Abstract
Introduction
Functional iron deficiency parameters predict iron-restricted erythropoiesis more precisely than ferritin. Ferritin and erythropoiesis can be affected by inflammation and hormonal alterations. We hypothesize that the association between low ferritin concentrations and iron-restricted erythropoiesis is not comparable between pregnant and non-pregnant women.
Materials and methods
Pregnant women (n = 926) were included at week 12 of gestation. Ferritin concentrations, %hypochromic erythrocytes (%HYPO), %microcytic erythrocytes (%MICRO), reticulocyte haemoglobin content (MCHr), mean cell volume (MCV) and mean cell haemoglobin (MCH) were analysed. Data were compared with non-pregnant women (n = 1302).
Results
Functional iron deficiency parameters (%HYPO, %MICRO, MCHr) were present in, respectively, 3.9%, 14.3% and 2.3% off all pregnant women. Univariate analysis of low ferritin (<20 μg/L) showed significant differences between non-pregnant versus pregnant women; %HYPO (10.92% vs. 0.92%), increased %MICRO (4.33% vs. 1.00%) and decreased MCHr (24.9 pg vs. 29.5 pg), respectively. In the logistic regression analysis, MCHr, %MICRO and MCV were independently associated with low ferritin concentrations in pregnant women, while %HYPO and %MICRO were independently associated variables in non-pregnant women with low ferritin concentrations.
Discussion
Functional iron deficiency is significantly less frequent in pregnant women compared with iron-deficient non-pregnant women. During pregnancy, iron metabolism might be differentially regulated for optimal fetal growth and development despite low maternal iron stores.
Introduction
Iron deficiency anaemia (IDA) is the most common cause of anaemia during pregnancy and should be distinguished from physiologic anaemia during pregnancy. 1 Physiologic anaemia results from expansion of plasma volume by approximately 50% and increase of red blood cell (RBC) mass by 25%, while IDA is defined as an anaemia which responds to treatment with iron.2–4 Maternal anaemia is associated with low fetal birthweight and preterm delivery, 5 maternal postpartum depression 6 and neonatal impaired cognitive development and growth. 7
Serum ferritin concentration reflects iron stores and is the most used marker to diagnose iron deficiency. Currently, there are a variety of serum ferritin thresholds for the diagnosis of iron deficiency in pregnancy. As the WHO defines iron deficiency anaemia based on a ferritin threshold of <15 μg/L, 1 pregnant women with ferritin concentrations of <30 μg/L are considered to be at risk for IDA in the United Kingdom. 8 In Denmark, it is suggested that women with plasma ferritin <70 μg/L should be treated with iron supplements. 9 The variety in ferritin thresholds led to uncertainty regarding the diagnosis and optimal treatment of iron deficiency in pregnant women. 10 As ferritin concentrations are affected by a variety of factors especially inflammation and infection, it is suggested that corrected serum ferritin concentrations should be used when assessing iron status in these conditions.11,12 The diagnosis of IDA during pregnancy is of vital importance for both mother and child and due to the above limitations of ferritin, functional iron deficiency (FID) parameters might be useful.
Functional iron deficiency is defined as a state in which there is insufficient iron incorporation into erythroid precursor cells despite sufficient iron stores; impaired mobilization of iron from the stores is the cause for the low availability of iron for haemoglobin synthesis. 13 Current haematology analysers enable measuring erythrocyte and reticulocyte indices that can serve as markers of FID.14,15 Decreased mean cell volume (MCV), mean cell haemoglobin (MCH), increased percentages of hypochromic and microcytic erythrocytes reflect long-term iron-restricted erythropoiesis, while the reticulocyte haemoglobin content (MCHr) reflect short-term iron-restricted erythropoiesis. 16 MCHr is probably regarded as the most reliable diagnostic value to investigate short-term iron-restricted erythropoiesis at any given time. 17 MCV might be less reliable as marker of iron deficiency in pregnancy. 18 However, short-term iron-restricted erythropoiesis parameters were superior compared with ferritin, transferrin saturation and MCV to predict iron deficiency in patients with chronic renal failure, patient undergoing bone marrow examination and in pregnancy.19–21
We assessed the value of ferritin and RBC parameters to diagnose iron deficiency in non-pregnant and pregnant women. We demonstrated that the association between FID parameters and low ferritin concentration differs between pregnant and non-pregnant women.
Materials and methods
Study population
Pregnant women (n = 926) were included during the regular first prenatal visit at week 12 of gestation at the local Midwife and Birth Centre between 2015 and 2017. Inclusion was performed by the midwives of the Midwife and Birth Centre. All pregnant women received folate supplements according to the national guideline. Pregnant women did not receive iron supplements. No exclusion criteria were applied. In total, two pregnant women were suspected of thalassaemia based on erythrocyte indices; these patients were not excluded, as genetical testing was not performed. Both patients had normal ferritin concentrations. Non-pregnant women (n = 1302) were included from our local anaemia screening program between 2015 and 2017. Non-pregnant control women were age matched. The non-pregnant women were clinically suspected of chronic anaemia due to either low ferritin, vitamin B12/folic acid deficiency or other causes. The study was conducted in accordance with the Declaration of Helsinki and was approved by the institutional ethical review board (number: TCO14.02).
Laboratory analysis
Blood samples were collected and routinely processed within 4 h. Haematological parameters were determined in blood anticoagulated with K3-EDTA on a haematology analyser (CELL-DYN Sapphire; Abbott Diagnostics, Hoofddorp, The Netherlands). The following haematological parameters were analysed: haemoglobin, erythrocytes, mean cell haemoglobin (MCH), mean cell volume (MCV), percentages of hypochromic erythrocytes (%HYPO), percentages of microcytic erythrocytes (%MICRO) and reticulocyte haemoglobin content (MCHr). Serum ferritin was analysed using a chemiluminescent method on a clinical chemistry analyser (Architect; Abbott Diagnostics, Hoofddorp, The Netherlands). Daily quality control and bimonthly external quality control (SKML, Dutch Organization for Quality Assurance of Medical Laboratories) were performed for all investigated parameters. All parameters were analysed by several counters and routinely checked for harmonization. The following local reference values were applicable: MCV 80–100 fL, MCH 26.0–34.0 pg (1.60–2.10 fmol), %hypo 0.0–3.5%, %micro 0.0–1.4%, MCHr 26.2–31.9 pg (1.62–1.99 fmol) and ferritin 20–150 μg/L. 15
Statistical analysis
Normally distributed continuous variables were described with their means and standard deviations; not-normally distributed ones with the median and their 25th and 75th quartile range. Data were analysed for statistical significance using the Student’s t-test for parametric variables, and the Mann-Whitney U test for non-parametric variables. We made scatter plots to visualize the relation between ferritin and haematopoiesis variables. Logistic regression analysis was performed to assess the relation between low ferritin concentrations (<20 μg/L) and short-term and long-term erythropoiesis factors for pregnant and non-pregnant women. All statistical analyses were performed using SPSS version 23 (SPSS Inc., Chicago, IL, USA) and Graphpad Prism 5.01 software (Graphpad Software, La Jolla, CA). P values of less than 0.05 were considered significant.
Results
Characteristics of the study population
The characteristics of the study population are shown (Table 1). Even though the non-pregnant women were age matched with 12th week pregnant women, there was a small, but statistically significant difference in age. FID parameters were statistically significant different between non-pregnant iron deficient women and pregnant women. The number of patients exceeding reference values are presented in Table 2. Out of 926 pregnant women, 33 women were anaemic (Hb <114.4 g/L [<7.1 mmol/L]). Increased or decreased FID parameters (as defined by results falling outside the reference intervals for erythrocyte parameters) were present in only a small proportion of pregnant women. In total, 16% of the pregnant women had low ferritin concentrations, 2.3% had an MCV <80 fL, 1.2% had an MCH <26.0 pg (<1.60 fmol), 3.9% had increased %HYPO, 14.3% of the women had increased %MICRO and 2.2 of the pregnant women had an MCHr <26.2 pg (<1.62 fmol).
Patient characteristics.

Note: Data presented as mean (±SD) or amedian with 25th and 75th quartiles. Non-pregnant anaemia is defined as haemoglobin concentration <120 g/L (<7.5 mmol/L). Anaemia in pregnancy is defined as haemoglobin concentration <114 g/L (<7.1 mmol/L) at week 12 of gestation.
MCV: mean cell volume; MCH: mean cell haemoglobin; %HYPO: %hypochromic erythrocytes; %MICRO: %microcytic erythrocytes; MCHr: reticulocyte haemoglobin content.
Number of patients exceeding reference values.
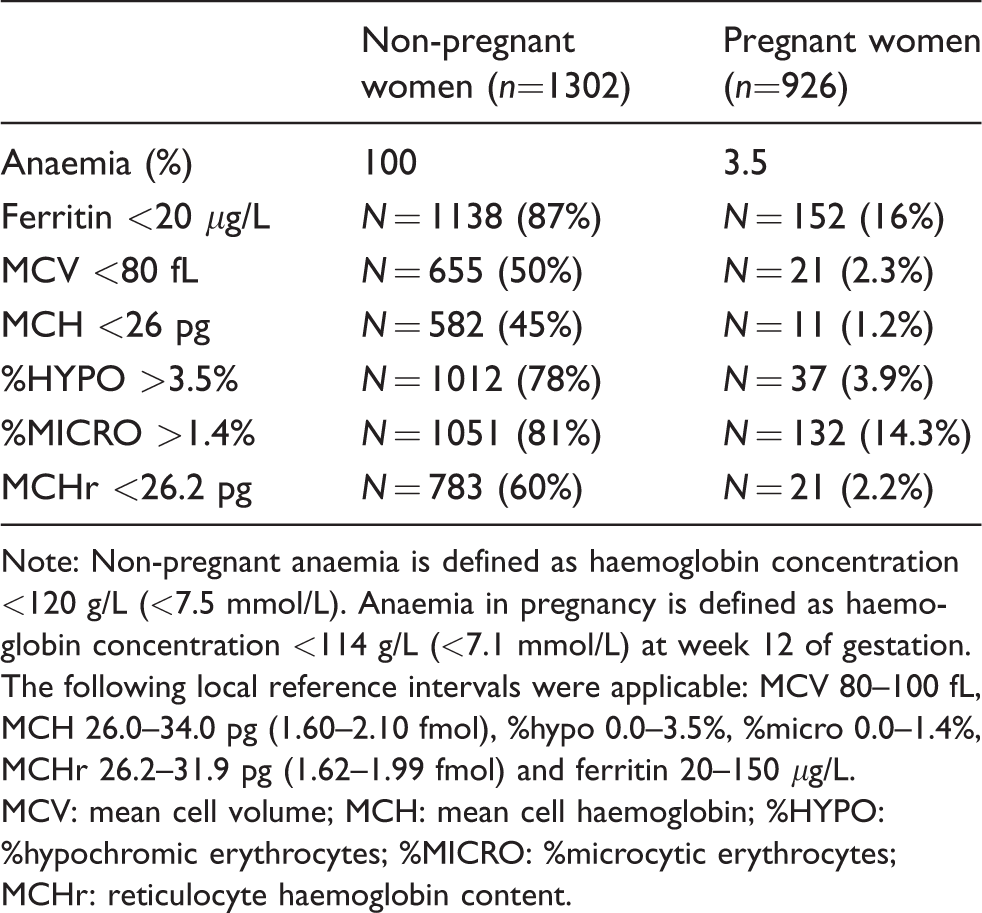
Note: Non-pregnant anaemia is defined as haemoglobin concentration <120 g/L (<7.5 mmol/L). Anaemia in pregnancy is defined as haemoglobin concentration <114 g/L (<7.1 mmol/L) at week 12 of gestation. The following local reference intervals were applicable: MCV 80–100 fL, MCH 26.0–34.0 pg (1.60–2.10 fmol), %hypo 0.0–3.5%, %micro 0.0–1.4%, MCHr 26.2–31.9 pg (1.62–1.99 fmol) and ferritin 20–150 μg/L.
MCV: mean cell volume; MCH: mean cell haemoglobin; %HYPO: %hypochromic erythrocytes; %MICRO: %microcytic erythrocytes; MCHr: reticulocyte haemoglobin content.
RBC parameters in pregnant and non-pregnant women with low ferritin concentrations
Figure 1 reflects the scatter plots percentage hypochromic erythrocytes (%HYPO), percentage microcytic erythrocytes (%MICRO), MCHr, MCV, and MCH in relation to ferritin concentrations in pregnant and non-pregnant women. Among the pregnant women, 152 had low ferritin concentrations and 808 ferritin concentrations within the reference interval. In contrast, the vast majority of non-pregnant women (1138 out of 1302) had low ferritin concentrations and only 164 had ferritin concentrations within the reference interval. The scatter plots patterns of the iron-restricted erythropoiesis markers are clearly different between non-pregnant and pregnant women.
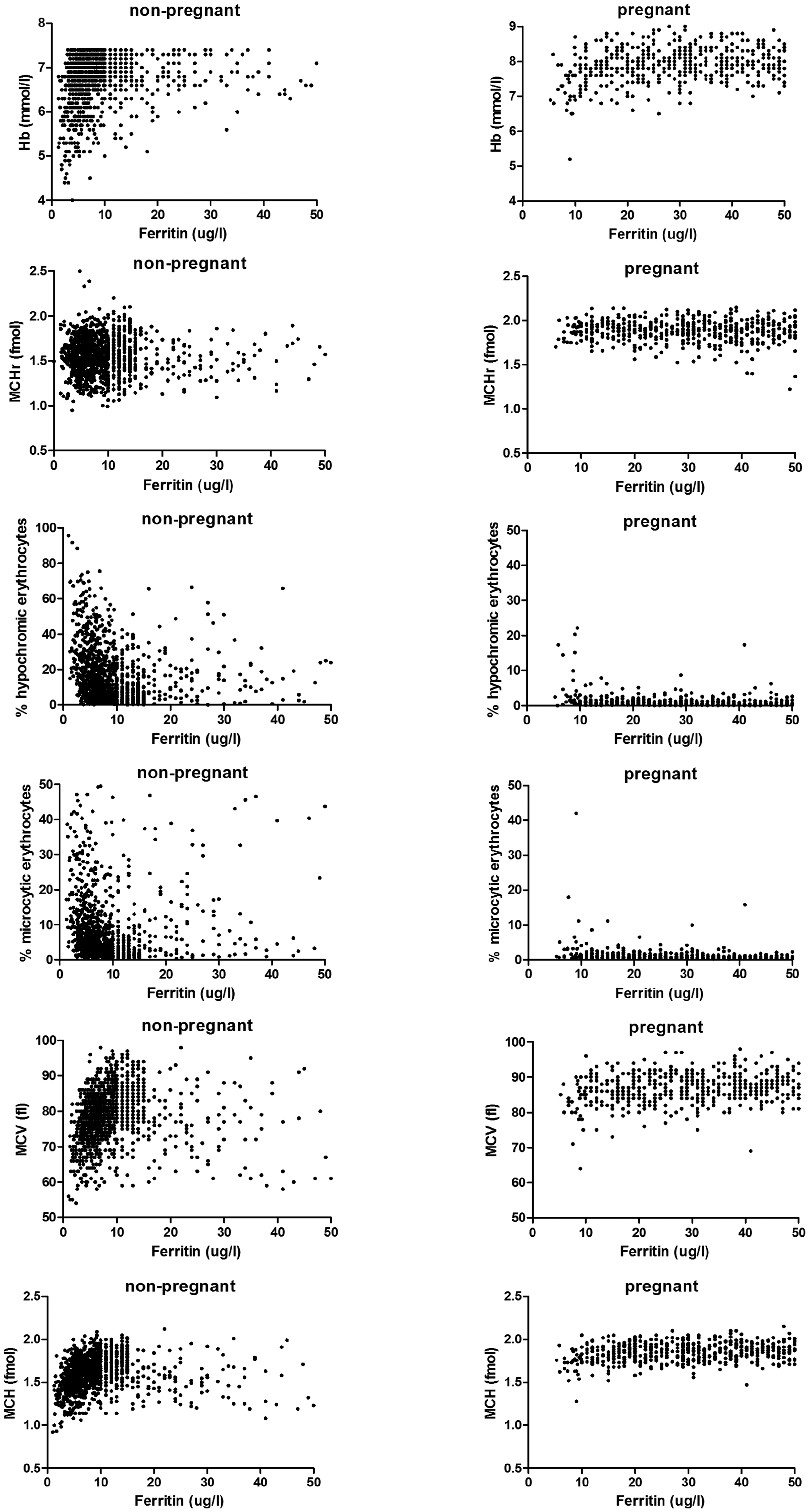
Scatter plot of ferritin with functional iron deficiency parameters. Ferritin and RBC iron parameters were compared between pregnant and non-pregnant women. Low ferritin concentrations in pregnant women are differently associated with functional iron deficiency parameters as compared with the functional iron deficiency parameters in non-pregnant women.
Frequency of FID parameters in pregnant and non-pregnant iron-deficient women
Low ferritin concentrations in pregnant women are not associated with FID, in contrast to iron-deficient non-pregnant women (Figure 1). We found significant differences in FID parameters in pregnant women with low ferritin concentrations (<20 μg/L) compared with non-pregnant women. Significant differences were also found in the pregnant population between low (<20 μg/L) and normal (>20 μg/L) ferritin concentrations, although all values were within the reference intervals (Table 3).
Frequency table of functional iron deficiency parameters.
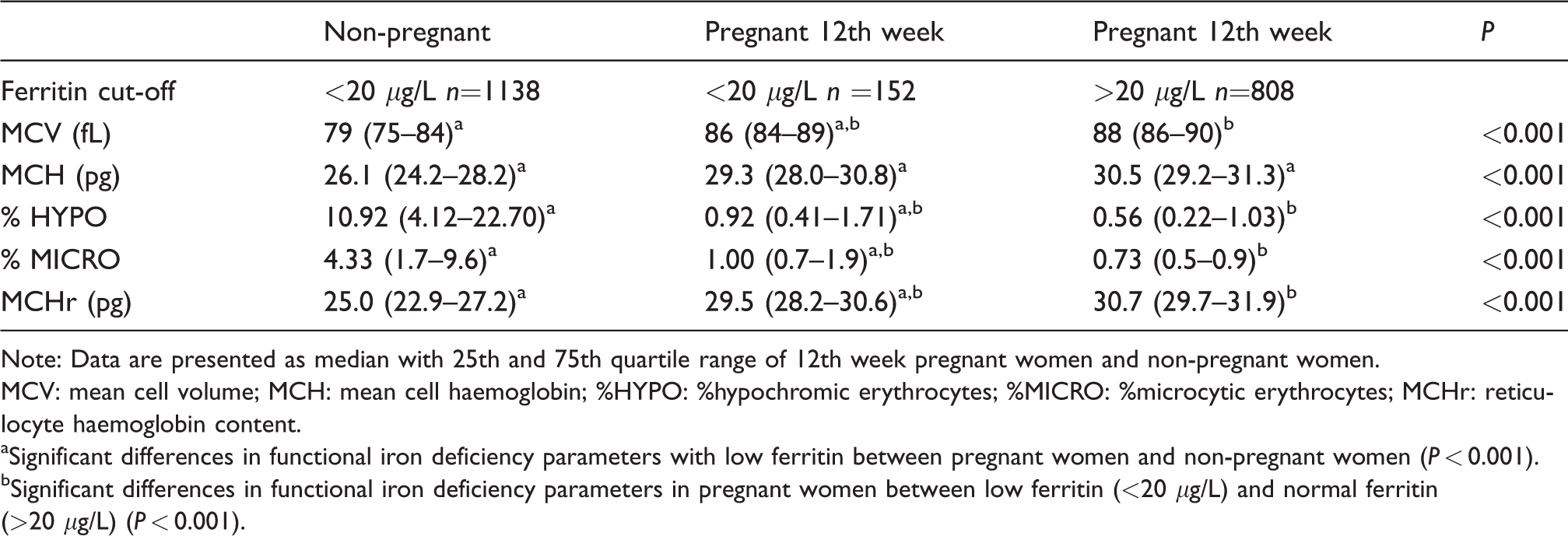
Note: Data are presented as median with 25th and 75th quartile range of 12th week pregnant women and non-pregnant women.
MCV: mean cell volume; MCH: mean cell haemoglobin; %HYPO: %hypochromic erythrocytes; %MICRO: %microcytic erythrocytes; MCHr: reticulocyte haemoglobin content.
aSignificant differences in functional iron deficiency parameters with low ferritin between pregnant women and non-pregnant women (P < 0.001).
bSignificant differences in functional iron deficiency parameters in pregnant women between low ferritin (<20 μg/L) and normal ferritin (>20 μg/L) (P < 0.001).
FID parameters in logistic regression analysis between pregnant women and non-pregnant women
In the non-pregnant iron-deficient anaemia population, the logistic regression analysis revealed hypochromic erythrocytes (P = 0.008) and microcytic erythrocytes (P < 0.001) as independently associated variables with the outcome of low ferritin. In contrast, in the pregnant women population, the logistic regression analyses revealed microcytic erythrocytes (P = 0.02), MCHr (P < 0.001) and MCV (P < 0.001) as independently associated variables with low ferritin (Table 4). In the non-pregnant logistic regression model, MCV, MCH and MCHr were not statistically significant, while in the pregnant logistic regression model, MCH and hypochromic erythrocytes were not statistically significant and thus not independently associated variables with low ferritin.
Logistic regression analysis of iron-deficient non-pregnant women and pregnant women.
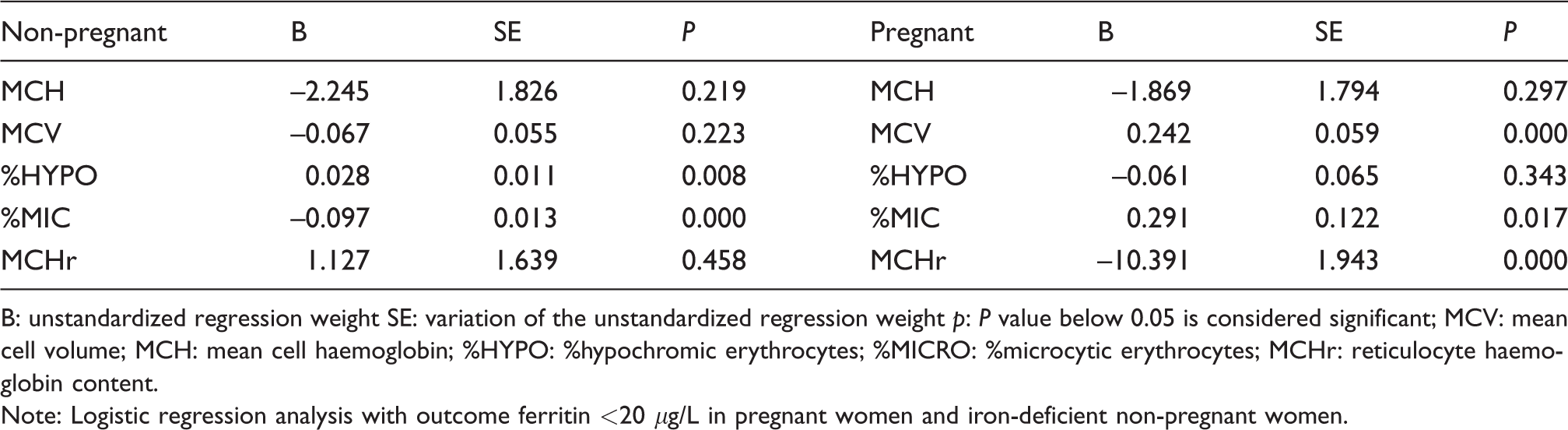
B: unstandardized regression weight SE: variation of the unstandardized regression weight p: P value below 0.05 is considered significant; MCV: mean cell volume; MCH: mean cell haemoglobin; %HYPO: %hypochromic erythrocytes; %MICRO: %microcytic erythrocytes; MCHr: reticulocyte haemoglobin content.
Note: Logistic regression analysis with outcome ferritin <20 μg/L in pregnant women and iron-deficient non-pregnant women.
Discussion
To date, iron deficiency during pregnancy is poorly defined and its mechanism to maintain fetal iron concentrations is not well understood. Iron deficiency during pregnancy is associated with low birthweight, preterm delivery, neonatal impaired cognitive development and growth, and impacts on maternal emotional stability.22–24 Maternal physiology might affect IDA biomarkers. We specifically investigated RBC parameters in non-pregnant women with iron deficiency and RBC parameters during gestation to define whether these parameters differ and to establish which parameter might be of essential need to determine iron deficiency during pregnancy.
In non-pregnant people, the best-established variables for the identification of FID are the percentage of hypochromic erythrocytes and MCHr.20,25 In the case of iron deficiency, the percentage of hypochromic and microcytic erythrocytes will increase and the MCHr will decrease. In the present study, these changes were clearly seen in the non-pregnant women with a low ferritin concentration. However, during pregnancy, low ferritin resulted in a minimal increase of hypochromic and microcytic erythrocytes or a decrease of MCHr; although statistically significant, all these values remained within their reference intervals. 15 We found in the univariate analysis of low ferritin concentrations (Table 3) that hypochromic erythrocytes, microcytic erythrocytes, MCHr, MCV and MCH were statistically significantly different between pregnant and non-pregnant women with a low ferritin. This suggests differentially regulated erythropoiesis and iron homeostasis during pregnancy compared with non-pregnant women.
What are the best markers to diagnose iron deficiency during pregnancy? According to Weyers et al., serum ferritin concentration has excellent sensitivity but poor specificity for detecting depleted iron stores during pregnancy, which is in line with our results. They proposed the reticulocyte haemoglobin equivalent (RET‐He) in combination with the ferritin index (Thomas plot) as the best FID parameters in pregnant women. 26 Ervasti et al. suggested measurement of hypochromic erythrocytes or reticulocyte haemoglobin content (CHr) to determine FID during pregnancy. 19 In our logistic regression analysis, microcytic erythrocytes, and especially MCHr and MCV proved to be independent variables associated with low ferritin in pregnant women. MCHr, RET-He and CHr are all markers of short-term iron-restricted erythropoiesis. These short-term iron markers should be preferred over ferritin concentrations to diagnose iron deficiency anaemia during gestation.
Iron homeostasis during pregnancy is dependent on body distribution, transport, metabolism and regulation. 27 To maintain optimal iron homeostasis during gestation, hepcidin is completely downregulated to achieve maximum iron absorption during the second and third trimesters. 28 van Santen et al. suggested an inhibiting effect of oestrogens on hepcidin synthesis.29,30 Other in vitro experiments suggest that oestrogens and progesterone could have a role in the growth of peripheral burst‐forming unit-erythroid concentrations. 31 Interestingly, it was recently shown that oestrogens increased haematopoietic stem cell self-renewal during pregnancy in mice, resulting in increased splenic erythropoiesis. 32 It is also demonstrated that testosterone has a dose-dependent stimulatory effect on erythropoiesis in men. 33 Whether human chorionic gonadotropin affects erythropoiesis during pregnancy remains to be investigated. Taken together, it is suggested that during pregnancy erythropoiesis is affected by several hormonal factors, and this might explain the differences of the RBC parameters between pregnant and non-pregnant women with low ferritin concentrations.
Weyers et al. and Ervasti et al. found decreased ferritin concentrations despite adequate iron stores and normal RBC indices in late pregnancy.19,26 Our results clearly demonstrated that this phenomenon is already present at week 12 of gestation; However, we do not have data on women in late pregnancy. Well-established specific cut-off levels of MCHr and percentage microcytic erythrocytes should be determined to allow clinical diagnosis of iron deficiency at later time points of gestation and postpartum to improve the health of women and neonates. In addition, hepcidin concentrations during gestation could be determined to assess iron deficiency and its response to oral iron therapy. In our study, we were limited by the lack of a healthy non-pregnant women control group without clinical evidence of anaemia. However, the outcome of this study would not be affected. We were specifically interested to understand whether low ferritin concentrations and RBC parameters would be comparable between pregnant and non-pregnant women.
In conclusion, RBC markers indicating short-term and long-term iron deficiency are seen less frequent in pregnant women despite low ferritin concentrations compared with iron-deficient non-pregnant women. During pregnancy, iron metabolism is probably differentially regulated than in a non-pregnant state. From the point of view of developmental biology, this might secure an optimal fetal growth and development despite low maternal iron stores. Moreover, during pregnancy, ferritin concentrations might not reflect short-term iron-restricted erythropoiesis and RBC parameters are needed to diagnose iron deficiency.
Footnotes
Acknowledgements
The authors would like to thank all patients, midwives and technical staff of the clinical laboratory of Gelre Hospital.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study was conducted in accordance with the Declaration of Helsinki and was approved by the institutional ethical review board of Gelre Ziekenhuizen (number: TCO14.02).
Guarantor
HJA.
Contributorship
MWHJD performed statistical analysis, interpreted the data and wrote the article.
MN: designed the study and critically revised the article.
GvdH: collected all data and patients.
HJvdZL: performed statistical analysis and interpreted the data.
JJMLH: interpreted the data and critically revised the article.
HJA: designed the study, critically revised the article and final approval of the version to be published.
