Abstract
Introduction
Serum ferritin is routinely used as a first line test for iron status. Testing subjects with low pre-test probability often results in unexpected abnormal results. Raised ferritin is typically found in subjects with iron overload, liver disease, malignancy or inflammation. We sought to determine whether primary care patients with high ferritin had either porphyria cutanea tarda (PCT) or hereditary haemochromatosis (HH).
Methods
Redundant serum samples were collected from consecutive specimens with high ferritin (>500 µg/L) which had been received from primary care sources. Samples were analysed for serum iron and iron-binding capacity and for porphyrins by fluorescence scanning and HPLC.
Results
There were 240 samples (91 females, 149 males) which represented 2.7% of total over the collection period. Serum iron was 17.3 (18.9) µmol/L (median (IQR)), TIBC 47.3 (14.2) µmol/L and transferrin saturation 35.7 (41.1) %. There were 87/240 (36%) with transferrin saturation >45% (57 males, 30 females). Of the samples 19/236 (8%) were positive for porphyrins by spectrofluorimetry and 14/15 (4 insufficient sample) had total porphyrins >11.2 nmol/L (40(63) median (IQR)) with 3/15 (1.25%) having a typical pattern for PCT.
Discussion
This study demonstrates the feasibility of cascading tests using laboratory protocols and confirms the ability to identify potential cases. However, further studies for HH genotype and urine and stool porphyrin analysis will be necessary to confirm the diagnoses.
Introduction
Serum ferritin is routinely used as a test for iron status. Currently, it is often requested with a full blood count as a first line test when anaemia might be considered as it is faster than awaiting the haemoglobin result before cascading to a second line test. This strategy of performing tests with low pre-test probability often results in unexpected abnormal results. Raised ferritin is typically found in subjects with iron overload, liver disease, malignancy or inflammation whereas low ferritin is more commonly a sign of iron deficiency or low protein states.
A relationship exists between porphyria cutanea tarda (PCT) and hepatic iron overload due to haemochromatosis. 1 PCT is a blistering skin condition related to inherited or acquired deficiency of hepatic uroporphyrinogen decarboxylase and hereditary haemochromatosis (HH) is an inherited condition causing unregulated intestinal iron absorption. In order to explore whether hyperferritinaemia was related to either PCT or HH, O'Reilly et al. 2 identified subjects presenting to the departments of emergency care, gastroenterology, and dermatology in an acute hospital. Over a four-month period they identified 74 subjects with elevated ferritin (>500 µg/L) and determined that 18 (24%) had either PCT or HH.
Since most laboratory requests for ferritin originate from primary care where there is a lower prevalence of inflammatory disease and acute ill health, we sought to determine if cascade testing of samples with elevated ferritin would be a valuable means of diagnosing undetected cases of HH and PCT.
Methods
Serum samples were collected from the Leeds General Infirmary Blood Sciences laboratory. Consecutive samples with high ferritin (>500 µg/L) were recruited from routine specimens after all requested analyses had been performed. Samples were excluded on all hospital in- and out-patients and any samples with clinical details stating haemochromatosis or porphyria. Serum samples were anonymised and stored at −20℃ until analysis. Institutional research governance approval was not granted for the collection of samples for DNA analysis. Ferritin samples were measured on the day of arrival whereas iron and porphyrin studies were performed in batches after sample collection.
Ferritin was measured using Siemens Centaur XP and iron and transferrin saturation were measured using a Siemens Advia 2400 using manufacturer’s reagents (Siemens Healthcare Diagnostics, Camberley, UK). Ferritin between batch precision was 5.6% at 398 µg/L; iron was <0.5% at 17 µmol/L and <1% at 52 µmol/L and transferrin saturation was <0.23% at 22 µmol/L and <0.5% at 80 µmol/L. Iron overload was considered a likely diagnosis if the transferrin saturation was >45% in subjects selected with hyperferritinaemia. 3
Fluorescence emission scanning for serum porphyrins was performed on all samples as an initial screening test. 4 Serum was diluted 10-fold in phosphate buffered saline (pH 7.4) and then scanned spectrofluorimetrically from 550 nm to 670 nm with the excitation monochromator set at 400 nm. Samples with increased porphyrin concentration were identified by the presence of a discernable emission peak at 619 nm and these were further investigated by HPLC analysis. 5 Serum was acidified, deproteinized and then following separation by reverse-phase HPLC the porphyrin fractions were quantified using a modified chromatographic marker standard (Frontier Scientific Europe Ltd., Lancashire, UK). PCT was considered a likely diagnosis if the total serum porphyrins were elevated (reference range <11.2 nmol/L) in conjunction with a characteristic HPLC pattern demonstrating pronounced increase in uroporphyrin I & III and heptacarboxyl III fractions. Samples are exposed to light during processing in the routine laboratory which may degrade the porphyrins. The effect of light exposure was determined in serum samples from known cases of active PCT (n = 4) which had been exposed to 5 h of ambient light during daylight hours (August). The peak height of the serum scan fell by 16% (range 13–28) and the serum porphyrin concentration fell by 18.5% (range 13–22).
Results
A total of 11,230 samples (8960 from primary care) were collected over a period of 57 days. Of these 1524 samples had elevated ferritin >500 µg/L. Following exclusion of samples from hospital in- and out-patients, known haemochromatosis, duplicate samples, haemolysed samples and those with insufficient serum remaining (Figure 1), 240 samples (91 females, 149 males) were available for further analysis. Iron studies showed serum iron 17.3 (18.9) µmol/L (median (IQR)), TIBC 47.3 (14.2) µmol/L and transferrin saturation 35.7 (41.1) %. There were 87/240 (36%) samples with transferrin saturation >45% (57 males, 30 females).
Sample identification flowchart.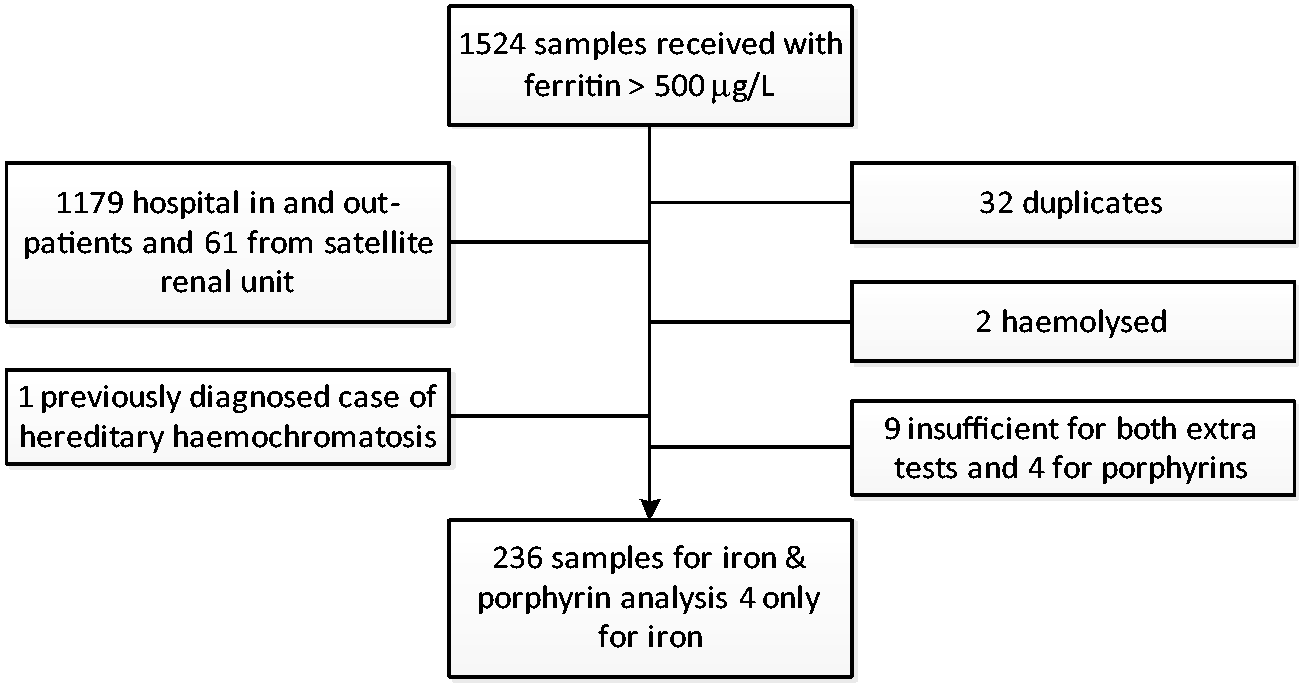
Nineteen of 236 (8%) samples were positive for porphyrins by spectrofluorimetry and 12/15 (four insufficient samples) had total porphyrins >11.2 nmol/L. Three out of 15 samples (1.25%) had a typical pattern of porphyrins to diagnose PCT. The total porphyrin concentrations in cases diagnosed with PCT were 77 nmol/L (43–87) (median (range)) versus 16.5 (12–76) nmol/L for those cases judged not to have PCT.
Discussion
Ferritin requests are routinely made in the investigation of anaemia. In practice, haematinic requests are often made simultaneously with full blood counts rather than waiting for confirmation of anaemia and then cascade testing. This practice therefore means that many patients who are not anaemic have ferritin measurements and may additionally have conditions that cause elevated ferritin concentration. These include liver disease, alcohol excess, iron overload syndromes and any inflammatory condition since ferritin is an acute phase reactant. The aim of our study was to determine how many of these fortuitously identified cases of hyperferritinaemia samples could be given a diagnosis within the laboratory by cascade testing. We chose iron overload and PCT as these could be diagnosed using the same serum sample. We were unable to confirm a diagnosis of HH as our institution would not permit genotyping the sample without consent. The merit of screening for these two conditions is that they are both treatable and morbidity from both is reduced by early diagnosis and treatment.
We used a ferritin concentration of 500 µg/L as a first line step to discriminate cases since this was also used by O’Reilly et al. 2 which allows comparison between the two studies. Our findings are supportive as the identification rate of 36% with iron overload and 1.25% with PCT are not dissimilar from their findings of 17.5% and 6.7% with iron overload and PCT respectively. Further studies will be necessary to confirm these diagnoses but if these are confirmatory, the identification of iron overload in 87/8960 (1%) of routine ferritin samples represents the potential for considerable disease prevention if patients are followed up and treatment is instituted.
The identification of patients with HH and PCT would seem to be a valuable exercise since the morbidities of both diseases can be reduced when treated early. Diagnosis of HH using DNA testing has been shown to be cost-effective. 6 To date no cost-effectiveness studies appear to have been performed for early recognition of PCT but a proportion of patients have underlying hepatitis C and early treatment will prevent disease spread and secondly untreated PCT carries a risk of hepatoma which might be avoided by early detection and treatment of PCT or its underlying disorders.
This study demonstrates the feasibility of cascading tests using laboratory protocols and confirms the ability to identify potential cases. However, further studies for HH genotype and urine and stool porphyrin analysis will be necessary to confirm the diagnoses.
Footnotes
Acknowledgement
This study is part of an MSc thesis to Nottingham Trent University.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
This study was approved by the Leeds Central Research Ethics Committee (Ref 11/YH/0149).
Guarantor
JHB.
Contributorship
The study was designed by JHB and EB, all analyses were performed by EB and TD. All authors have contributed to the final manuscript.
