Abstract
Introduction
Laboratories are recommended to determine their own local reference intervals (RIs) to embrace the variations in local populations. We have assessed local RIs for thyroid function tests using two different approaches to selection of reference populations and also searched the literature for studies using the Advia Centaur methods.
Methods
Two independent populations were made of redundant serum samples from primary care in which exclusion criteria were used to reduce the inclusion of patients with thyroid disease. A further population of healthy subjects were recruited. All groups were restricted to 18–65 years and thyroid peroxidase antibodies (TPOabs) positive subjects were excluded. All samples were analysed using Advia Centaur reagents. A literature search was made for RI studies on non-pregnant adults using Advia Centaur.
Results
Redundant data sets consisted of 219 and 222 subjects and a healthy population of 280. Comparison of variance of all three groups showed differences for free T4 (fT4) and total T3 (TT3) (analysis of variance P < 0.0001) but thyroid-stimulating hormone (TSH) was similar across all three groups (P = 0.7656). RI for TSH, fT4 and TT3 all fell within the 95% confidence interval for each other for all three analytes. Published RIs give wide variation although their mean is similar to the prospective data reported here.
Discussion
Our data suggest that a consensus set of RIs for Advia Centaur can be adopted from the prospective studies and literature search in this paper and we would suggest the following RIs: TSH 0.5–4.4 mIU/L; fT4 10–20 pmol/L; and TT3 1.1–2.4 nmol/L.
Keywords
Introduction
Interpretation of laboratory test results is usually aided by the offer of reference intervals or decision limits. Laboratories are recommended to determine their own reference intervals in order to embrace the variations in local populations. In contrast, decision limits are generally introduced by national or international guidelines which do not necessarily consider any local population variation or any method-related variation. Over the years, there has been considerable debate regarding reference intervals (RIs) for thyroid hormones. First, there are clear method-related variations as demonstrated by a number of well-designed studies, and this is compounded by the lack of any accepted international standards.1–3 Second, the choice of statistical tests has been raised as a factor. 4 Finally, and possibly as important, the situation is clouded by disagreement amongst clinicians over the decision value for thyroid-stimulating hormone (TSH) that should be used for defining hypothyroidism.5–8
The choice of reference populations on whom the RI is derived is problematic. Purists would demand that reference individuals should be completely healthy whereas pragmatists choose populations that represent the case mix attending the local health system. The argument for the latter is that a RI describes local populations rather than defining health.
We have tried to determine the effect of reference population choice by studying prospective populations which represent both local case mix and a cleaner healthy population. In addition, we have searched the literature for all studies of thyroid hormone RIs using the same analytical methods that we have used.
Methods
Patient samples
Series 1.1. Redundant serum samples from primary care were collected from the routine biochemistry service. No samples had TFTs requested, and our laboratory computer (Telepath) was interrogated to ensure that no sample came from an individual who had had TFTs requested in the previous 3 years. Additionally, clinical details were examined for any indication of oral medication that might interfere with TFTs or of any obvious systemic disease. The samples were anonymized, stored at 4°C, and analysed within 24 h of receipt. Series 1.2. Repetition of series 1.1 after an interval of 12 months. Series 2. A prospective reference population of healthy individuals was recruited using an International Federation of Clinical Chemistry Committee on Reference Intervals and Decision Limits (IFCC-CRIDL)-approved health status questionnaire for a worldwide reference interval study. Ineligible candidates included, inter alia, pregnant or lactating women, blood donors and individuals on medication or with long-term conditions such as diabetes. Sample collection and preanalytical processes were standardized according to an IFCC-CRIDL protocol.
9
Sample exclusion:
1. All samples with overt hyperthyroidism or TSH > 10.0 mIU/L 2. All samples with thyroid peroxidase antibodies (TPOabs) greater than 100 IU/mL. 3. All samples from subjects aged 66 years or older.
Analyses
Assays for TSH, free T4 (fT4), total T3 (TT3) and TPOabs were performed on all samples on a single ADVIA Centaur XP with Siemens reagents and using the manufacturer’s protocols (Siemens Healthcare Diagnostics, Camberley, UK). TPOabs on series 2 were measured using Beckman reagents (Beckman Coulter Ltd, High Wycombe, UK). The two studies were not performed concurrently and on both occasions, the between-batch precision (% coefficient of variation [CV]) was TSH <6.0%, fT4 <6.4% and TT3 <7.1%. Internal quality control reagents were from BioRad Laboratories Ltd. (Hertfordshire, UK).
For the RI study (series 2), samples were centrifuged at 3000 g for 10 min at room temperature within 6 h of collection and stored at −80°C until analysis. On the day of analysis, samples were thawed at room temperature for 1 h and protected from direct sunlight. Homogenization was achieved by inverting samples 10 times and analysis undertaken within 4 h from the start of thawing. Analysis was undertaken on an ADVIA Centaur XP instrument which was maintained as per the manufacturer standards with no significant deviations observed over the duration of the study (CV TSH 3.85%, fT4 5.25%, TT3 3.85%).
Literature search
Search strategy was <<thyroid[Title] AND Centaur[Title/Abstract] AND reference[title/abstract]>>. All studies related to children and elderly people, and pregnant women were excluded. Further relevant papers cited in these articles were added.
Statistics
Statistical analyses were performed using the Analyse-it v. 4.51 add-in package for Microsoft Excel (Analyse-it Software Ltd, Leeds, UK). RIs were calculated using the Quantile method.
Results
Sample history
Series 1.1 (dataset 1): Of the 393 samples collected, 47 samples were excluded as they were positive for TPOab and a further 127 were excluded as they came from subjects aged 66 or greater (total 219). Series 1.2 (data set 2): Of the 413 samples collected, 40 samples were excluded as they were positive for TPOab, and a further 151 were excluded as they came from subjects aged 66 or greater (total 222). Series 2: Of the 301 samples collected, one was excluded as the TSH was 70 mIU/L, 16 samples were excluded as they were positive for TPOab and a further four were excluded as they came from subjects aged 66 or greater (total 280).
Comparison of the means of all three groups using analysis of variance shows that fT4 and TT3 were different (P < 0.0001) but TSH was similar across all three groups (P = 0.7656). It should be noted that the reference limits all fell within the 95% confidence interval for each other for all three analytes.
Thyroid hormone RI for the three populations studied.
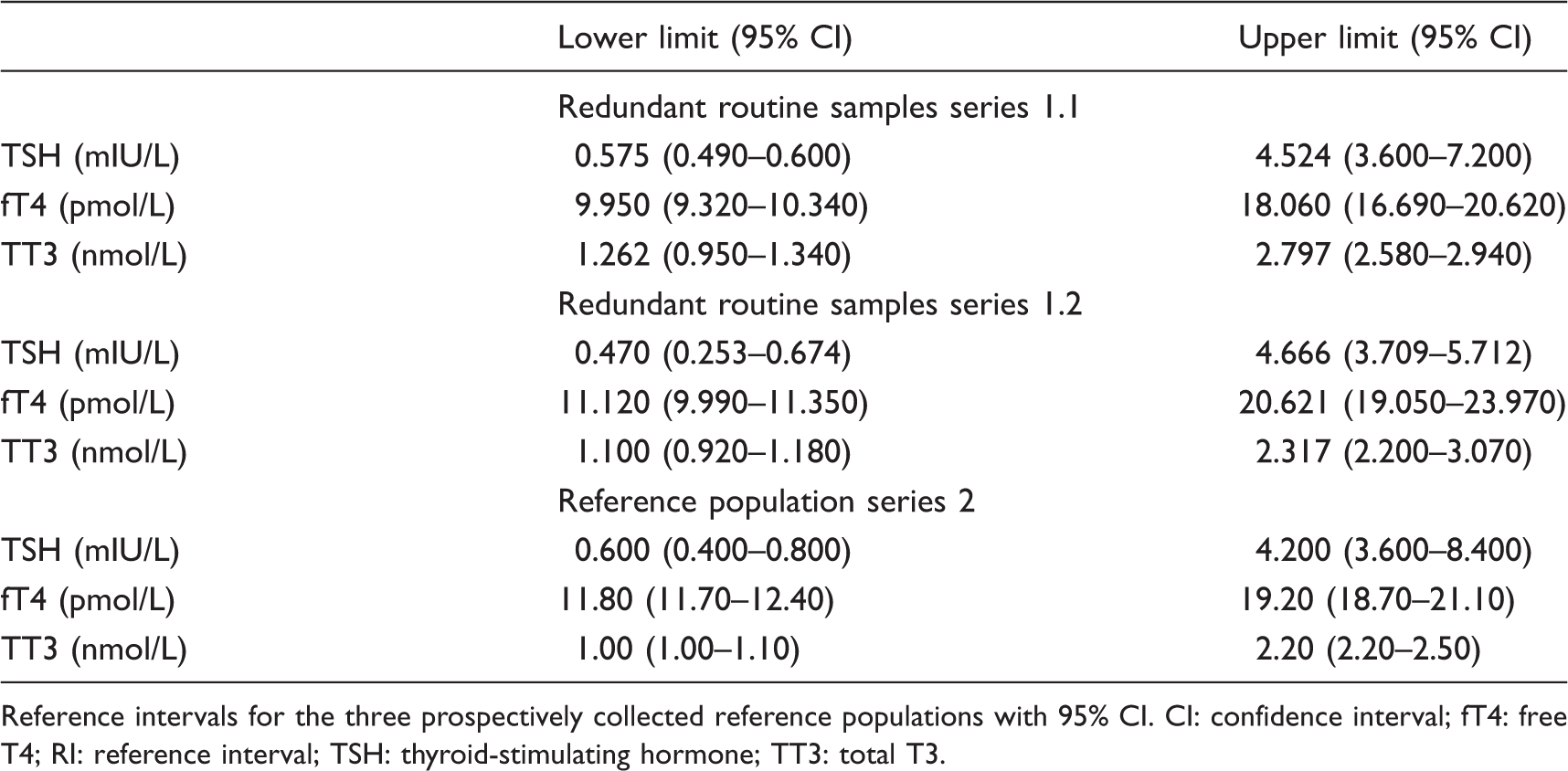
Reference intervals for the three prospectively collected reference populations with 95% CI. CI: confidence interval; fT4: free T4; RI: reference interval; TSH: thyroid-stimulating hormone; TT3: total T3.
TSH RIs for Advia Centaur from literature search.
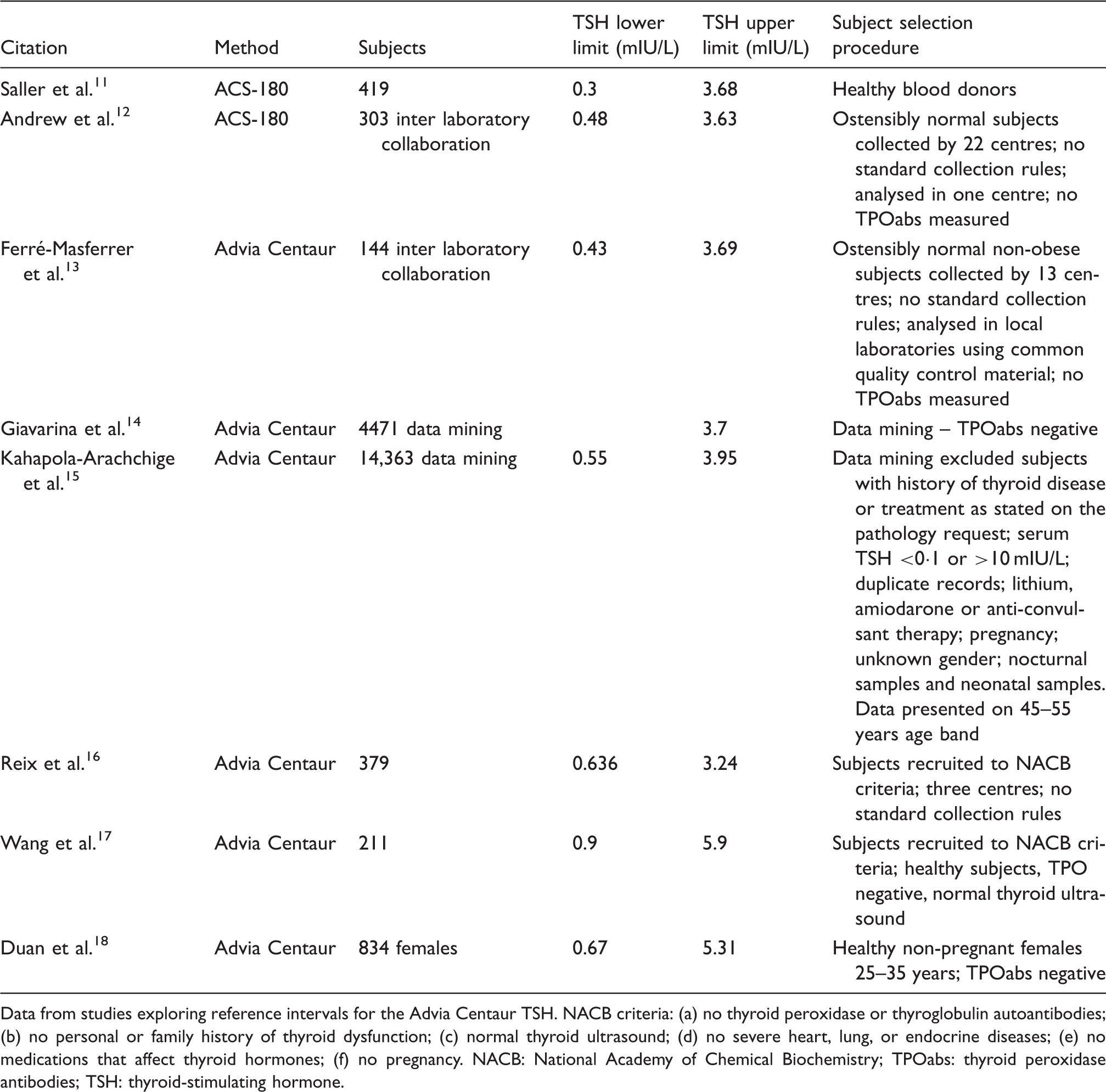
Data from studies exploring reference intervals for the Advia Centaur TSH. NACB criteria: (a) no thyroid peroxidase or thyroglobulin autoantibodies; (b) no personal or family history of thyroid dysfunction; (c) normal thyroid ultrasound; (d) no severe heart, lung, or endocrine diseases; (e) no medications that affect thyroid hormones; (f) no pregnancy. NACB: National Academy of Chemical Biochemistry; TPOabs: thyroid peroxidase antibodies; TSH: thyroid-stimulating hormone.
Discussion
We have explored three different populations which have been collected in a single centre. We found significant differences for each population in the values for fT4 and TT3 although not for TSH. Despite this, there were minimal and non-significant differences in the RIs for all the thyroid hormones. Two of the populations represented patients attending primary care who did not appear to have thyroid disease, and the third was a more carefully selected healthy population.
Our findings highlight the problems with the assessment of RIs. We have used a single analytical method and a single statistical approach. Despite this, slight changes in population selection have shown differences in RIs. These differences are not statistically significant but make specific intervals difficult to construct. Population differences may explain the mismatch with the published literature. This may relate to regional variations or iodine status which has not been formally assessed in any series. Another factor is that the assay for TSH was changed to TSH ultra at the end of 2010. Our personal evaluation of this change suggests that the newer TSH assay showed a reduction in approximately 10% (Passing & Bablock y = 0.90x + 0.03, n = 406, unpublished data). However, this bias is not reflected in the published literature in Table 2, as three papers were published both before and after the new assay introduction.
The importance of assay variation on thyroid hormones has been well studied by the International Federation of Clinical Chemistry Working Group,1–3 but this has not been absorbed by the latest guidelines on hypothyroidism from the American Association of Clinical Endocrinologists and the American Thyroid Association. They make recommendations regarding the upper TSH reference interval (?) but do not even mention analytical differences. 10 Interestingly, their recommendation that laboratories use an upper TSH reference limit of 4.12 mIU/L would be consistent with our data using the Advia Centaur method.
We would propose that Advia Centaur uses the following RIs: TSH 0.5–4.4 mIU/L; fT4 10–20 pmol/L; and TT3 1.1–2.4 nmol/L.
Footnotes
Acknowledgements
Reagents were provided by Siemens Healthcare Diagnostics.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The reference interval study on volunteers was approved by the Bradford Research Ethics Committee (ref 11H/1302/5).
Guarantor
JHB.
Contributorship
JHB, JDS and SRG devised and conducted the studies of redundant serum samples. JHB and AL devised and conducted the prospective study. JHB researched the literature. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
