the Triton X-100 detergent. The mean uPA values were found to be higher by a factor of 2 when Triton was present (Figure 4(a)). In contrast, the presence of the detergent resulted in a 20% decrease of the mean PAI-1 recovery, but this difference was not statistically significant (Figure 4(b)). The mean uPA/PAI-1 ratio was significantly higher in the presence of the detergent (Figure 4(c)), closely resembling the values previously established for deep frozen tissue homogenate before and after freeze-drying (Figure 2). Our data on the influence of detergent on uPA and PAI-1 recovery from prostate tissue are in good agreement with the results of Jänicke et al., on breast cancer tissue, who found a two-fold increase in the extraction yield of uPA and a 10% reduction of PAI-1 when detergent was present.
6
It can therefore be concluded that prostate and breast cancer tissues do not differ as far as the recovery of the two markers is concerned. In view of the very low uPA/PAI-1 ratio generated in the absence of detergent, we adopt the current consensus view for breast cancer tissue by retaining the detergent in the extraction buffer for prostate tissue.
Influence of the absence (Cyt) and presence (Tx) of 1% Triton X-100 detergent on the recovery of uPA and PAI-1, and uPA/PAI-1 ratio.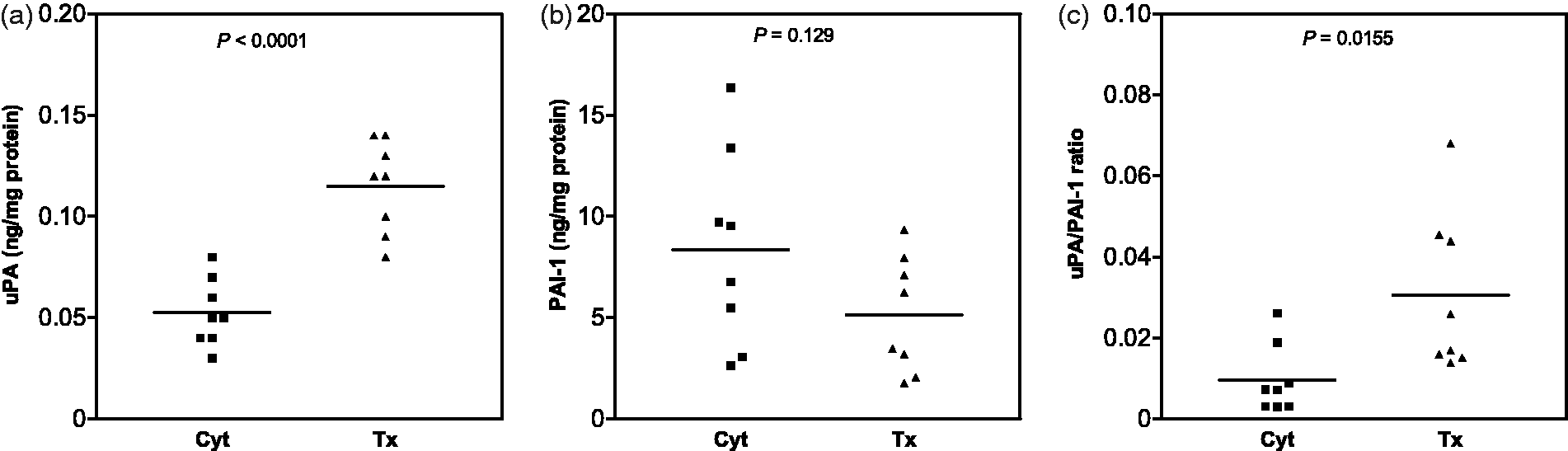
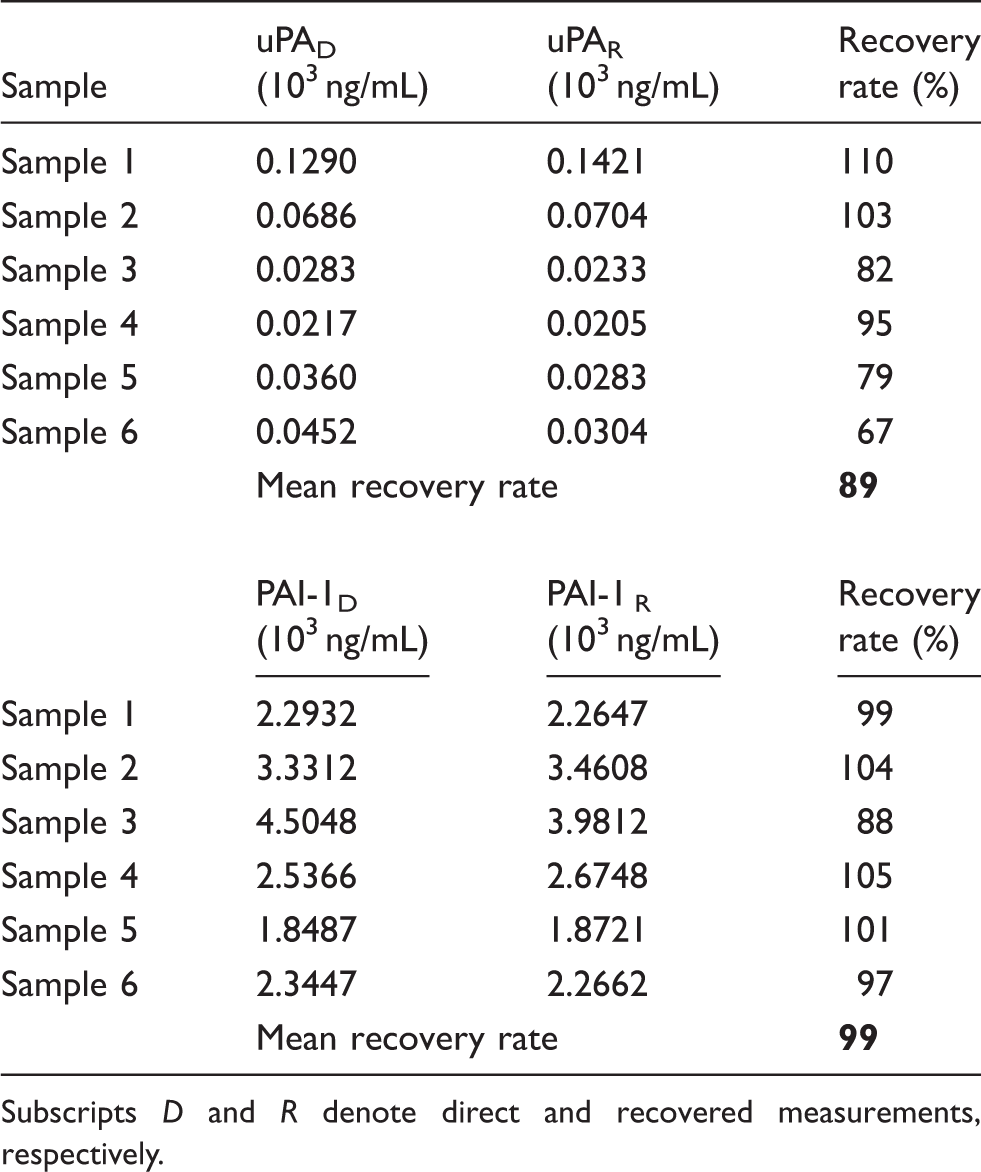
Lyophilisation and reconstitution of tumour tissue extract and cytosol do not affect the efficiency of the uPA and PAI-1 marker recovery (Figure 5). For uPA, recovery from lyophilised extracts was excellent for all samples (Figure 5(a)). Similar levels of PAI-1 recovery were evident for the majority of samples (Figure 5(b)). The uPA/PAI-1 ratio remained unaffected (Figure 5(c)). These findings further support the data in Figure 4. The recovery of the two markers was also assessed by the standard addition method using volumes of 90 µL of extract to which we added 10 µL of known amounts of standard made up in water, as defined in the FEMTELLE protocol. Table 2 depicts an experiment showing the mean recovery to be between 89 and 99% for uPA and PAI-1, respectively. This is fully in line with the predictable behaviour of the two markers as far as solubility and stability is concerned when the detergent method of extraction is adopted. The relatively low recovery of uPA is likely due to the low concentrations of this marker in prostate tissue.
Influence of freeze-drying and reconstitution of markers solubilised in the presence of 1% Triton X-100 detergent. Open symbols (Cyt) and solid symbols (Tx). Recovery of uPA and PAI-1 in samples of prostate tissue by the method of standard addition. Subscripts D and R denote direct and recovered measurements, respectively.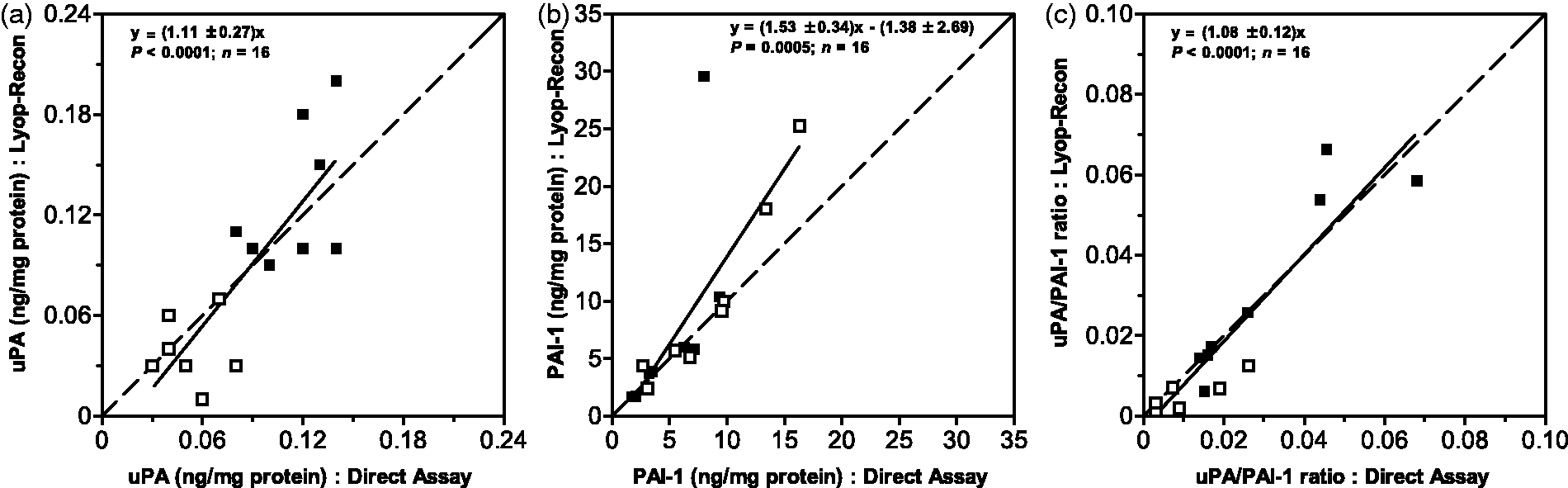
We conclude that the recovery of uPA and PAI-1 from deep frozen prostate tissue homogenate followed by freeze-drying and marker extraction by the FEMTELLE protocol proceeds with a reproducible loss of 10 and 11%, respectively, with no influence on the uPA/PAI-1 ratio. The freeze-dried tissue preparation has a shelf life of at least 30 months.
Footnotes
Acknowledgements
We thank Drs K du Toit, J Basson, H van Heerden, S Wessels, P Fernandez, A van der Merwe and C van der Walt of the Urology Department, University of Stellenbosch, and Dr NA Aziz of the Gatesville Medical Centre (Athlone, SA) for prostate resections. We are particularly indebted to Dr M Vetter for advice on the utilisation of the FEMTELLE assay.
Declaration of conflicting interests
None delcared.
Funding
Financial support was received from the Faculty of Medicine and Health Sciences (Stellenbosch University), the Cancer Association of South Africa (CANSA) to LB and the South African National Research Foundation to JMA (NRF: grants No. 85703 and No. 92741). Material and travel support from Sekisui Diagnostics (Lexington, MA, USA) to AMS is also gratefully acknowledged.
Ethical approval
This study was approved under reference NO9/11/330 of the Ethics Committee of the Faculty of Medicine and Health Sciences (Stellenbosch University, South Africa).
Guarantor
LB.
Contributorship
LB researched the literature, conceived the study and wrote the manuscript. JMA performed the data analysis. AMS collected, prepared the samples and performed the assay. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
