Abstract
Background
The measurement of cardiac troponin is central for the diagnosis of myocardial infarction (MI). It is recommended that a coefficient of variation of ≤10% is achieved at the diagnostic threshold and significant change between serial measurements reported. Many modern laboratories use multiple analysers linked by automation where samples are randomly assigned to an analyser. It is therefore important to consider the combined effect of all analysers on the analytical performance of troponin measurement.
Method
The performance of a contemporary troponin-I (cTn-I) assay run on three analysers, linked by an automated track, was undertaken across a range of cTn-I concentrations. The data for the three analysers were aggregated to obtain the combined analytical coefficient of variation (CVA) and reference change values (RCVs).
Results
The CVA improved with increasing concentration and calculated RCVs ranged from 67.2% (±13 ng/L) to 32% (±160 ng/L) between cTn-I values 20 ng/L and 500 ng/L. Although there were significant differences in cTn-I measurement between analysers around the diagnostic threshold (
Conclusions
We demonstrate that there are significant differences between the performances of analysers which can impact the biochemical criteria for the diagnosis of MI. We also show that the RCV varies according to baseline cTn-I values and that reporting a single RCV across the analytical range of cTn-I may not be appropriate.
Introduction
An accurate and precise measurement of troponin is crucial for the diagnosis of myocardial infarction (MI). The diagnosis requires serial measurements of troponin between 3 h and 12 h that show a significant rise and/or a fall in troponin values, with at least one measurement above the diagnostic threshold.1,2 It is also recommended that laboratories define significant change between serial troponin measurements. 3
Troponin assays are expected to have an analytical coefficient of variation (CVA) of ≤10% at the diagnostic threshold, although assays with a CVA between 10% and 20% are clinically acceptable. 2 The high-sensitivity troponin (hsTn) assays achieve coefficients of variation of <10% at troponin concentrations below the diagnostic threshold. At these concentrations, contemporary troponin assays have higher imprecision.4,5 Although hsTn assays are available in mainland Europe and the United Kingdom, the use of contemporary assays remains widespread. Currently around 78% of users in the United Kingdom use contemporary troponin assays (based on recent United Kingdom National External Quality Assessment Service (UK NEQAS) data). In addition, the hsTn assays are not available in the USA and await Food and Drug Administration (FDA) approval.
Many laboratories utilize two or more analysers for the measurement of troponin, and samples are randomly distributed by automation to any analyser. It is therefore important to consider the impact of the combined analytical performance when multiple analysers are used upon serial troponin measurement, especially where contemporary troponin assays are utilized. In this study, we investigated the performance of a contemporary troponin-I (cTn-I) assay, operated across three analysers, connected to an automated track and sample manager, in order to established reference change values (RCVs).
Method
Following routine cTn-I analysis, serum samples that had been stored at 4–8℃ for <24 h were anonymously pooled to produce samples with target cTn-I concentrations between 20 ng/L and >500 ng/L. Aliquots of each pool were prepared and stored frozen at −20℃ until analysis. They were defrosted once and front loaded onto one of three ADVIA Centaur XP (XP1, XP2 and XP3) instruments (Siemens, Erlangen, Germany) for analysis with a contemporary4,5 TnI-Ultra assay. The diagnostic threshold (where CVA ≤ 10%) and the limit of detection are reported as 40 ng/L and 6 ng/L, respectively.4,5 One of the above pools, therefore, had a target cTn-I concentration of 40 ng/L, the diagnostic threshold. The analysers were programmed to measure 200 μL aliquots, with different cTn-I concentrations, simultaneously at variable times on different days for 10 days. All runs were performed using the same reagent lot number. The results obtained on running internal quality control and external quality assurance samples, at the time of each sample measurement, were confirmed to be within acceptance. A total of 210 inter-batch data points were obtained. All statistical analyses were performed using R Project for Statistical Computing.
The statistical difference between the mean values at each concentration was computed by analysis of variance (ANOVA). In addition, performance in terms of accuracy of the three analysers was compared across the concentration range by analysis of covariance (ANCOVA). Overall performance of the three analysers was ascertained by considering them as a single unit and aggregating the data at each target troponin concentration. Precisions plots (reciprocal model) were generated for the aggregated CVA values and used to predict inter-assay CVAs for the three analysers combined.
RCV was calculated using the formula RCV = 2.77√(
Results
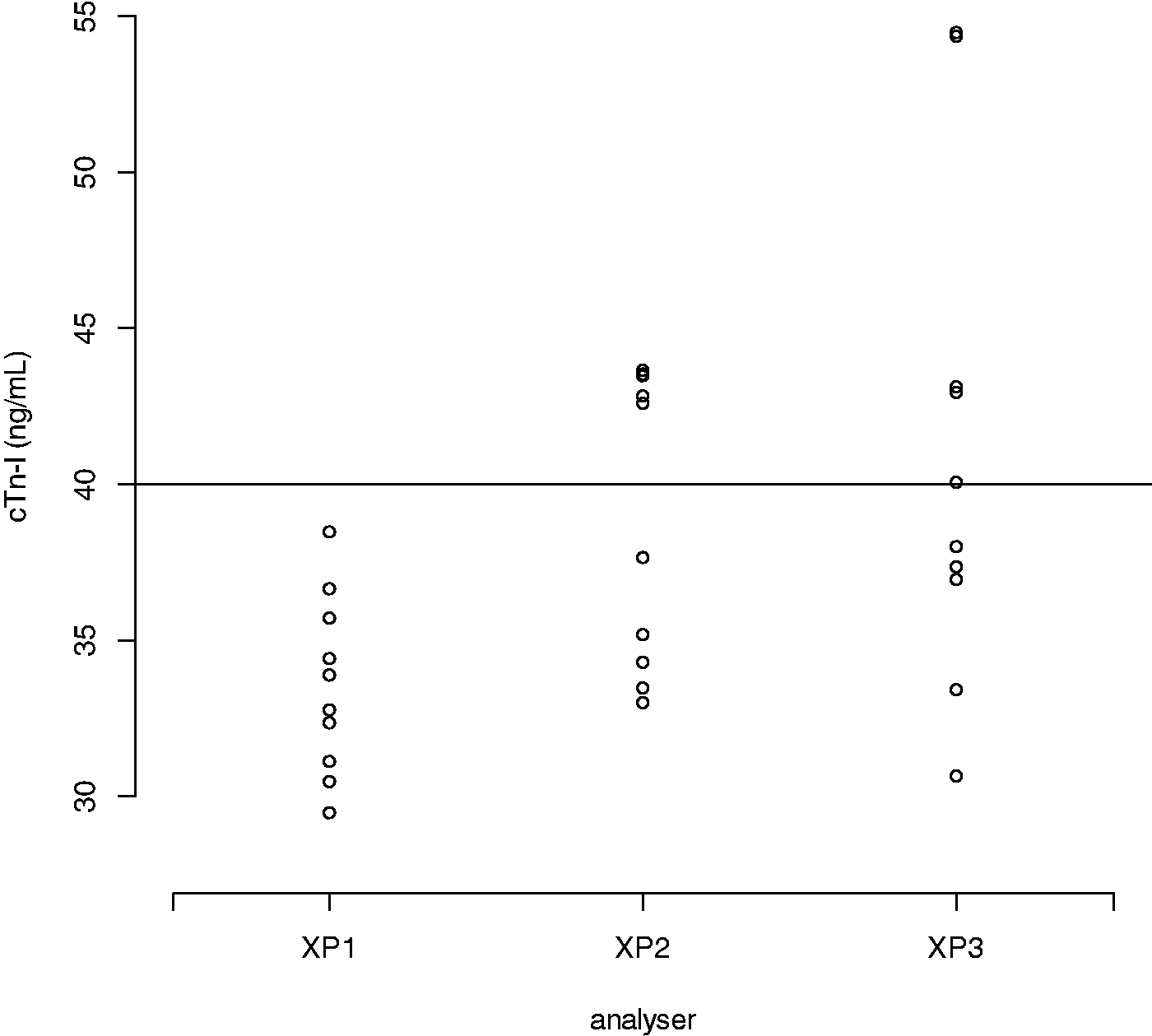
CVAs improved with increasing cTn-I concentration and ranged from 22.1% to 5.8% at target cTn-I concentrations 20–500 ng/L. This gave RCVs of 67.2% (±13 ng/L) to 32% (±160 ng/L) across this concentration range. At the diagnostic threshold of 40 ng/L, the CVA of the aggregated data for all three analysers was 13.6%. RCV was therefore estimated as 46.8% (±19 ng/L) at the diagnostic threshold (Figure 1). Figure 1 also shows that the CVA and RCV are somewhat consistent at cTn-I concentrations over 100 ng/L). With aggregated mean value of 38 ng/L (target: cTn-I 40, range: 29.5–54.5 ng/L), the overall proportion of cTn-I measured at >40 ng/L was 0.33. Broken down by analyser (XP1, XP2, XP3), the proportions were 0.00, 0.50 and 0.50, respectively (H0 proportions are equal, Reciprocal regression model for inter-assay CVA and RCV for aggregated data from all three analysers. CVA: analytical coefficient of variation; RCV: reference change value.. Scatter plot of cTn-I measurement on each analyser at approximate target concentration 40 ng/L. cTn-I: contemporary troponin-I.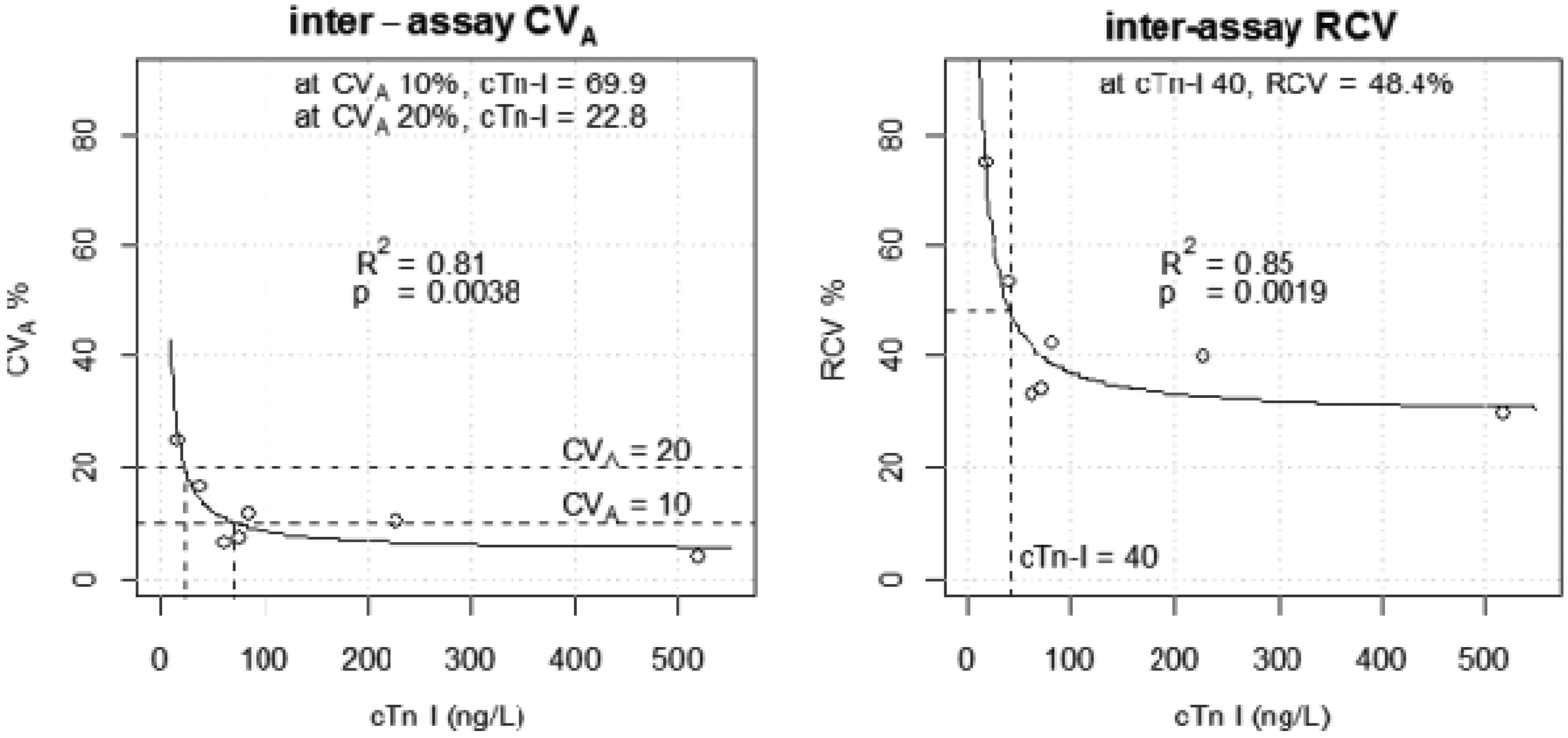
Discussion and conclusions
This study demonstrates that, in terms of bias, differences in the performance of different analysers has clinical implications at the diagnostic threshold. The differences in cTn-I measurement when multiple analysers are used can generate false positive or false negative biochemical criteria for the diagnosis of MI. Despite this, the combined CVA across our three analysers at the diagnostic threshold is 13.6%, within the clinically acceptable range for cTn-I assays.
Laboratories are expected to report significant change values between serial troponin measurements, 3 but there is no clear consensus on best practice for this. There is debate on whether to report a single or multiple RCVs for troponin measurement and whether to report absolute or percentage change. 8
We determined a range of RCVs between 67.2% and 32% for the cTn-I concentrations we investigated for our three combined analysers. We therefore propose that reporting a single RCV as a measure of significant change for cTn-I may not be appropriate at cTn-I concentrations below 100 ng/L in routine practice, in those laboratories where multiple automated analysers are used.
We believe this to be the first report, of the impact, on analytical performance, of running a cTn-I assay simultaneously on multiple analyser platforms. Our findings should be of interest to the many routine service laboratories, which like our own, operate a cTn-I assay on multiple platforms.
Footnotes
Acknowledgements
We would like to thank Dr Paul Smith and Ms Laura Hikin for aiding the review of this report.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
PG.
Contributorship
JP and VL designed and carried out investigation. JP wrote the first draft, subsequent revisions and approved the final draft of the manuscript. PP helped in result analyses, revised drafts and approved the final draft. MN, AB, PC and VL revised drafts and approved the final draft. TD performed statistical analysis, revised drafts and approved the final draft. JT helped review the statistical analysis and approved the final draft. PG helped in the design, data analysis, write up plan, revised drafts and approved the final draft.
