Abstract
Introduction
Triage of patients with suspected colorectal cancer (CRC) utilises a single faecal immunochemical test (FIT) at a defined threshold. Limited evidence exists regarding whether replicate FIT improves the positive and negative predictive value in symptomatic patients. This study examines urgently referred symptomatic patients undergoing replicate FIT. Primary aim is to assess two FITs and CRC/serious bowel disease. Secondary aims are to determine correlation and utility of replicate FIT.
Methodology
Patients carried out one additional FIT during COVID-19 pandemic. FIT 1 and FIT 2 (the replicate sample) were analysed in relation to symptoms, diagnoses, investigations, future colonoscopy and missed CRC. Study period was 01/03/2020–31/07/2020. Three subgroups were compared; double positive (≥10 μg Hb/g faeces), double negative, and discordant FIT (one positive).
Results
111 patients had replicate FIT (50 male, 61 female). 43 (38.7%) patients had double negative, 32 (28.8%) double positive and 36 (32.4%) had discordant FITs. Median time between FITs was 14 days (IQR = 11–19). 83% of double positive patients underwent colonoscopy/virtual colonoscopy (61% in double negative patients). Six CRC and one high-risk polyp were in double positive patients (none in other groups). One discordant patient was not investigated and a CRC missed.
Conclusions
Replicate FIT as a triage strategy appears most effective where both FITs are negative. CRC risk is low when FIT results are discordant. Double negative FITs are reassuring given benign associated diagnoses, or for patients where endoscopic investigation is high-risk. Larger studies are required to evaluate discordant FITs, enabling refinement of urgent investigation pathways.
Introduction
The use of faecal immunochemical tests (FIT) for triaging patients with symptoms suggestive of colorectal cancer (CRC) is currently based on a single FIT test at a defined threshold.1,2 There is a limited understanding of replicate and repeat faecal immunochemical tests in symptomatic patients. Replicate FIT describes more than one test done prior to decision-making. Repeat FIT refers to at least one additional FIT performed following a clinical decision. 3 Evidence pertaining to the performance of quantitative, replicate FIT in clinical practice is largely restricted to small studies and anecdotal evidence. 3 The authors recently published a systematic review that summarised the data for this topic and highlighted a lack of consensus regarding its role in the diagnostic algorithm. 3
A large retrospective service evaluation by Hunt et al (2022) showed a duplicate FIT strategy to be superior to single FIT (negative predictive value of 0.999 where both FIT were negative), enabling a significant proportion of suspected colorectal cancer patients to be safety-netted and managed in primary care. 4 Similarly, a large Scottish multicentre retrospective observational study highlighted that serial FIT is already being undertaken in nearly 10% of primary care patients. 5 The authors state that two faecal haemoglobin results of less than 10 μg Hb/g faeces (μg/g), in a 12-month period, confers a very low CRC risk (0.1%). Furthermore, 0.4% of the 944 patients with a f-Hb ≥ 10 μg/g followed by f-Hb < 10 μg/g were found to have CRC. Therefore, performing two FIT tests within 2 months could “add an additional layer of safety netting”. 5
There is a growing need to deliver cost effective strategies to tackle mounting urgent referrals and the associated burden on endoscopy services. 6 Repeat and replicate FIT tests may be utilised by clinicians for reassurance, decision to investigate, or surveillance. However, as demonstrated by the omission of replicate FIT from the recent joint guidance issued by the Association of Coloproctology of Great Britain and Ireland (ACPGBI) and British Society of Gastroenterology (BSG), little understanding exists pertaining to the role that replicate FIT plays in clinical decision-making. 2 Current National Institute for Health and Care Excellence (NICE) guidelines recommend a single FIT at a threshold of 10 μg/g for referral. 7 This recommendation was endorsed in recent guidance published by BSG/ACPGBI. 2
This study looks to examine the utility of replicate FIT at a single centre in a cohort of urgently referred patients. The primary aim is to assess the correlation between two FIT values and diagnosis/presence (or absence) of CRC, or serious bowel disease (CRC and high-risk polyps). Secondary aims are to determine the demographics and presenting symptoms of this cohort.
Methodology
Data from the CRC database, held at Royal Surrey Hospital, relating to all urgent referrals were accessed. Analysis of patients from 01/03/2020 to 31/07/2020 identified those with more than one FIT result. These dates were chosen as a new referral pathway was implemented at our centre during the COVID pandemic. In addition to the FIT (FIT 1) undertaken in primary care prior to referral, packs containing an additional FIT (FIT 2) were sent out to all patients referred on the urgent referral pathway. 8 These included instructions for use, an OC-Auto Sampling Bottle, (Eiken Chemical Co Ltd, Tokyo, Japan) and a pre-paid envelope. Patients were asked to collect the samples for f-Hb (FIT 2) in a dated OC-Auto sampling bottle, before returning via post to the biochemistry laboratory at the Royal Surrey Hospital. Samples were analysed using an OC-Sensor™ Pledia analyser (Eiken Chemical Co Ltd, Tokyo, Japan). The limit of detection is 1 μg/g and limit of quantification is 6 μg/g. In line with national guidance, 7 a f-Hb less than 10 μg/g was defined as negative, and a f-Hb greater than or equal to 10 μg/g faeces as positive. This pathway was introduced to determine those who required urgent assessment and endoscopic investigation.
Data from patient letters and investigation requests was collected relating to; demographics, FIT 1 value (μg/g), FIT 2 value (μg/g), presenting symptoms (abdominal pain, weight loss, change in bowel habit (CIBH), per rectal bleeding), anaemia, endoscopic, radiological and histological findings. All patients with two FIT results were included, even if the patient was not further investigated, or no final diagnosis was established. Medical records were checked 24 months after the initial FIT to ensure patients were not subsequently diagnosed with serious bowel pathology. Analysis of data comparing three groups was undertaken: double negative FIT, double positive FIT and discordant FIT (patients with one positive and one negative result). Comparison of FIT was made with endoscopic, radiological and histological findings. Potential missed CRC were identified by cross-referencing the endoscopic database at our centre (until 01/07/2022).
Statistical analysis was undertaken using GraphPad Prism v8.4.3. Bland-Altman plots were used for agreement between FIT 1 and FIT 2. Where appropriate, FIT values greater than 500 μg/g were capped for graphical demonstration. The paper was written in line with STROBE guidelines. 9
Results
In total, 727 patients were referred to our centre, of whom 111 patients (50 male, 61 female) returned two FIT samples and were included in the study cohort. Median overall age was 71 years, ages ranged from 32 to 93 years (range = 61), median female age was 68 years, ages ranged from 32 to 93 years (range = 61) and median male age was 76 years, ages ranged from 36 to 89 years (range = 53). The double negative FIT group contained 43 (39%) patients; the double positive FIT group 32 (29%), and a further 36 (32%) patients had discordant results (either a positive FIT 1 and negative FIT 2, or a negative FIT 1 and positive FIT 2). The median time between FIT 1 and FIT 2 was 14 (IQR 11–19) days.
Diagnoses categorised by FIT outcomes.
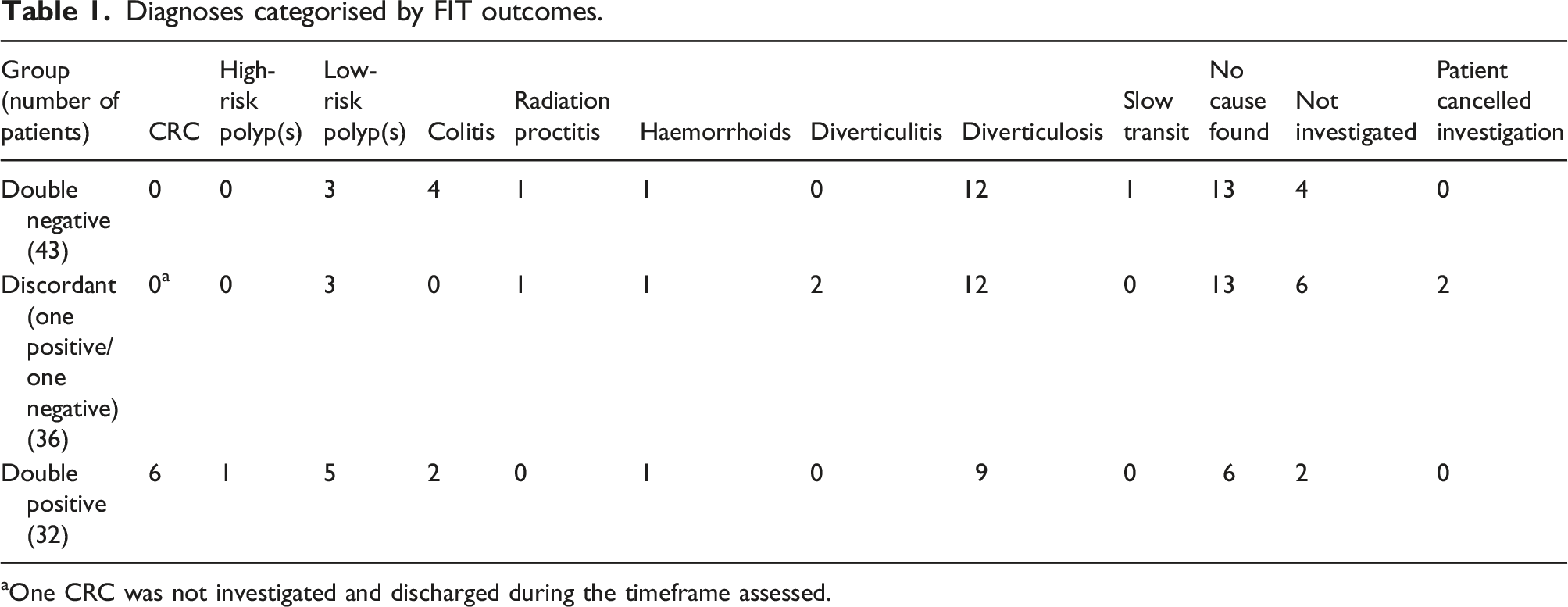
aOne CRC was not investigated and discharged during the timeframe assessed.
Data (excluding double negative FIT patients) was analysed to assess the relationship between a positive or negative FIT 2 and the haemoglobin concentration of FIT 1 (Figure 1). Patients (n = 30) who had a negative FIT 2 had a lower median FIT 1 concentration (23.5 μg/g), interquartile range (IQR) 14–73 μg/L. Those patients (n = 38) with a positive FIT 2 had a higher median FIT 1 concentration (30 μg/g) and a wider range, IQR = 12–341 μg/L. Several elevated outliers were present in this group. Mann–Whitney U test was performed which did not demonstrate any difference between the two groups, p = 0.27. Box and whisker plot of FIT 1 concentration and FIT 2 outcome.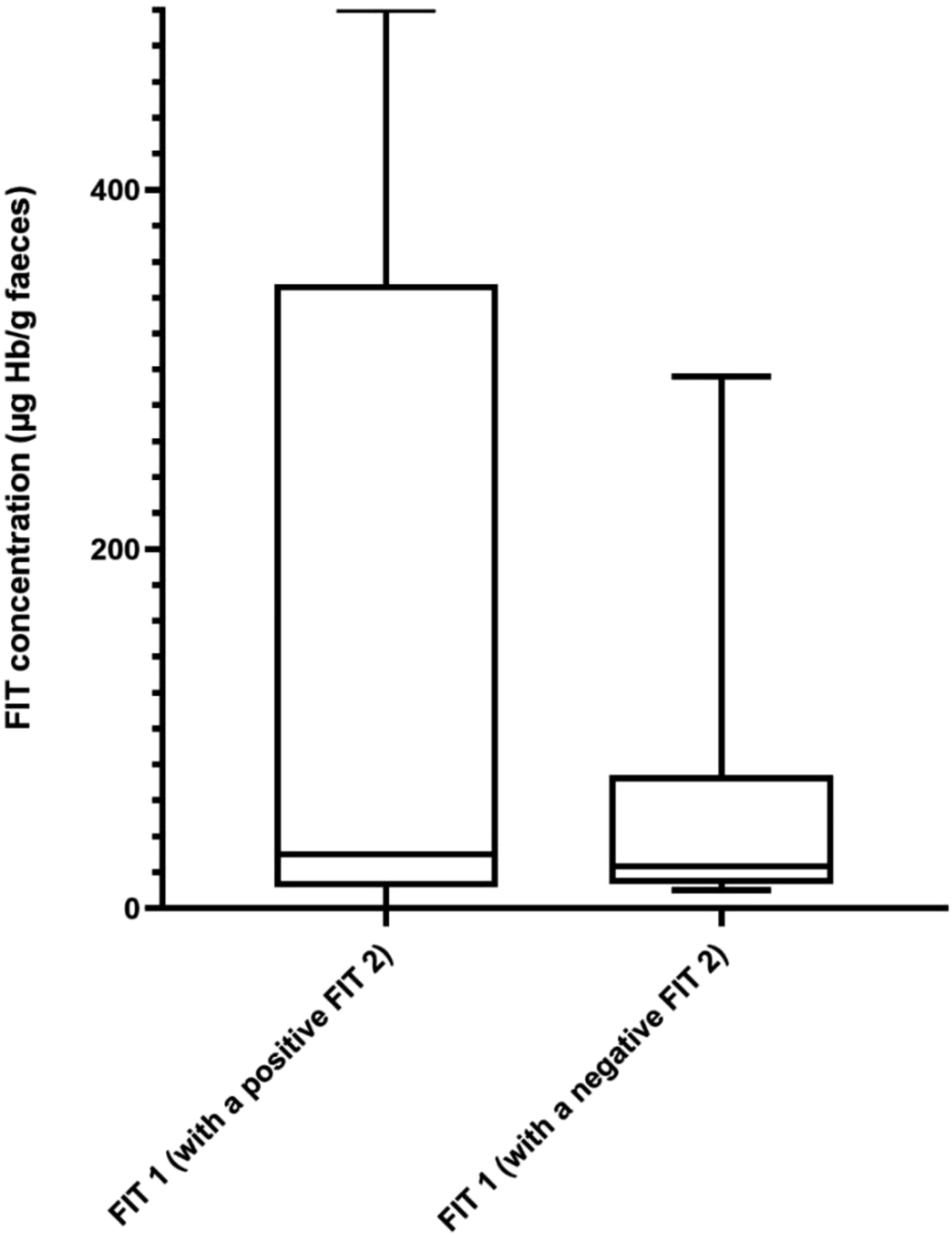
A Bland–Altman plot (Figure 2) was used to demonstrate the repeatability of FITs 1 and 2. The level of agreement was wide (95% limits of agreement −5588–6160 μg/g) and bias was 286.1 μg/g. One extreme outlier was excluded to allow for graphical representation (FIT 1 = 3962 μg/g, FIT 2 = 20,165 μg/g). Bland–Altman plot of double positive FIT results.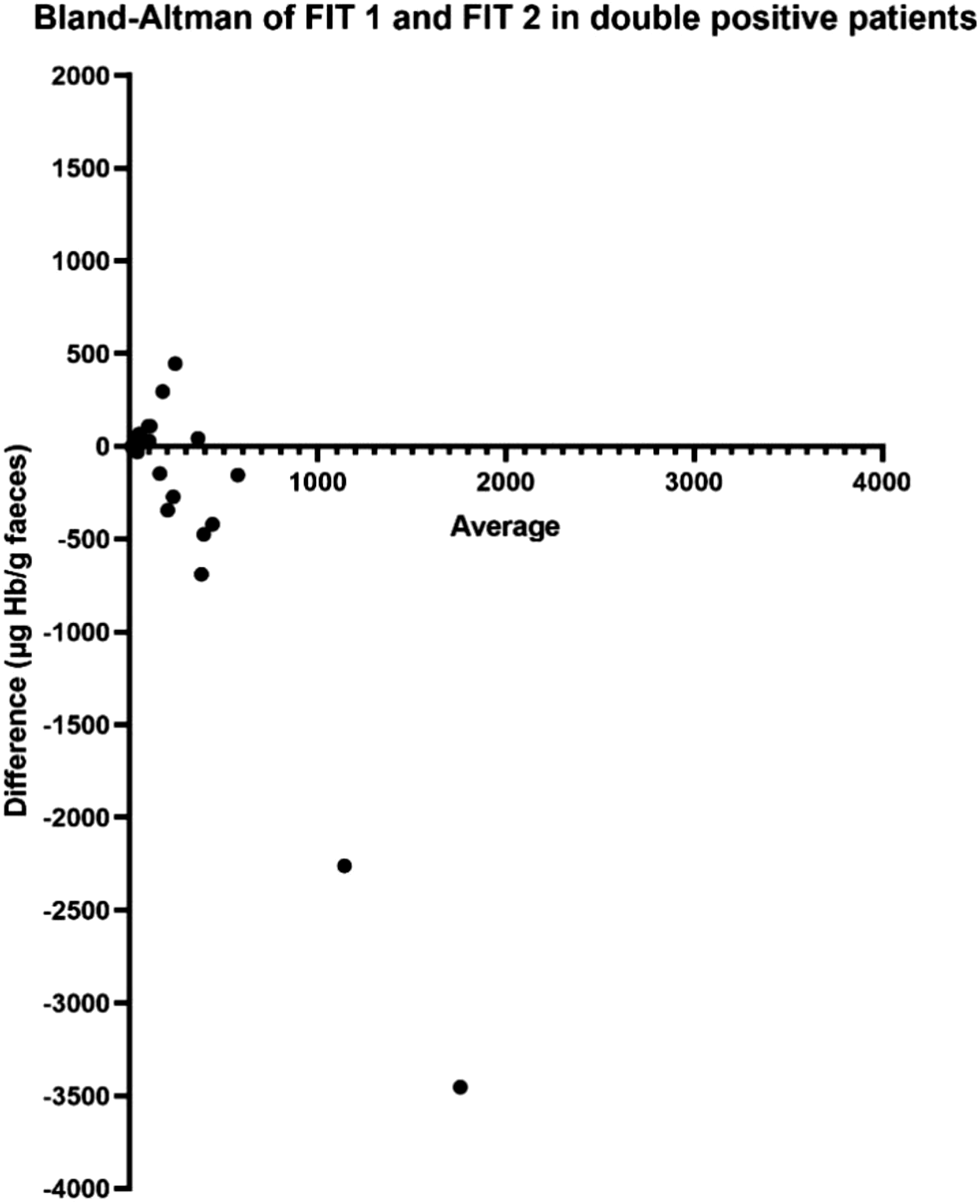
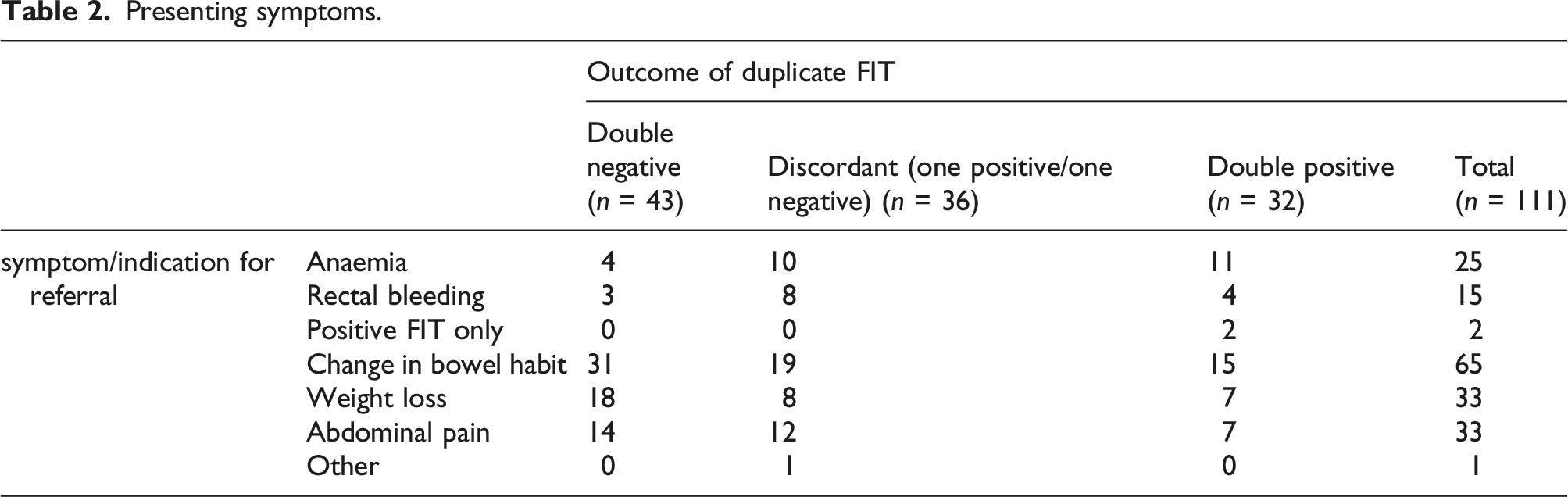
Presenting symptoms.
Discussion
In this study, patients with a double positive FIT result have more serious underlying pathology than those with two negative FIT, or discordant FIT. All CRC and high-risk polyps diagnosed in the cohort had a double positive FIT. This suggests that there should be a high level of clinical suspicion for serious bowel pathology within this group.
Auge et al. evaluated the diagnostic yield of two FIT samples in 208 patients and concluded that in 29 patients with advanced colorectal neoplasia (ACRN), FIT/1 and FIT max (the higher concentration of two FIT samples) were significantly higher than in patients with low-risk adenoma and normal colonoscopy. 10 Douglas et al. detected six CRC within a cohort of 746 patients with two FIT results. 11 Three had both FIT less than 80 μg/g and three had both FIT greater than 80 μg/g. The retrospective cohort study by Hunt et al. highlighted 86 patients (0.3%) with CRC and two FIT less than 100 μg/g. 60 patients (4.2%) had CRC with one FIT greater than 100 μg/g and one FIT less than 100 μg/g, 14% (173 patients) had two FITs greater than 100 μg/g. 4 The positive predictive value (PPV) and likelihood ratio was higher when both FITs were positive than one positive FIT only.
In our study, 39% of the total cohort were double negative. Given that the final diagnoses in this subgroup represented benign, or no underlying pathology, investigation may be less urgent or even unnecessary for such patients.
The relationship between a low positive (or negative) FIT 1 and a subsequent negative FIT 2 is highlighted in Figure 1. A lower FIT 1 (when compared to a more positive FIT 1 value) is usually followed by a negative FIT 2. In this discordant subgroup, six patients (18%) had a negative FIT 1 result followed by a positive FIT 2. The FIT 2 values of these patients were 14, 15, 23, 26, 39, and 50,000 μg/g, respectively. The patient with the significantly elevated FIT 2 had a diagnosis of radiation proctitis following flexible sigmoidoscopy. Therefore, larger studies are needed to determine whether an alternative threshold for FIT 2 could be used to improve triage in secondary care.
Furthermore, the management of this group may have differed if only the first FIT result was available when determining whether a patient should be reassured or investigated. This would not have resulted in any missed CRC in this group. However, the findings of Högberg et al. highlight the potential for missed CRC and adenomas in larger cohorts when management relies on a single FIT result. 12 The false negative rate of FIT is well documented at 10%. 13 Replicate FIT testing may help reduce this rate as indicated by the results of Hunt et al., where 0.04% of patients with two negative tests had CRC, compared to 0.5% in patients with a single positive and 5.2% in those with two positive FIT results. 4
It is evident for double positive patients that replicate FIT has less clinical utility in selecting for investigation. The presence of a single significantly elevated FIT (>100 μg/g) raises clinical suspicion for serious pathology and therefore mandates investigation. We are unable to determine from this small observational study whether a low positive FIT combined with a negative can downgrade or adjust clinical suspicion. When one FIT is significantly elevated, even if the other is negative, concern should remain.
The commonest presenting symptoms in double FIT negative patients were change in bowel habit, abdominal pain and weight loss. In the double FIT positive group, 34% of patients had anaemia, which was higher than in other groups. The paper by Chapman et al. reported that (single FIT) patients, with anaemia, had significantly higher FIT values, 14 whilst Mashlab et al. report that anaemic patients were three times more likely to have CRC than non-anaemic. 15
Given that microscopic bleeding can be transient and vary in volume and additional pre-analytical factors can impact f-Hb concentrations, 16 the outlying FIT 1 and FIT 2 values of 3962/20,165, 3485/31 and 2272/11 μg/g observed in Figure 2 are to be expected. This data contributes to the limited evidence base regarding repeatability of FIT in the symptomatic population. Whilst over half of the cohort had a FIT 1 and FIT 2 within 50 μg/g of each another and a quarter within 10 μg/g, there remains the potential for wide variation between samples. This highlights the risk of significant pathology being missed if management is reliant on single FIT.
The prevalence of benign conditions such as diverticulosis were evenly distributed between groups. A cause was not found in 19% of patients with double positive FIT; this was higher in the double negative (30%) and discordant (36%) groups. The double positive FIT cohort had a higher prevalence of underlying pathology, supporting the suggestion that less urgent investigation may be acceptable only in those with double negative FIT.
This cohort supports studies that advocate the introduction of safety netting and reassurance for patients, particularly with double negative FIT. 17 This may enable endoscopy services to be utilised in a targeted manner, providing a more effective service for those symptomatic patients at high risk. Triaging non-urgent patients who still require investigation will enable urgent referral pathways to function more efficiently despite increasing demand. Miller et al. highlight a service where two FIT results (<80 μg/g) with low-risk symptoms served as reassurance and investigation was not undertaken. 18
Three patients with double negative FIT had colitis, although no inflammation was seen at time of colonoscopy. Microscopic lymphotcytic colitis, collagenous colitis and “mild acute inflammation” were diagnosed following biopsy. A fourth patient, known to have collagenous colitis, was treated without repeat investigation. Two patients in the double positive group had campylobacter and microscopic colitis. It remains difficult to ascertain the role of FIT in the diagnosis of colitis, particularly without overt inflammation or bleeding.
12 patients with reassuring FIT and low-risk symptoms were not investigated. One patient was reassured and discharged with an initial positive FIT (25 μg/g) and a further negative FIT. The patient presented with blood in his urostomy bag on a background of bladder cancer having undergone a cystoprostatectomy in 2011. There were no constitutional bowel symptoms at the time of presentation. 10 months later a CRC was diagnosed. We advise caution for management based solely on two FIT samples. The pandemic is likely to have had a significant impact on this case given restricted availability of endoscopy. However, a single missed cancer illustrates the need to consider the patient as a whole, irrespective of FIT value. Correlation with symptoms, blood tests and examination is still required and where concerns remain, investigation is warranted.
Currently, we are unable to determine whether replicate discordant FIT results can be managed differently in symptomatic patients. There is a need for reproducible studies with greater power to enable further analysis of such patients, specifically, when a positive FIT 1 is followed by a negative FIT 2. Developing this understanding may enable additional refinement of urgent investigation pathways. Similarly, more evidence is required to support the triage of double negative FIT patients away from endoscopy. Addressing these areas will support clinical decision-making and offers an effective strategy, particularly for those patients in whom endoscopic investigation is high risk or not clearly indicated.
There are limitations to this study. The small data set limits the power and reliability of the results. The study is written in retrospect, consequently, the non-standardised recording of data and reliance on patient letters and investigation requests may result in missing or inaccurate data. The results of this study require contextualisation given that the data was taken from a period during the COVID-19 pandemic when clinical practice was significantly modified. Referrals to secondary care may have changed during the time period studied; the symptom profile observed may not be truly representative of urgently referred patients pre-pandemic. Consequently, the conclusions of this paper are tempered somewhat by these factors and interpretation of the data should be undertaken cautiously.
Conclusion
Replicate FIT as a triage strategy appears most effective where both investigations are negative or discordant. An initial elevated FIT 1 will likely to lead to onward investigation, thereby rendering a replicate FIT less useful. The promising outcomes support replicate FIT as a reassuring adjunct when two values are negative given the benign associated diagnoses. With further research, it is possible that replicate FIT will play a significant role in delivering effective and safe patient centred strategies to address the increasing number and cost of urgent symptomatic referrals.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
No ethical approval was required for this paper.
Guarantor
SCB.
Contributorship
NGF, SCB conceptualisation of study; NGF data collection; NGF and JO'B analysis and write up of manuscript. MW, IJ, TR, SCB review and editing of manuscript.
