Abstract
Background
Faecal calprotectin has been shown to be useful as a non-invasive screening test to differentiate functional from organic bowel disease, and it has been noted to be elevated in colorectal cancer. The aim of this study was to describe concentrations of faecal calprotectin in patients with oesophago-gastric cancer and investigate any potential discriminatory power of the test.
Patients
Faecal calprotectin was measured in samples from 39 patients with known oesophago-gastric cancer and in 191 samples from control subjects.
Results
The median calprotectin concentration was < 20 µg/g (range < 20–421 µg/g) in control subjects and 97 µg/g (range < 20–940 µg/g) in patients with oesophago-gastric cancer (P < 0.001). A receiver operating characteristic curve gave an area under the ROC curve of 0.84 and a sensitivity of 76.9% (95% CI: 63.7–90.1%) and specificity of 88.0% (95% CI: 83.3–92.6%) at a cutoff of 50 µg/g.
Conclusion
Faecal calprotectin is elevated in patients with cancer of the upper gastrointestinal tract. This study suggests that calprotectin may be promising in discriminating cancer patients from controls, but further work is required to explore any potential role of faecal calprotectin in screening for, or diagnosis of, oesophago-gastric cancer.
Introduction
Faecal inflammatory markers have been shown to be useful as non-invasive screening tools to differentiate patients with functional from organic bowel pathology. Of these markers, calprotectin has been the most intensively studied. Calprotectin is a calcium- and zinc-binding protein, a member of the S100 family. It is derived predominantly from neutrophils and constitutes about 60% of their cytosolic proteins. 1 It is found in various body fluids in proportion to the severity of any existing inflammation. Calprotectin has bacteriostatic and fungistatic properties and is also able to inhibit fibroblastic cell growth and to induce apoptosis in various host cells. 2 Faecal calprotectin tends to be significantly elevated in patients with inflammatory bowel disease (IBD) compared with healthy subjects, but for clinical purposes it is particularly useful as a marker of inflammation in patients presenting with similar symptoms, particularly those presenting with irritable bowel syndrome. Using statistical techniques, a pooled sensitivity of 93% and specificity of 96% have been calculated for the use of calprotectin in the diagnosis of IBD. 3
Subsequently, attention turned to whether calprotectin could be of use in the differentiation of organic from functional bowel disease. Even though not all organic bowel diseases would be expected to be associated with an inflammatory response, mean sensitivity and specificity have been calculated to be 83% and 84%, respectively, for use of calprotectin in the diagnosis of organic intestinal disease. 4 Faecal calprotectin also has been found to be elevated in patients with colorectal cancer.5–7
The aim of this study was to describe concentrations of faecal calprotectin in patients with oesophago-gastric cancer and controls and to investigate any potential discriminatory power of the test.
Methods
Patients
Patients with a history of oesophago-gastric cancer were asked to provide a faecal sample for measurement of calprotectin. Samples were taken at least two weeks after any endoscopic procedure and prior to any definitive treatment for the malignancy being given. Patients undergoing surgery for uncomplicated abdominal wall hernia acted as controls and provided a faecal sample prior to operation. Patients and controls were excluded if they had previous investigation or diagnosis of IBD or other gastro-intestinal disease, co-existing serious illness or medication known to be associated with intestinal inflammation. 8
Sample analysis
Samples were received in the laboratory with minimal delay and certainly within 24 h. On receipt, samples were frozen at–20 C prior to analysis which was performed within six months. One hundred milligrams of faeces were extracted in 5 mL of extraction buffer. The supernatant was analysed by an enzyme-linked immunosorbent assay (Phical, Calpro, Lysaker, Norway) using wells coated with polyclonal rabbit antibody and immunoaffinity purified ALKP-labelled anti-calprotectin, with detection of the enzyme reaction with a substrate at 405 nm. The limit of detection of the assays was <20 µg/g. The manufacturer states a cutoff for calprotectin of <50 µg/g faeces. Previously published age-related reference ranges for healthy subjects are <51 µg/g in those aged 10–50 years and <112 µg/g in those aged ≥60 years. 9
Statistical analysis
For statistical purposes, samples below the limit of detection of the assay were set at this lower limit. Calprotectin concentrations in the two disease groups (cancer and control) were summarized in terms of median and range and compared using a Wilcoxon rank-sum test. The discriminatory power of calprotectin was investigated using empirical estimates of area under the ROC curve (AUC) and sensitivity and specificity, with 95% CI, for a given cutoff value. Statistical analysis was performed using R (version 3.0.2). 10
Results
Demographic characteristics of oesophago-gastric cancer patients and control subjects.
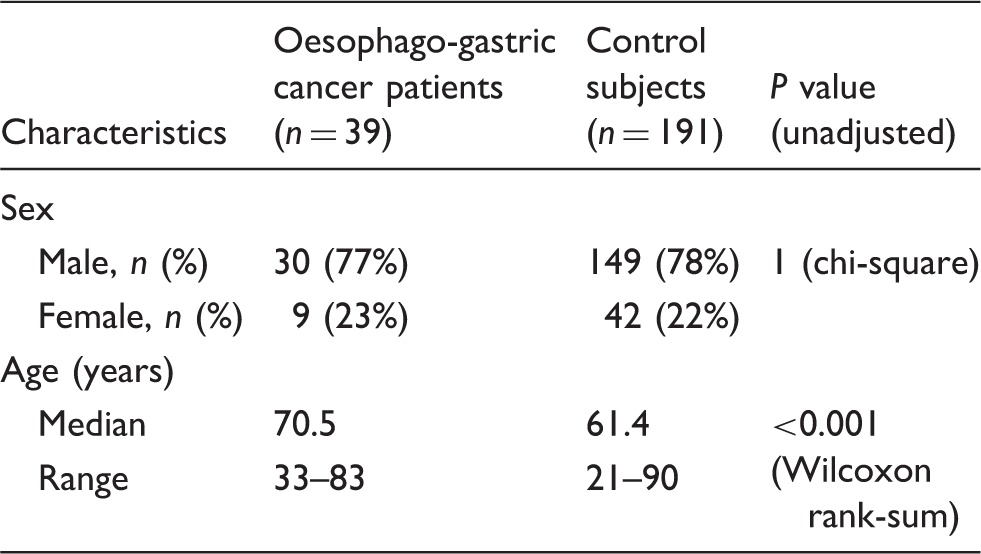
The concentrations of calprotectin in the faecal samples from the patients and controls are shown in Figure 1. For the control subjects, the median faecal calprotectin concentration was <20 µg/g (range < 20–421 µg/g), and for the cancer patients, the median calprotectin concentration was 97 µg/g (range < 20–940 µg/g) (P < 0.001, Wilcoxon rank-sum test). Using the manufacturer’s cutoff, faecal calprotectin was elevated in 30 (22 males, 8 females) of the 39 patients with oesophago-gastric cancer and in 23 controls (19 males and 4 females). Using age-related reference ranges, faecal calprotectin was elevated in 22 patients and 13 controls. Faecal calprotectin was elevated in patients with oesophago-gastric cancer in both early and late disease (Figure 2).
The concentration of calprotectin in the faeces of oesophago-gastric cancer patients (n = 39) and control subjects (n = 191). Concentrations < 20 µg/g are set at 20 µg/g (n = 125; six cancer patients, 119 controls). The concentration of calprotectin in the faeces of oesophago-gastric cancer patients classified according to the stage of their disease.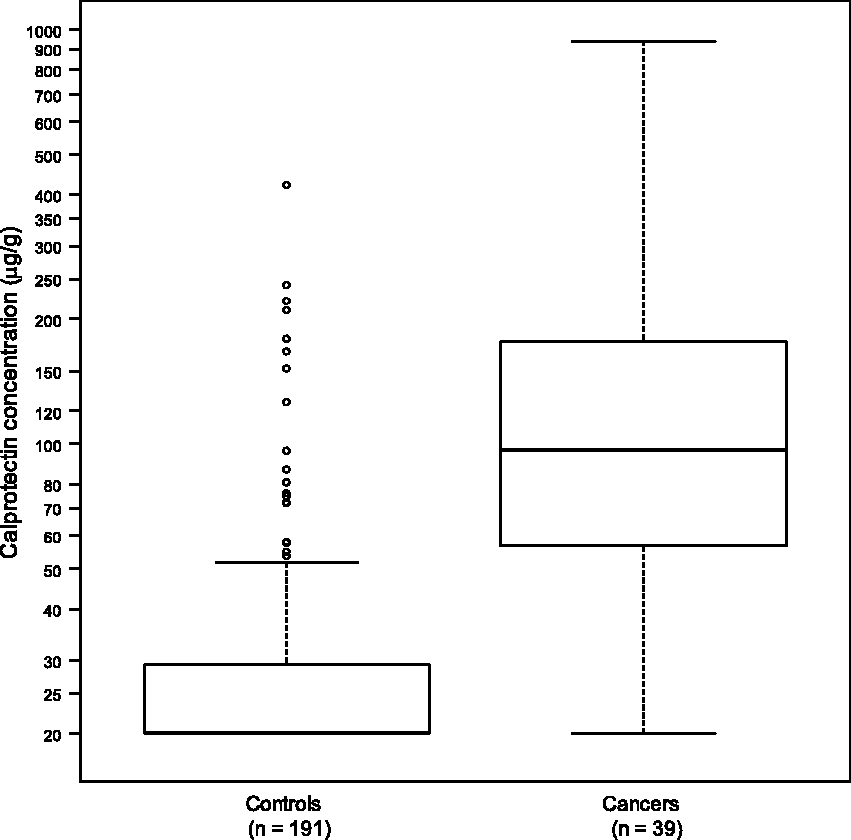
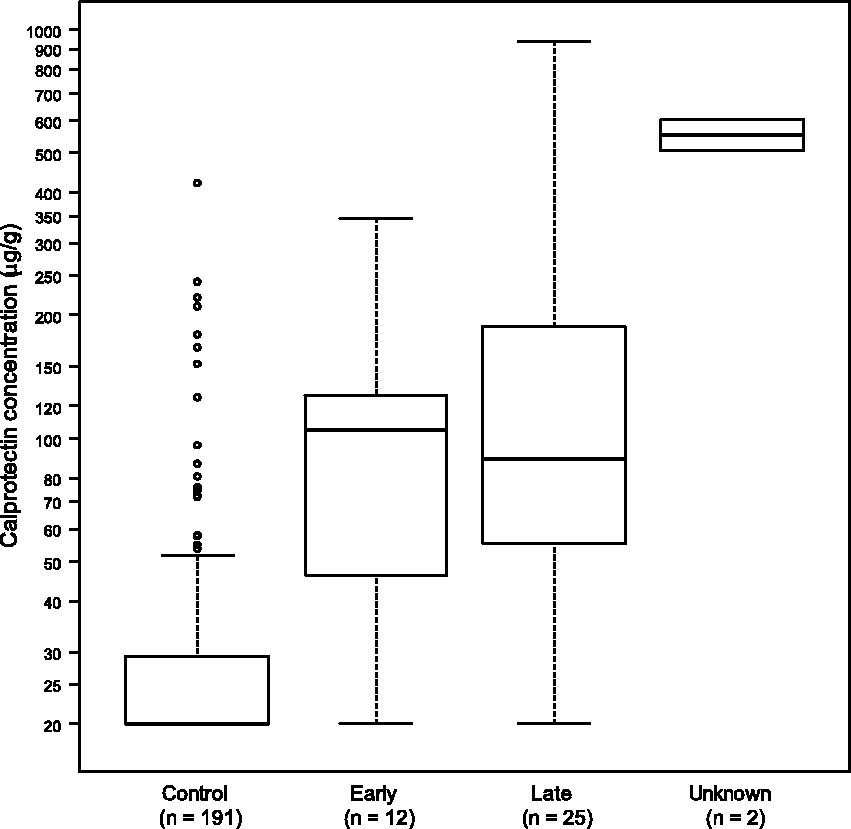
Figure 3 shows a ROC curve for calprotectin in discriminating between cancers and controls. At the manufacturer’s cutoff of 50 µg/g, the sensitivity and specificity of faecal calprotectin in the diagnosis of oesophago-gastric cancer were calculated to be 76.9% (95% CI: 63.7–90.1%) and 88.0% (95% CI: 83.3–92.6%), respectively. The area under the curve was 0.84. Increasing the cutoff to 55 µg/g maintained sensitivity and increased specificity marginally to 89.5% (95% CI: 85.2–93.9%). Using age-related cutoffs lowered sensitivity to 56.4% (95% CI: 40.8–72.0%) but increased specificity slightly to 93.2% (95% CI: 89.6–96.8%). Any differences in specificity were not statistically significant.
Receiver operating characteristic curve for calprotectin in the diagnosis of oesophago-gastric cancer. Vertical and horizontal dotted lines indicate the performance achieved at a cutoff of 50 µg/g.
Discussion
The main diagnostic use of calprotectin is as a surrogate marker of inflammation in the diagnosis and management of IBD. Calprotectin has also been found to be elevated in patients with colorectal cancer. In 814 patients at high risk for colorectal cancer, undergoing colonoscopy, faecal calprotectin was found to have a sensitivity for cancer of 74% and for adenoma of 43%. 5 A study, which measured calprotectin in 62 patients with known colorectal cancer and 233 patients referred for colonoscopy with features suggestive of the disease, found the overall sensitivity and specificity for cancer and adenomatous polyps to be 79% and 72%, compared with 43% and 92%, respectively, for guaiac faecal occult blood testing. 6 A study specifically looking at calprotectin in the context of population screening in 2321 asymptomatic individuals found a sensitivity of 76% for any neoplasia and 27% for advanced neoplasia, compared with 90% and 35%, respectively, for immunochemical faecal occult blood testing. 7 However, the results of a meta-analysis involving 297 patients concluded that the overall sensitivity and specificity of faecal calprotectin for the diagnosis of colorectal cancer were 36% and 71% and that the test could not be recommended for use as a screening test in the general population. 11
Although calprotectin is present in blood, it has been estimated that blood loss of more than 300 mL/24 h would be required to produce significant elevation in faecal calprotectin. 12 Any increase in faecal calprotectin observed in patients with gastrointestinal cancer is likely to represent polymophonuclear cell infiltration of the tumour, with subsequent shedding into the bowel lumen; certainly colon cancer may be associated with a local acute inflammatory reaction that can be visualized by white cell neutrophil scanning. 13
The data presented here have shown that faecal calprotectin has a sensitivity of 76.9% (95% CI: 63.7–90.1%) and a specificity of 88.0% (95% CI: 83.3–92.6%) in the diagnosis of oesophago-gastric cancer, using the manufacturer’s cutoff of <50 µg/g. This cutoff is in widespread use in screening for gastrointestinal inflammation and has been used in the investigation of calprotectin in colorectal cancer. 6 This adds to the previous reports of elevated faecal calprotectin in patients with upper gastrointestinal malignancy.14–16
An early study noted two patients with gastric and one with oesophageal cancer to have elevated calprotectin, 14 and the same group later reported calprotectin to be elevated in all 20 of a series of newly diagnosed patients with gastric cancer. 15 Of 116 patients studied, when attending for endoscopy, two with elevated calprotectin were found to have upper gastrointestinal cancer. 16 Twenty-two patients with oesophageal polyps or neoplasms were found to have calprotectin concentrations significantly higher than healthy controls and patients with non-malignant disorders (chronic gastritis/ulcer/pancreatitis). 17
In the UK, a national screening programme has been introduced for colorectal cancer with colonoscopy offered to those who are positive on initial faecal occult blood testing 18 but, due to the relatively low incidence of oesophago-gastric cancer, population screening by endoscopy is not feasible. It may be that other screening tools, such as use of the ingestible Cytosponge, 19 may prove a viable option or that further definition of populations at additional risk will enable targeted screening.
Further work is required to explore the potential of faecal calprotectin in screening for, and diagnosis of, oesophago-gastric cancer.
Conclusion
Faecal calprotectin is used as a surrogate marker of inflammation, particularly in the diagnosis and management of IBD. However, calprotectin may be raised in various other gastrointestinal pathologies including colorectal cancer. This study confirms previous reports that faecal calprotectin is elevated in gastric cancer, and this finding merits further study. Oesophago-gastric cancer should be considered as a potential cause of a raised calprotectin, particularly in patients with normal colonoscopy findings.
Footnotes
Declaration of conflicting interests
None declared.
Funding
Steve Hornby received a grant from the Bardhan Research and Education Trust.
Ethical approval
This study was approved by the South West Regional Ethics Committee reference code 10/HO102176.
Guarantor
RMA.
Contributorship
All authors contributed to the design and execution of the study. All authors reviewed and edited the manuscript and approved the final version.
