Abstract
Background
We aimed to determine whether the discrepancy between haemoglobin A1c values determined by high-performance liquid chromatography and enzymatic haemoglobin A1c measurements in diabetic patients was clinically relevant.
Methods
We randomly recruited 1421 outpatients undergoing diabetic treatment and follow-up who underwent at least three haemoglobin A1c measurements between April 2014 and March 2015 at our clinic. In 6369 samples, haemoglobin A1c was simultaneously measured by HA-8160 and MetaboLead (enzymatic assay), and the values were compared.
Results
haemoglobin A1c measurements by high-performance liquid chromatography and enzymatic assay were strongly correlated (correlation coefficient: 0.9828, linear approximation curve y = 0.9986x − 0.2507). Mean haemoglobin A1c (6.8 ± 1.0%) measured by high-performance liquid chromatography was significantly higher than that measured by enzymatic assay (6.5 ± 1.0%, P < 0.0001). During the sample processing, four (0.3%) subjects presented consistently lower haemoglobin A1c values (<0.7%) by high-performance liquid chromatography than those from enzymatic assay. Of these, three had Hb Toranomon [β112 (G14) Cys→Trp]. The fourth had Hb Ube-2 [α68 (E17) Asn→Asp]. One other subject presented consistently higher haemoglobin A1c values (>1%) by high-performance liquid chromatography than those from enzymatic assay and was diagnosed with a −77 (T > C) mutation in the δ-globin gene. These unrelated asymptomatic subjects had normal erythrocyte profiles, without anaemia.
Conclusions
We showed that haemoglobin A1c values measured by high-performance liquid chromatography were significantly higher than those measured by enzymatic assay in diabetic subjects. However, when an oversized deviation (>0.7%) between glycaemic control status and haemoglobin A1c is apparent, clinicians should check the methods used to measure haemoglobin A1c and consider the possible presence of a haemoglobin variant.
Introduction
More than 1200 haemoglobin variants, which lead to changes in amino acids, have been reported thus far. 1 About 20% of patients with haemoglobin variants have haemolytic anaemia, polycythaemia or methemoglobinaemia and the remaining 80% are asymptomatic.2,3 Haemoglobin variants are often discovered incidentally based on abnormalities of red blood cells, such as anaemia and polycythaemia. However, in recent years, high-performance liquid chromatography (HPLC) has become widely and routinely used to measure haemoglobin A1c (HbA1c) to evaluate glycaemic control. This recent practice has led to the discovery of several asymptomatic haemoglobin variants.4–7 This has occurred because the HbA1c value measured by HPLC is often abnormally higher or lower than expected compared with the glycemic status in subjects with haemoglobin variants. Such discrepancies raise suspicion of the presence of haemoglobin variants. 8
Although the recent advances in HPLC devices decreased the deviation between the HbA1c values and glycaemic control status in subjects with haemoglobin variants compared with the values obtained using earlier devices, 9 deviations caused by haemoglobin variants are still observed in clinical practice. However, in the Diabetes Control and Complications Trial (DCCT), HPLC was used as the designated comparison method; 10 therefore, HPLC has been routinely and widely used to measure HbA1c.
In fact, even with the modern HPLC devices, discrepancies exist between reported HbA1c values measured by HPLC and those obtained by enzymatic assay, which theoretically should not be influenced by the existence of haemoglobin variants.4,9,11,12 These discrepancies in HbA1c values may be clinically significant. Such discrepancies are particularly significant when subjects with haemoglobin variants have impaired glucose tolerance given the likelihood of missing the diagnosis of diabetes because of falsely low HbA1c values. 8
Therefore, we aimed to compare the HbA1c values measured by a modern and worldwide available HPLC device with that yielded by an enzymatic assay to determine whether the discrepancy between HbA1c values obtained by these two methods in diabetic patients is clinically relevant.
Subjects and methods
Subjects
In total, 1421 subjects, under treatment for diabetes with or without medication and followed up at our outpatient clinic, were randomly recruited if they had undergone at least three HbA1c measurements between April 2014 and March 2015. According to the findings of a previous report, 2 this number of subjects was sufficient to conduct haemoglobin variant analyses. Informed consent was obtained from the all participants in this study.
Procedures
From 1421 patients, who were all of Japanese ethnicity, 6369 samples in total were collected for HbA1c measurement during the 12-month period. In the same samples, HbA1c was measured by enzymatic assay (MetaboLead- HbA1c, Kyowa Medex, Tokyo) and by the Arkray Adams HA-8160 HbA1c analyzer, which is a newly developed HPLC device (Arkray, Kyoto, Japan).
The MetaboLead-HbA1c was calibrated with two prescript standard substances in faithful accordance with the manual. Calibration procedures of the Arkray Adams HA-8160 HbA1c analyzer are fully automated, and thus, they reduce manual operations to the minimum. All reagents, controls and calibrators used for these methods followed the National Glycohemoglobin Standardisation Programme (NGSP) guidelines.
Additionally, glycated albumin (GA) was measured at the time of blood collection. The GA is a ketoamine formed via the non-enzymatic glycation reaction of serum albumin, and it is used for patients likely to have inaccurate results of clinically measured haemoglobin HbA1c. On the presumption that a maximum margin of relative error of HbA1c measurements was within ±10%, the presence of a haemoglobin variant was suspected, if there was a consistent difference of more than 0.5% of the absolute value between HbA1c values measured by HPLC and enzymatic assay. Then, direct base sequencing was performed for the α1, α2, β and δ globin genes13,14 to confirm the presence of the haemoglobin variant. The GA concentration was measured by using albumin-specific protease, ketoamine oxidase and an albumin assay reagent (Lucica GA-L kit, Asahi Kasei Pharma Corporation, Tokyo, Japan) with an automatic spectrophotometer. 15 The conversion formula described by Tahara. 16 was used to calculate the HbA1c value based on the GA concentration. All samples were analysed blindly, and the procedures were in accordance with the Declaration of Helsinki and the local ethics committee recommendations.
Statistics
Data are expressed as mean and standard deviation (SD). Comparisons between the two groups were performed with the Wilcoxon–Kruskal–Wallis test. A P value <0.05 was considered statistically significant. All analyses were performed using StatView software (SAS Institute, Cary, NC, USA). The regression equation was obtained via simple linear regression. Bland–Altman plots were generated using Microsoft Excel.
Results
HbA1c was measured in all 6369 blood samples by both HPLC and enzymatic assay simultaneously, and a strong correlation was found between the methods (correlation coefficient: 0.9828; linear approximation curve: y = 0.9986x − 0.2507). However, the mean HbA1c (6.8 ± 1.0%) value measured by the HPLC method was significantly higher compared with the mean HbA1c (6.5 ± 1.0%) obtained by the enzymatic assay (P < 0.0001) (Figure 1). Moreover, the Bland–Altman plot shows the distinct discrepancy between two methods (Figure 2).
Correlation between HPLC and enzymatic assay for the measurement of HbA1c. When HbA1c was measured by high-performance liquid chromatography (HPLC) and enzymatic assay simultaneously, there was a strong correlation between the methods (correlation coefficient: 0.9828; linear approximation curve: y = 0.9986x − 0.2507). The mean HbA1c value (6.8 ± 1.0%) measured by HPLC was significantly higher compared with the mean HbA1c (6.5 ± 1.0%) measured by enzymatic assay (P < 0.0001). Bland–Altman plot.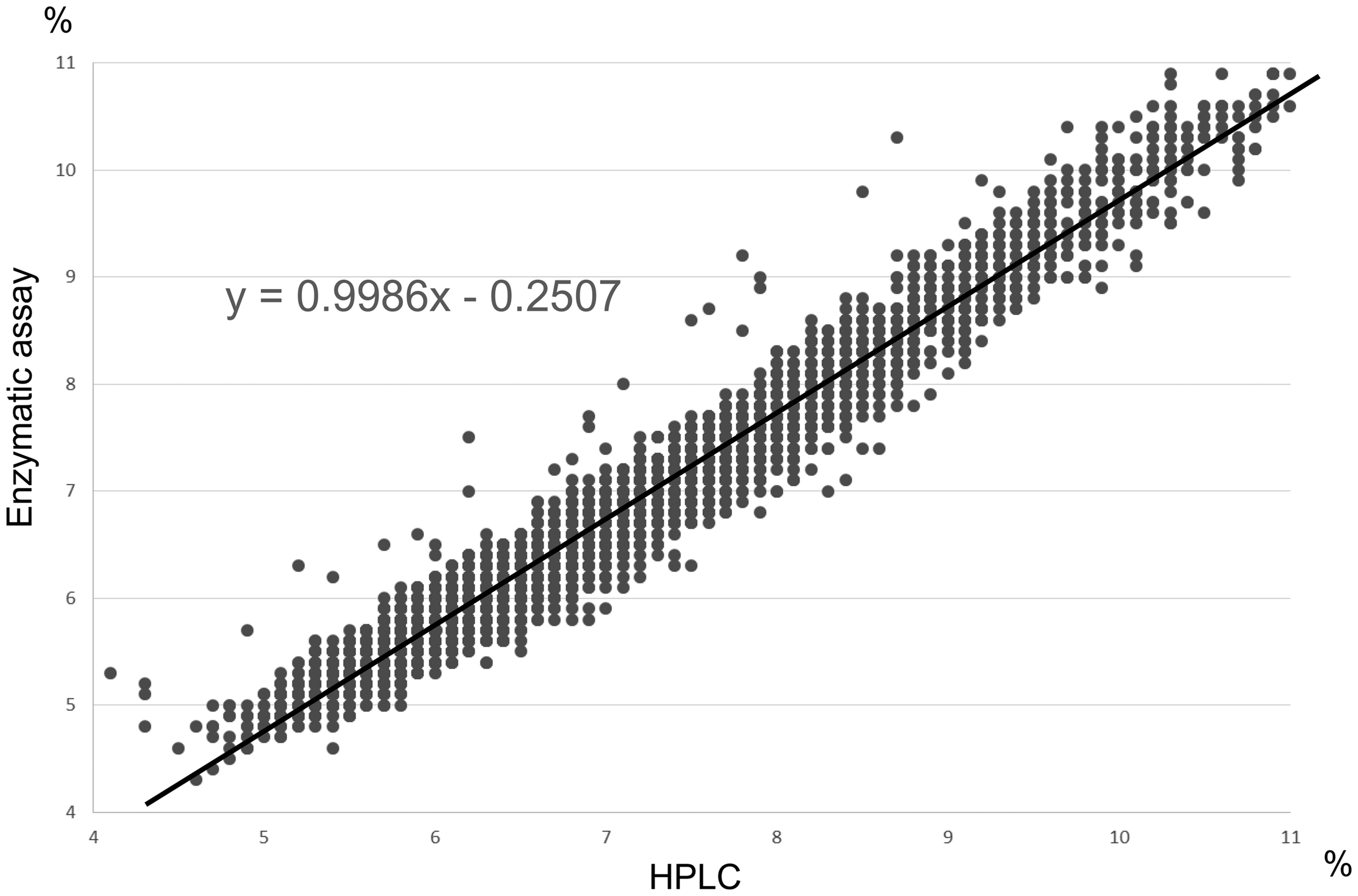
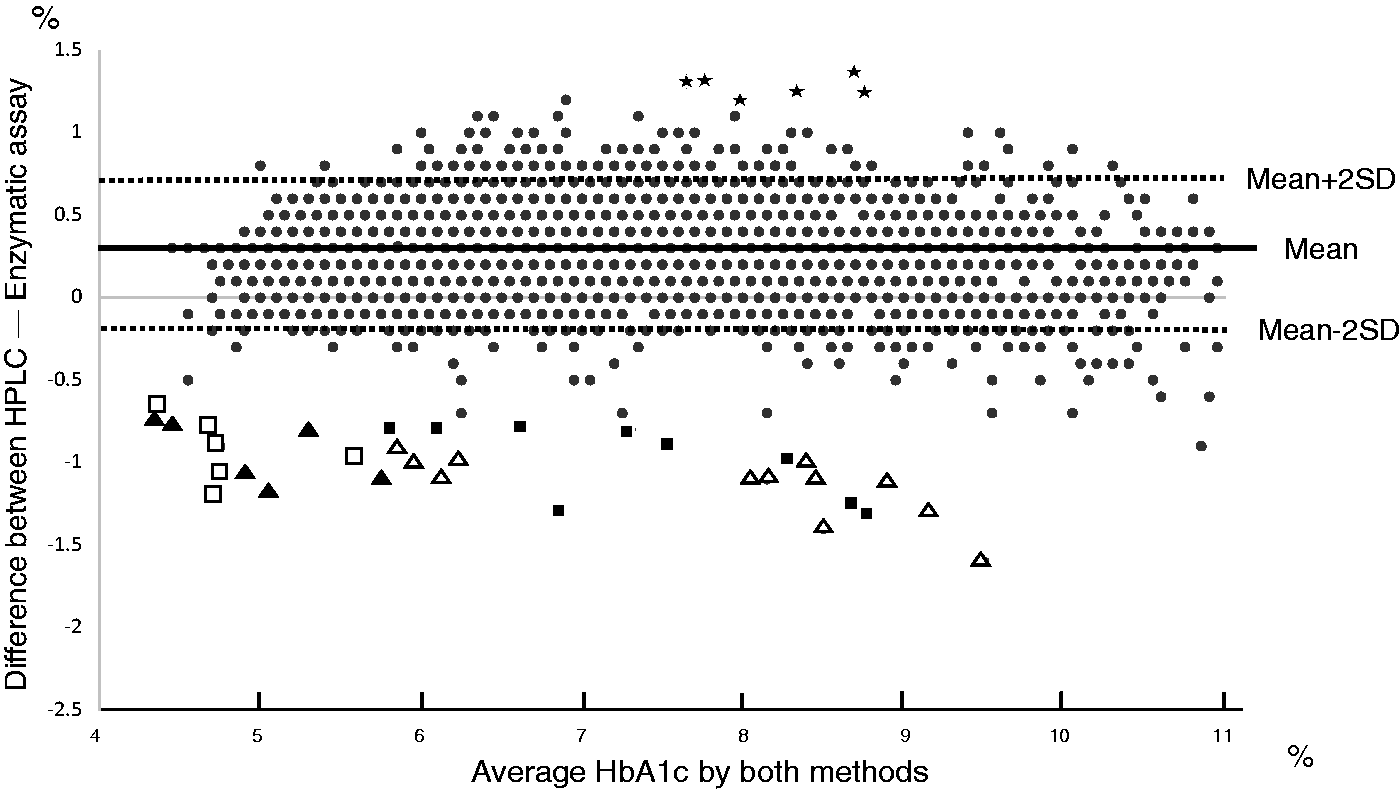
Because we presumed that HbA1c measurements had a maximum margin of relative error of ±10%, we further evaluated the subjects with a constant difference greater than 0.5% between HbA1c values measured by the two methods. If we identified such subjects in the process, we then suspected the existence of a haemoglobin variant in these subjects.
Of the 1421 subjects assessed, none of the subjects presented an inconsistency much greater than 0.5% in the HbA1c values measured by the two methods at any time point throughout the year, except for the five subjects with haemoglobin variants. Although there were several other subjects who occasionally had different values larger than 0.5%, these subjects presented one or several factors that influenced HbA1c, such as iron deficiency anaemia, alcoholism, chronic renal failure, chronic liver dysfunction, ingestion of aspirin or rheumatoid arthritis.
Five subjects with newly detected haemoglobin variant.
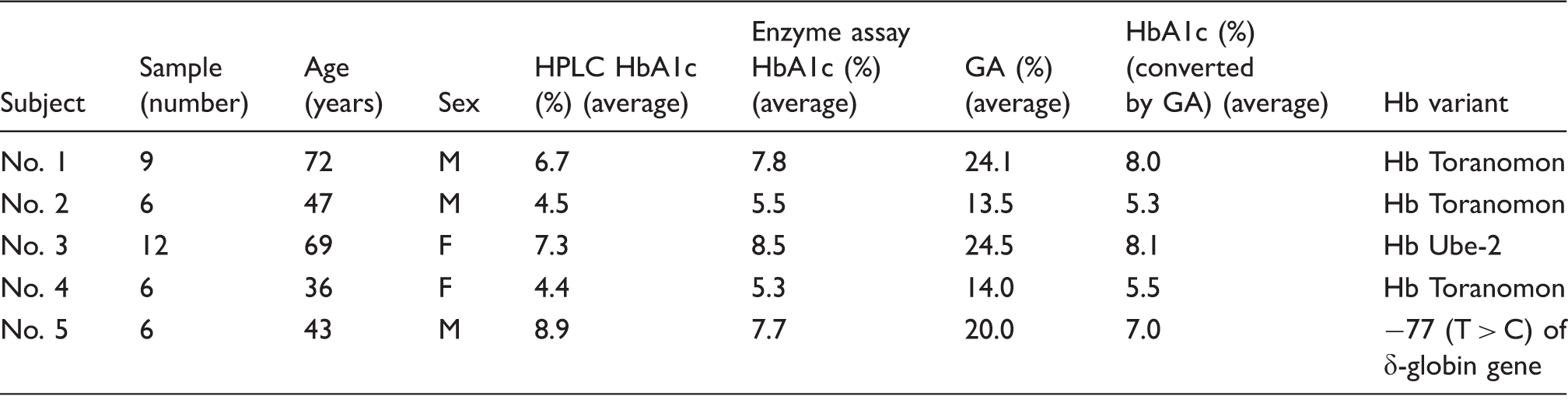
HPLC: high-performance liquid chromatography; GA: glycated albumin; Hb: haemoglobin.
The HbA1c value of another subject was always higher than 1.0% by HPLC than by the enzymatic assay. The haemoglobin analysis of this subject showed that his HbA2 was 1.4%, which was lower than the normal value (between 2 and 3.5%). Therefore, an abnormality in the δ-globin gene was suspected. After genetic testing, a mutation in −77 (T>C) of the δ-globin gene was found (Table 1), and the subject was diagnosed with abnormality in the δ-globin gene.
These five subjects were from unrelated families. Their peripheral blood profiles were normal, and they did not have anaemia and or any clinical characteristics of thalassemia. According to these results, at least 5 of the 1421 subjects had haemoglobin variants. Table 1 shows the differences between HbA1c values measured by HPLC and enzymatic assays in this one-year study.
Discussion
The HbA1c value is widely and routinely used as a biochemical marker for the diagnosis of diabetes and risk assessment of complications when monitoring the treatment of diabetic patients.17,18 According to the guidelines by the American Diabetes Association, diabetes can simply be diagnosed if the HbA1c value is ≥6.5% without the need for further plasma glucose concentration measurements. 19 The HbA1c value can be measured by various methods based on different principles, such as the HPLC method, immunoassays, electrophoresis and enzymatic assays, and these methods are in constant advancement and improvement.11,20 Our results indicated that the HbA1c values measured by the HPLC method strongly correlated with those measured by the enzymatic assay, and the value obtained by HPLC was significantly higher. The reason why these two methods yielded different HbA1c values in the same sample is unknown.
In the present study, 4 out of 1421 subjects presented persistently higher HbA1c values measured by enzymatic assay than by HPLC. Interestingly, the four subjects had a haemoglobin variant. Three of them had Hb Toranomon [β112 (G14) Cys→Trp] and the other one had Hb Ube-2 [α68 (E17) Asn→Asp]. These mutations have been previously reported.21–23 However, we could not utilize a β-thalassemia program (Variant II analyzer, Bio Rad Laboratories, Munich, Germany) to screen for β-thalassemia minor. 7 This system does not exist in Japan because β-thalassemia is very rare in the Japanese population, and most patients not only present anaemia but other clinical symptoms as well.
The relationship between the HbA1c value and daily average blood glucose concentrations is reported to be 126 mg/dL when HbA1c is 6%, 154 mg/dL when HbA1c is 7% and 183 mg/dL when HbA1c is 8%; therefore, a 1% difference in the HbA1c value particularly for diabetic patients is not necessarily a small difference. 24 Therefore, if the HPLC value is higher than the enzymatic value by 1% or more, excessive antidiabetic drugs may be administered for the treatment of the condition. By comparing the HbA1c values by both methods in this study, we identified one patient whose HbA1c value by HPLC was always higher than that measured by enzymatic assay by more than 1%. The haemoglobin analysis of this subject showed a low HbA2 concentration (1.4%). In general, 96 to 98% of haemoglobin in adults is HbA (α2β2), 2% is HbA2 (α2δ2) and less than 1% is HbF (α2γ2). Thus, we suspected the presence of an abnormality in the δ-globin gene in this case, which was later confirmed to be a mutation in −77 (T > C) of the δ-globin gene. This gene mutation is located in the highly conserved core sequence (GATA-1 motif) of the δ-globin gene and impairs expression by affecting GATA-1 binding. 25 A recent study reported that this gene mutation accounts for about 80% of the δ-globin gene mutations and that the incidence is not necessarily low in the general population. 26 The δ-globin gene mutation, without clinical symptoms, is often diagnosed after the finding of a low HbA2 (α2δ2) value, but it is interesting that this gene mutation was found through the difference of HbA1c values obtained by two different measurement methods. Further research is anticipated to clarify the mechanism by which this gene abnormality leads to higher HbA1c values as measured by HPLC.
In the present study, we identified at least 5 (0.4%) out of 1421 subjects who had a haemoglobin variant. In Korea, a neighboring country in East Asia, it was reported that about 2700 individuals had a haemoglobin variant that was detected through an abnormal peak in chromatographic haemoglobin fraction patterns by the HPLC method. 27
Our present results suggest the possibility that haemoglobin variants may be more frequent than previously reported. However, in daily medical practice, medical staffs do not pay much attention to the HbA1c measurement methods and even less to the type of the measuring device used. Therefore, we must bear in mind that the prevalence of haemoglobin variants in this study might be underestimated as it is possible that some haemoglobin variants are missed if there are little or no differences between the HbA1c values measured by these two methods.
In this study, we monitored GA, which reflects intermediate- or short-term glycaemic control, and it is not affected by haemoglobin metabolism. 28 We found that the HbA1c values calculated based on GA values were more similar to those obtained by the enzymatic assay than HPLC (Table 1). Therefore, when there appears to be an inconsistency between the HbA1c value and the glycaemic control, we should check the method used to obtain such values.
The abbreviated report of a WHO Consultation in 2011 29 stated that we should be aware that the most common and important factors worldwide affecting HbA1c concentrations are haemoglobin variants, although the measurement of HbA1c is recommended as an additional test to diagnose diabetes mellitus.
Furthermore, in the NGSP web site (www.ngsp.org), it is mentioned that the purpose of the NGSP is to standardize HbA1c test results based on those of DCCT and United Kingdom Prospective Diabetes Study (UKPDS), which established the direct relationships between HbA1c concentrations and outcome risks in patients with diabetes. Measuring HbA1c by other methods approved by NGSP, such as immunoassay and enzymatic assay, seems to reduce the risk of incorrect medical treatments in consequence of the false HbA1c concentrations. However, the fact remains that the HPLC method has been used according to the method formerly applied in the DCCT and UKPDS. In the future, the measurement method of HbA1c may be replaced by methods other than the HPLC to avoid the inadvertent failure to establish the diagnosis of haemoglobinopathies.
Therefore, the manufacturers should advise the medical staffs to keep in mind the possible presence of haemoglobin variants. When a haemoglobin variant is suspected, other glycaemic parameters besides HbA1c, such as GA, 1,5-anhydroglucitol and fructosamine, should also be examined.
Footnotes
Acknowledgements
We would like to thank Ms. Kumi Otabe for her assistance in this research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was funded by Otabe Clinic.
Ethical approval
The ethics committee of Otabe Clinic approved this study (REC number: 20150101).
Guarantor
SO.
Contributorship
KY and YT researched literature and conceived the study. TO, HN and ES were involved in protocol development, gaining ethical approval, patient recruitment and data analysis. SO wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
