Abstract
Background
Assessing prognosis is important in patients with neuroendocrine tumours of the small bowel as disease progression and survival is variable. We previously identified raised Neurokinin A as an independent indicator of poor prognosis and have shown that prognosis worsens when circulating Neurokinin A rises ≥50 ng/L. In the present study we have examined survival in relation to Neurokinin A concentrations.
Methods
Patients in whom Neurokinin A rose ≥50 ng/L between January 1989 and December 2010 were identified. All circulating Neurokinin A concentrations were recorded and survival was followed up to 31 December 2014 or to death.
Results
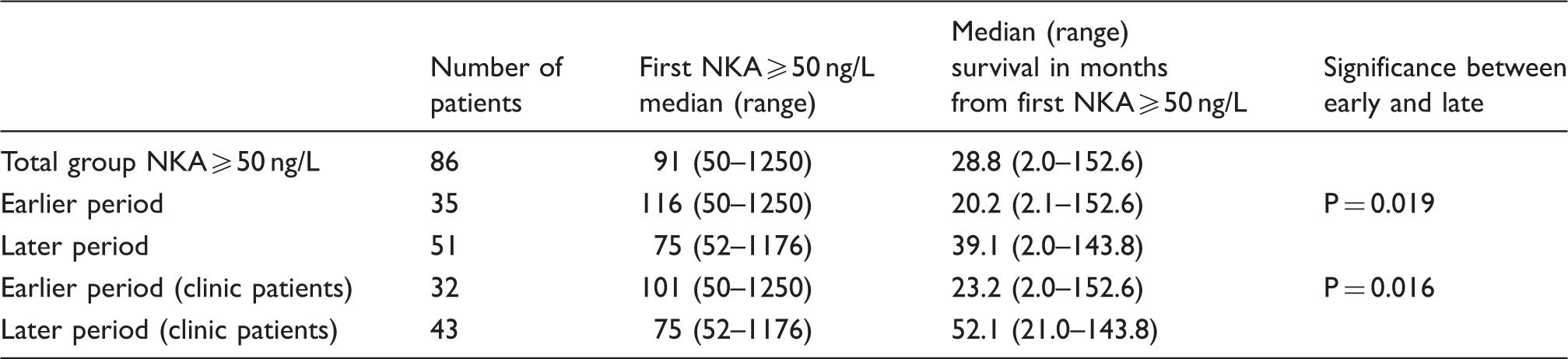
Median survival, from the date when Neurokinin A was first ≥50 ng/L was 11.1 (2.0–117.8) months if Neurokinin A remained ≥50 ng/L and 72.4 (4.8–152.6) months when Neurokinin A was reduced below 50 ng/L and controlled below that concentration for ≥3 months (P < 0.001). Survival was significantly better for patients attending the neuroendocrine tumour specialist clinic than for those not attending (P = 0.009). Comparing patients identified during 1989–2000, and those during 2001–2010, Neurokinin A was successfully reduced in the earlier period in 30.3% patients with median survival 23.2 (2.0–152.6) months and this improved in 58.1% with median survival of 43.3 (2.0–141.1) months in the later period (P = 0.019). Significance was greater between the earlier and later periods when only patients attending the neuroendocrine tumour clinic were compared (P = 0.016).
Conclusions
Circulating Neurokinin A ≥ 50 ng/L is a strong indicator of poor prognosis when Neurokinin A remains above this concentration. Lowering Neurokinin A below 50 ng/L indicates a significant improvement in prognosis (P < 0.001). This prognostic indicator reflects improved treatment and survival in more recent years.
Introduction
Neuroendocrine tumours (NETs) of the ileum and colon are one of the more common groups of NETs with an incidence of approximately 1:100,000.1,2 Prevalence is high (5–6:100,000) as survival is good when compared to gastrointestinal tract adenocarcinoma. The approach to treatment has become much more pro-active in the last decade.3–5 With the exception of tumours of the appendix which are not the focus of this paper, the majority of cases have metastases by the time of diagnosis and surgical cure is usually not possible. However, there is increasing evidence that excision of the primary and de-bulking of metastatic deposits improves survival.6,7–9 Somatostatin analogue therapy is the mainstay of treatment with peptide receptor radionuclide therapy,10–13 hepatic embolization, ablation,14,15 selective internal radiation therapy (SIRTs), interferon alpha therapy 16 and chemotherapy, including, mTOR inhibitors, and tyrosine kinase inhibitors being further options in selected patients. 17 Diagnosis is often delayed and patients may have experienced abdominal symptoms for many years before the diagnosis of a NET is secured.
We have previously published the findings of a retrospective study of NETs of the ileum and colon which collected data to 31 December 2001. 6 Using multivariant analysis, circulating Neurokinin A (NKA) and age were shown to be the only independent indicators of poor prognosis in these patients. With longitudinal sampling, the patient’s most recent NKA was shown to be the most accurate predictor of outcome. Circulating NKA ≥50 ng/L indicated a median survival of less than two years. That study also showed that treatment with somatostatin analogues significantly lowered circulating NKA in 43 of 52 patients but did not correlate with survival.
Since 2001, we have sought to reduce and maintain circulating NKA below 50 ng/L using somatostatin analogues and other treatment interventions including surgery, ablation, microwave, hepatic embolization, SIRTs, radio-nucleotide targeted therapy, interferon alpha and chemotherapy if appropriate. The present paper reports the findings of a study of patients in Northern Ireland, presenting between 1 January 1989 and 31 December 2010 whose circulating NKA concentration rose ≥50 ng/L at any stage between those dates. Patients were followed up to 31 December 2014. The aim of this study was to assess survival in all patients whose NKA rose ≥ 50 ng/L and to compare the survival of patients with NKA ≥50 ng/L in the earlier period (1 January 1989–31 December 2000) and later period (1 January 2001–31 December 2010) when, at the NET clinic, NKA ≥50 ng/L was used as an indicator of poor survival and a rise in NKA towards and above that concentration initiated treatment intervention even in the absence of escalating symptoms. 18
Methods
All NET biochemistry was performed in a single laboratory. This study therefore represents a population-based investigation of all patients in whom circulating NKA rose at any stage ≥50 ng/L, during a 22-year period (1 January 1989 and 31 December 2010). All subjects with NKA ≥50 ng/L at any stage during this time period were considered for inclusion.
The following patients were excluded from analysis. (i) Any patient who did not have the diagnosis of NET confirmed histologically, or by raised CgA and urinary 5HIAA with carcinoid syndrome. (ii) Any patient who survived for less than two months from their first recorded NKA, as these patients were considered to be at a terminal stage at the time of initial presentation.
The following data were recorded for each patient. Date of birth, gender, date of diagnosis, age at diagnosis, date of death when relevant, dates and concentration of NKA analyses, that included the initial NKA, the first NKA to rise ≥50 ng/L and the final NKA which rose ≥50 ng/L but did not return below that concentration thereafter. Survival outcome to 31 December 2014 was also recorded.
NKA measurements were performed using in-house radioimmunoassay. The same NKA assay using the same antisera, radiolabelling procedure, standard preparation and quality control protocol was used across the years of this study. In addition, 22 specimens that had been stored for more than 10 years were assayed again. In each of these repeated specimens, concentrations of NKA varied by less than 8.6%. The sensitivity of the NKA assay was 7.4 ng/L. Inter- and intra-assay variation at the concentration 50 ng/L was 3.6 and 5.2%, respectively. All specimens that record a concentration greater than 100 ng/L were routinely repeated in dilution.19,20 For statistical methods median, range (minimum to maximum) and percentages were used to summarize the variables. The median and 95% confidence interval of the survival analysis were calculated using Kaplan–Meier method where survival was compared in two groups. Cox survival model was used where more than one variable was considered.
Ethical approval was obtained from the Northern Ireland Research Ethics Committee and Clinical Research Governance from Belfast NHS Trust. Informed consent was obtained from each patient in accordance with the ethics approval.
Results
Ninety-seven individual patients were identified with NKA ≥50 ng/L within the study dates. No histological diagnosis was secured in two, whose diagnosis had not been confirmed with CgA, urinary 5HIAA and syndrome. In nine patients, survival was less than two months from the original blood sampling. After exclusion of these 11 subjects, 86 patients were included in the study and their data were subjected to analysis.
Median age at diagnosis was 63.1 (26.5–95.8) years and M/F ratio was 46/40. Median survival for the 86 patients from when they presented first with NKA ≥50 ng/L was 28.8 (2.1–152.6) months. In 47 patients NKA rose to or above 50 ng/L and was not reduced below that threshold again. In these patients, median NKA was 142 (range 52–1250) ng/L and median survival 11.7 (2.1–75.4) months. In 39 patients NKA was reduced back below 50 ng/L (median period 14.5 and range 3–144.0 months). In these patients, median NKA concentration was 63 (range 50–390) ng/L pre-NKA reduction and median survival from the first NKA recorded at ≥50 ng/L was 72.4 (4.8–152.5) months. Survival was significantly improved when NKA was reduced by treatment for three months or longer (p < 0.001) (Figure 1). When the first high NKA ≥50 ng/L was between 50 and 75 ng/L a fall in NKA was achieved in 60.5% of patients and when the first high NKA was above 75 ng/L then a reduction was achieved in 25% of patients. However, whether or not NKA was reduced below 50 ng/L had more impact on survival than the starting concentration of NKA when Cox Survival Method was used. In 72 patients NKA ultimately reached and was not reduced below 50 ng/L thereafter, to death or to the end of follow up. Of these, 66 died within the study period and six remained alive at the end of 2014. Fourteen patients had maintained a circulating NKA concentration controlled <50 ng/L to death or to the end of follow up. Of these patients six died while NKA remained <50 ng/L (in 4/6 the cause of death was not NET related). Eight patients remained alive at the end of follow up with NKA <50 ng/L.
Survival in months of patients with small bowel NETs in whom NKA was reduced below 50 ng/L for more than three months compared to those in whom NKA was not reduced (P < 0.001).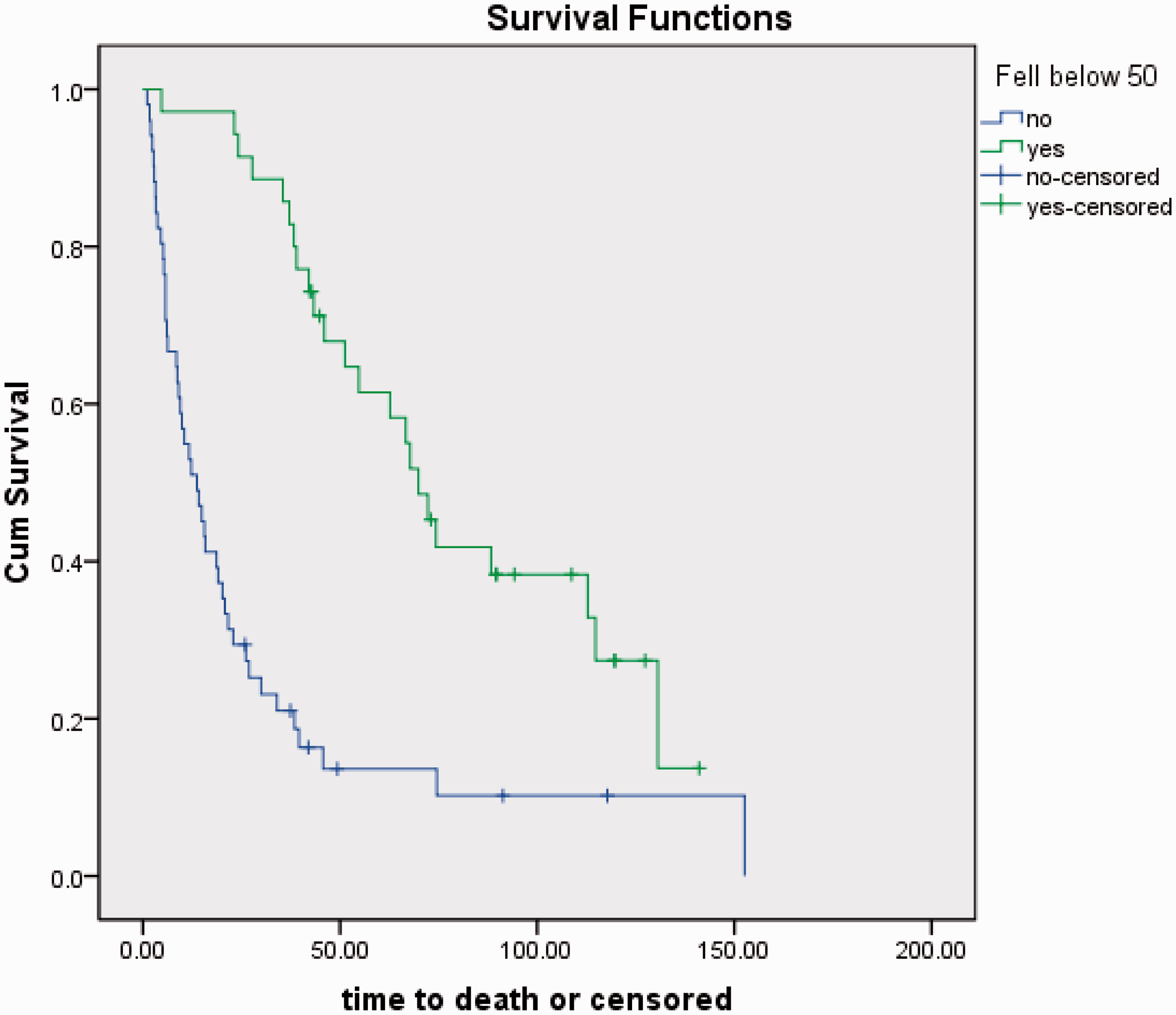
Thirty-five patients in the earlier period (pre December 2000) had a rise in NKA ≥50 ng/L and 51 patients in the later period (1 January 2001–31 December 2010). When compared median NKA was 116 (50–1250) ng/L in the earlier period with median survival 20.2 (2.1–152.6) months. In the later period, median NKA was 75 (52–1176) ng/L with median survival of 39.1 (2.0–141.1) months. This showed a significant difference between median survival from the first NKA concentration recorded ≥50 ng/L during the two time periods (P = 0.019) (Figure 2).
Survival in months of patients with small bowel NETs comparing those diagnosed pre- and post-2001 (P = 0.019).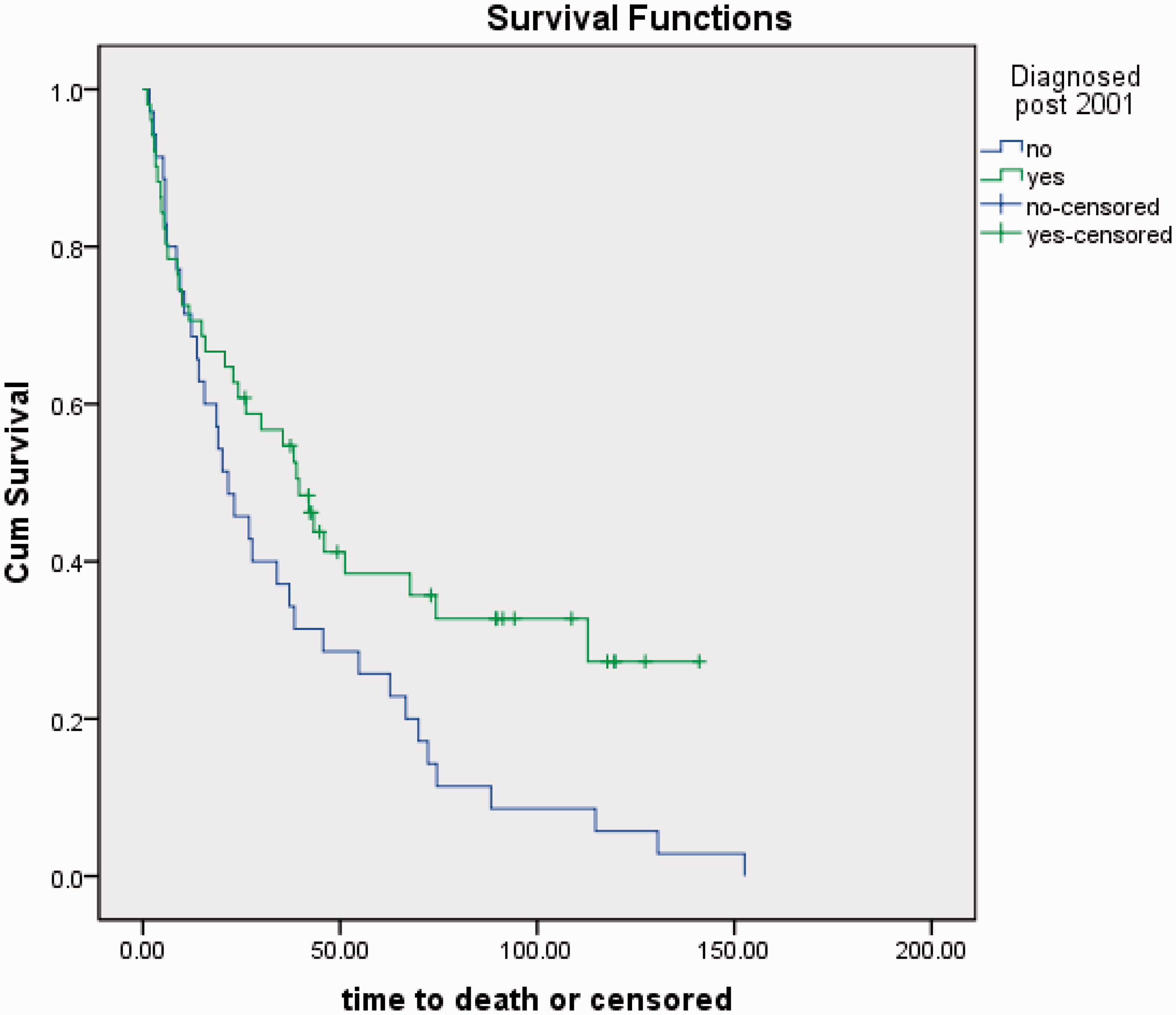
In Belfast there has been a tertiary referral centre for patients with NETs since 1975. Eleven patients were not referred to the NET clinic, three in the earlier period and eight in the later period of this study. Circulating NKA was reduced <50 ng/L in only one of these patients and median survival for these 11 patients was 5.7 (2.9–42.1) months. This survival was significantly worse than for those who attended the NET clinic (P = 0.009).
Comparison between survivals from first NKA recorded ≥50 ng/L in the earlier (1989–2000) and later (2000–2012) periods.
Discussion
This study confirms that a circulating NKA concentration ≥50 ng/L is a useful indicator of poor prognosis in small bowel NETs. This was first reported by Turner et al. in 2006 6 and supported by Diebold and colleagues in 2012 21 who showed in a retrospective study that patients with circulating NKA >50 ng/L had poor survival and survival was significantly improved in those in whom NKA had been reduced below that concentration.
NKA is of use both in patients at the time of diagnosis and also post-therapy. NKA controlled below 50 ng/L reflects significantly improved prognosis. For patients who attended the NET clinic, survival was significantly improved for those in whom NKA rose to ≥50 ng/L after 2001 as compared to those pre-2001. This may be due to earlier diagnosis reflected in the lower NKA concentrations in the more recent patients and also by a more proactive use of treatment as NKA rose.
Accurate prognostic assessment has been difficult in NETs. For small bowel NETs circulating markers are a useful method for assessing tumour bulk. Urinary 5HIAA, circulating CgA and NKA show an elevation in proportion to the number of liver metastases.18,19 CgA is also reported to be a good prognostic indicator for NETs.9,22 However, CgA is a general marker for all NETS metastatic to liver and is also significantly raised in other common conditions including atrophic gastritis and in patients, treated with proton pump inhibitors drugs. In addition, in many patients with NETs, CgA rises to thousands or tens of thousands of units per litre making accurate analysis and small rises or falls in concentration challenging using the assay kit methods employed in most laboratories. In contrast, NKA is a specific biomarker for NETs of the small bowel6,23 and is rarely secreted from tumours outside this anatomical region. The reference range in this laboratory is ≤20 ng/L and concentrations ≥50 ng/L have been associated with poor survival. 6 The previous study has shown that circulating concentration of NKA shows a negative correlation with survival but the most recent NKA measurement is the most significant and renders earlier measurements irrelevant. Circulating NKA is suppressed by treatment with somatostatin analogues24,25 and it has been established also that survival is improved with somatostatin analogue therapy in patients with NETs.26,27 This improved survival is despite little or no change in tumour size. Radiological imaging is used to assess tumour progression in many centres. However, an accurate prognostic circulating marker, such as NKA has added merit, since the test can be performed at more frequent intervals and with less expense.
The current study confirms that when NKA is raised ≥50 ng/L, prognosis is poor. Furthermore, the study shows that when NKA can be returned to below that limit, prognosis improves significantly. These data are consistent over a 20-year period. In the last 10 years, more pro-active approach to treatment and attention to rising NKA resulted in a significant increase in the percentage of patients that move from poor prognosis into better prognosis as identified by measurement of NKA. Those that achieve this reduction in NKA live significantly longer.
In summary, through more proactive and aggressive treatment it has been possible to control circulating NKA concentrations in an increased proportion of patients with small bowel NETs. Achieving a reduction in NKA concentration below 50 ng/L is associated with improved prognosis. Careful monitoring of NKA and appropriate intervention as it rises and particularly when it rises above 50 ng/L is recommended for management of patients with small bowel NETS.
Footnotes
Acknowledgements
We wish to thank Claire Irwin for her technical assistance and Mike Stevenson for his statistical advice.
Declaration of conflicting interests
There is no conflict of interest. JA was in receipt of an unrestricted grant from Ipsen UK.
Funding
This work was funded by the Royal Victoria Hospital and Ipsen UK.
Ethical approval
Ethical approval was obtained from The Office for Research Ethics Committees Northern Ireland (ORECNI) (12/NI/0098).
Guarantor
JESA.
Contributorship
JESA, BTJ and DRM conceived and designed the study. JESA gained ethical approval, enrolled patients and gained written informed consent from patients. JESA and WVS collected and compiled data. JESA wrote the first draft of the paper and BTJ, WVS and DRM edited the manuscript.
