Abstract
Reverse T3 (3,3′,5′-triiodothyronine or rT3) is the third most abundant iodothyronine circulating in human blood and is produced by the inner ring deiodination of the pro-hormone thyroxine (T4). Unlike the more abundant and active metabolite T3, the measurement of serum rT3 is yet to find a routine clinical application. As rT3 binds weakly to the T3 thyroid nuclear hormone receptors, it is thought to represent an inactive end-product of thyroid hormone metabolism, diverting T4 away from T3 production. The analysis of serum rT3 has, up until recently, been measured by competitive radioimmunoassay, but these methods have been superseded by mass-spectrometric methods which are less susceptible to interference from other more abundant iodothyronines. Serum rT3 concentration is increased as part of the non-thyroidal illness syndrome, and by administration of common medications such as amiodarone which inhibit the metabolism of rT3. Serum rT3 concentration is also affected by genetic conditions that affect the iodothyronine deiodinases, as well as thyroid transporters and transport proteins. Analysis of rT3 can provide a useful diagnostic fingerprint for these conditions. rT3 has been shown to bind extra-nuclear iodothyronine receptors with a potential role in cell proliferation; however, the clinical relevance of these findings awaits further study.
Keywords
Introduction
The analysis of circulating rT3 is controversial; most clinical laboratories do not measure this metabolite, as its relevance to routine diagnosis or therapeutic intervention is questionable. 1 However, some practitioners advocate its measurement to optimize thyroxine replacement in hypothyroid patients, which is controversial, given the current evidence available. The aim of this review is to provide the laboratory clinician with a summary of recent literature concerning the measurement and utility of serum rT3 analysis.
rT3 is the third most abundant iodothyronine circulating in human serum. Like the most active thyroid hormone, T3, it is generated via deiodination of the thyroid prohormone thyroxine (T4) by membrane-bound selenocysteine-containing deiodinase enzymes.2,3 Unlike T3, which is produced by outer-ring de-iodination of T4, rT3 is formed by the removal of iodine from the inner ring (Figure 1). This has an opposite effect on the affinity of each triiodothyronine to bind and activate the principle nuclear thyroid hormone receptors (thyroid hormone receptors alpha and beta, THRα and THRβ), rT3 having an association constant several hundred-fold lower than T3. As such, rT3 is likely to represent an inactive product of thyroid hormone metabolism, at least regarding the THR receptors, and is also unlikely to act as a physiologically relevant competitor for T3 binding to THRβ. While rT3 has little effect at the nuclear THR receptors, it has been shown to interact with the more recently described extra-nuclear thyroid hormone receptors. 4 The function of these potentially fast acting thyroid hormone receptors and the potential for rT3 to act as a physiologically relevant ligand are yet to be established.
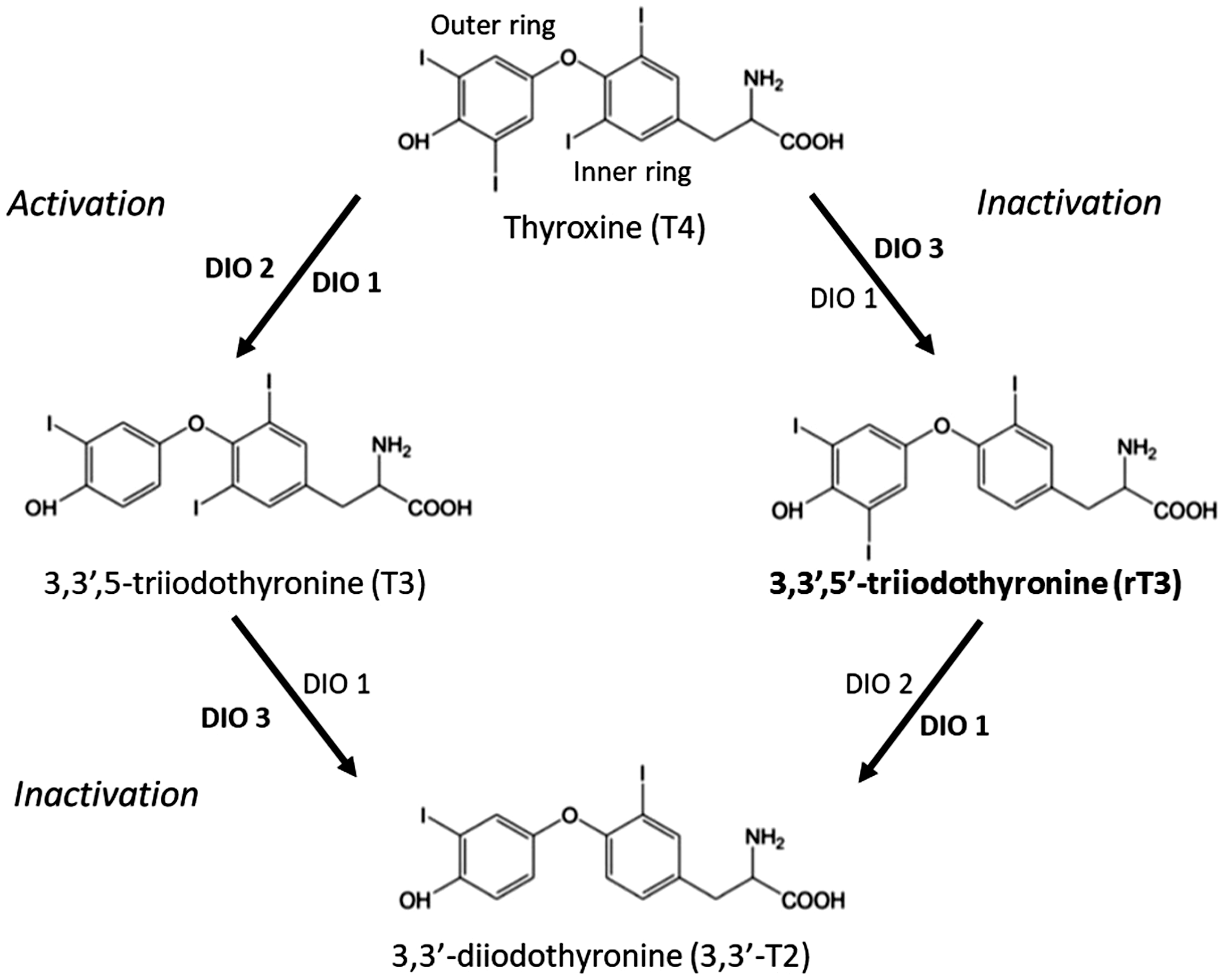
Synthesis and metabolism of Reverse tri-iodothyronine and the roles of the Deiodinases DIO1,2 and 3. Bold fonts indicate proposed major physiological roles. 2
The inactivation of T4 to rT3 is an established physiological response to acute illness (the non-thyroidal illness syndrome or NTIS) or starvation, although a clinical need for rT3 analysis in this situation has yet to be established. 5 Commonly used pharmacological agents such as amiodarone can inhibit 5′ deiodination of T4, resulting in high serum T4 and rT3 concentration at the expense of T3. This can confound the diagnosis of thyroid disease in patients taking amiodarone, which is unfortunate as this is a common side-effect of this drug. The past 10 years have seen a rapid advancement in understanding the process of thyroid hormone transport, metabolism and signal transduction, also the effects of human genetic variation on these processes.6–8 Serum rT3 analysis may have an emerging role in establishing a signature of thyroid hormones which can be used to diagnose defects in these recently discovered pathways.
Analytical
The first quantitative assay for rT3 was a radioimmunoassay (RIA) developed by Chopra in 1974. 9 The consensus from these studies and results from other RIAs subsequently developed was that rT3 is a normal constituent of healthy human plasma, and that the reciprocal conversion of T4 to T3 or to rT3 represents activating and inactivating pathways of the thyroidal prohormone T4. Unfortunately, the original RIAs were susceptible to both binding protein interferences and cross-reactivity with other far more abundant iodothyronines such as T4 itself. 10 Also, the RIA format is manual, time-consuming and requires the use of radioisotopes. Unlike T3, other more convenient immunoassay formats have not been developed for rT3, in part due to the lack of obvious clinical application. Like T4 and T3, rT3 is greater than 99% protein bound in serum, and RIA methods require displacing agents such as 8-anilino-l-naphthalenesulfonic acid to release rT3 prior to assay. 11 Considerable efforts have been expended to measure both the free fraction of T4 (fT4) and T3 (fT3), as this best correlates with biological effects. 12 This has not been pursued for rT3 due to the lack of an obvious biological target, although equilibrium dialysis methods would be appropriate for this task. As the availability and performance of mass spectrometers have improved, rT3, as well as other thyroid hormone metabolites, became an obvious target for highly selective mass-spectrometric methods. T3 and rT3 are isobars so liquid chromatographic separation of these isomers is generally, but not exclusively 13 required prior to mass-spectrometric analysis. Several laboratories have developed mass-spectrometric methods for the analysis of rT3 in human serum,14–16 but the first method to provide a substantial validation including method comparison with RIA was by Sakai et al. 17 Since then, several rT3 methods have been published, which are amenable to the routine clinical chemistry laboratory including methods for use in human tissue samples18,19 and urine. 20 With the discovery of a variety of other biologically active thyroid metabolites (reviewed by Köhrle) 21 and their potential role in peripheral regulation of thyroid hormone activity (reviewed by Little 2018), 22 the selectivity of MS methods has been exploited to for the analysis of large panels of thyroid hormone metabolites including rT3.14,23 While a promising area of research, the clinical utility of these parallel analytical methods is yet to be demonstrated.
Physiology
Synthesis and regulation
rT3 is a short-lived thyroid hormone metabolite whose concentration in serum is determined by competing activities of the membrane-bound selenocysteine-containing deiodinase enzyme DIO1 and DIO2 and that of DIO3.2,3 DIO3 is expressed predominantly in the brain and skin and generates rT3 by the by inner ring deiodination of T4 (Figure 1). rT3 is rapidly cleared by further deiodination to 3,3-T2 by DIO1, expressed predominantly in the liver and kidney, and also possibly DIO2 which is widely expressed in other tissues. 2 The contribution of each deiodinase to rT3 clearance is yet to be fully established and varies with overall thyroid hormone status.2,3 The serum concentration of rT3 is typically 1/10 of that of T3 (approximately 17 ng/mL vs. 140 ng/mL), but in some situations may exceed that of T3. The most common perturbation of serum rT3 concentration occurs in response to carbohydrate deprivation during starvation or severe illness (the non-thyroidal illness syndrome or NTIS). 24 In this situation, rT3 concentration increases due to the inhibition of DIO1 which reduces the conversion of T4 to T3 and the deiodination of rT3 causing the typical picture of low serum T3 and high serum rT3. Depending on the severity of starvation or illness, a secondary hypothalamic response to supress both the TSH circadian rhythm and reduce the concentration of circulating TSH concentration can occur with a consequent reduction in serum T4. While NTIS is thought to be an adaptive response to starvation, NTIS can persist in critically ill patients even in the presence of adequate nutrition, low T3 and raised rT3 being a poor prognostic indicator in this situation. As to whether these patients benefit from thyroid hormone replacement is debateable, reports published to date show no obvious improvements, 25 but most authors would agree that definitive studies have not been done in the group of patients with the more extreme low TSH form of NTIS with low circulating T4 and associated poorer outcomes.
DIO3 is responsible for the production of rT3 with the reciprocal actions of DIO2 and DIO3 now widely accepted as a mechanism to regulate tissue-specific concentrations of thyroid hormone metabolites independently of the hypothalamically regulated circulating thyroid hormone concentration. While the balance of deiodinase activity is critical for both tissue specific and temporal regulation of thyroid hormone action, the serum concentration of rT3 is typically unaffected. Notable exceptions are during pregnancy and in the neonate, as DIO3 is widely expressed in the placenta. This is thought to protect the foetus from maternal T3 during development and causes elevated circulating rT3 in both the pregnant woman and subsequently the neonate. Following parturition, there is a precipitous drop in neonatal serum rT3, 26 as DIO3 expression is supressed and DIO2 is induced which establishes tissue responsiveness to thyroid hormone. DIO3 is an oncofoetal protein, 27 as it is generally expressed during times of cell proliferation but can be re-expressed during oncogenesis. The most extreme example is that of haemangiomas which can express DIO3 to such an extent that circulating as well as peripheral T4 thyroid hormone is inactivated to rT3 leading to overt hypothyroidism.28,29
In similar fashion to T4 and T3, rT3 is extensively protein bound in serum (>99%) to the major serum-binding proteins thyroid binding globulin (TBG), transthyretin (TTE) and albumin (Alb), having higher affinities for all three proteins than T3, but considerably less than T4 (Table 1).
The relative binding affinities of rT3 and T3 compared with T4 for the major thyroid hormone serum-binding proteins.
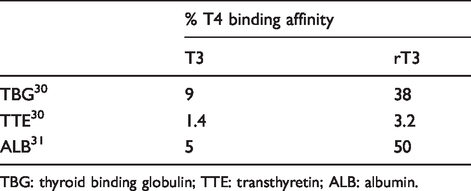
TBG: thyroid binding globulin; TTE: transthyretin; ALB: albumin.
Function
As rT3 has very low affinity for the nuclear hormone receptors THRα and THRβ, it is widely assumed to be an inactive byproduct of thyroid hormone metabolism and to have no function apart from the diversion of T4 from its active metabolite, T3. 21 The best demonstration of the lack of stimulatory activity of rT3 is the frank hypothyroidism that is associated with DIO3 expressing haemangiomas, as described above. DIO3 can be upregulated to such an extent that the concentration of rT3 far exceeds that of T3, with no obvious effect at the nuclear hormone receptor. It is cited in the lay press that rT3 can act as a competitor of T3 action and as such is a potential cause of hypothyroid symptoms despite adequate serum fT3 concentration. Given the binding affinity of rT3 is at least 200-fold weaker than that of T3 for the thyroid hormone nuclear hormone receptors, this is improbable. 21 rT3 has been shown in vivo to act as a competitive inhibitor of DIO1, so there is potential for rT3 to prevent DIO-mediated T4 to T3 conversion; however, this too is also thought unlikely at physiological hormone concentrations. 21
Considerable evidence shows that thyroid hormones can also act at non-nuclear receptors in the plasma membrane cytoplasm, or in mitochondria 4 as well as the more familiar nuclear hormone receptors. Non-nuclear receptors identified include non-transcriptionally active truncated forms of THRα such as p30 TRα14 and the plasma membrane integrin αvβ3. These receptors are thought to be mediators of the previously described rapid (and therefore non-transcriptional) effects of thyroid hormone, the best described being a rapid increase in mitochondrial respiration which is thought to be mediated by the truncated TRα receptor. Unlike the nuclear hormone receptors, these novel receptors are not specific for T3 and can be activated by T4 (previously assumed to be a prohormone) and rT3 (presumed to be an inactive metabolite) directly, and also by a plethora of other thyroid hormone metabolites and derivates that do not bind THRα or β. 4 rT3 and T4, but not T3 also have a striking effect on in vitro models of the neuronal actin cytoskeleton, 4 causing rapid repolymerization of actin derived from hypothyroid animal models which also likely act via the truncated THRα receptor. αvβ3 has been identified as a thyroid hormone receptor with regulatory effects on angiogenesis 32 ; this may have far reaching consequences on the impact of thyroid hormones on tumorigenesis. Whether these newly identified receptors will lead the identification physiological roles for rT3, or whether these are ‘in vitro’ effects of rT3 are due to cross-talk at non-specific receptors for other more abundant thyroid hormones is a topic for further study. However, rT3 has been shown to have a direct effect on the proliferation of human glioblastoma and breast cancer cell lines, 33 which may be of clinical consequence, particularly considering the NTIS where systemic rT3 concentrations can be dramatically elevated. 34
Xenobiotics and diet
Many drugs currently prescribed have an effect on thyroid function, 35 and some of these have a specific effect on deiodinase function so can affect the serum concentration of rT3 (Table 2).
The effects of common medications on serum rT3 concentration.
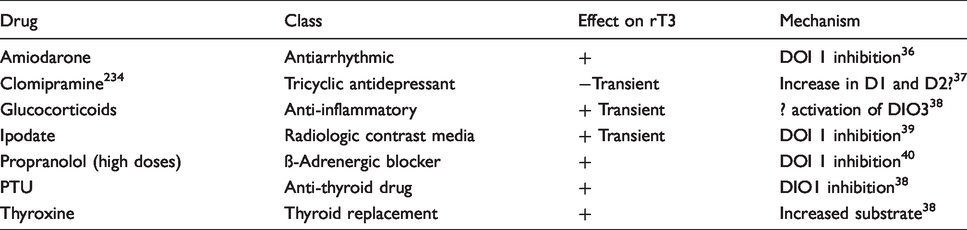
The best described effect is that of amiodarone. Amiodarone has complex effects on thyroid function in part due to its high iodine content, but also as an inhibitor of outer ring deiodination by DIOs 1 and 2. 40 Inhibition of DIO1 causes an increase in serum rT3, as rT3 clearance is diminished. Inhibition of DIO2 causes an increase in TSH secretion as T4–T3 conversion within the pituitary is reduced. Both mechanisms contribute to the raised serum T4 and reduced T3 seen in euthyroid patients taking amiodarone. The effect of dexamethasone is different, as production of rT3 rather than clearance is affected. This is likely to be due to effects on DIO3. 37
The isoflavones present in soy have been shown to cause a transient rise in rT3 with a concomitant increase in TSH. 41 However, concerns regarding a negative impact of soy consumption on thyroid function have not been substantiated by meta-analysis. 42
Reference intervals
Reference intervals for serum rT3 are method dependent, given the potential for cross-reactivity of other thyroid hormone metabolites with immunoassays for rT3. 17 Serum rT3 concentration can also be significantly affected by carbohydrate deficiency or concurrent illness. In general, rT3 is elevated (median ∼1 nmol/L) at birth due to the high expression of DIO3 in the placenta and then drops rapidly postparturition with a less dramatic decline to adult levels (median ∼0.25 nmol/L) throughout infancy. 43 Concentrations then increase with age, doubling in centenarians compared with young adults 44 ; this is thought to reflect a genuine age-related reduction in 5′ deiodination rather than confounding effects of carbohydrate intake or NTIS.
Clinical laboratory application for rT3 measurement
Unlike TSH, T4 and T3 serum, rT3 analysis has not found a role in the laboratory investigations for thyroid disease, and for this reason, high throughput methods for serum rT3 have not been developed. A recent review from the US showed that rT3 is requested rarely by most practitioners (80% never requesting), but favoured by a select number, 0.1% of requestors accounting for 29.5% of the rT3 orders rT3. 1 Frequent orders came from requestors defined as ‘functional medicine’ practitioners; this is a controversial patient-centred rather than disease-centred approach to medical therapy. 45 While it is now well described that 10–15% of patients on thyroid hormone replacement report hypothyroid symptoms, despite TSH being within the reference interval, evidence that the analysis of serum rT3 concentration can aid further modifications to treatment such as T3 therapy is currently lacking 1 and the use of rT3 in this manner cannot be recommended. This conclusion is largely based on the evidence previously discussed that rT3 has very low affinity for the nuclear TSH receptors and is unlikely to compete with T3 for these receptors or for the deiodinase enzymes at physiological concentrations.
While the utility of serum rT3 analysis to direct thyroid hormone replacement is far from established, the measurement of circulating rT3 does reflect central 5′ deiodinase activity and may be relevant in several clinical situations as detailed below. In particular, the analysis of rT3 has found a more positive role in the identification of rare genetic syndromes associated with the thyroid axis. As these defects can present with often subtle alterations in circulation thyroid hormones, more accurate methods for rT3 as well as other thyroid hormone metabolites enables the identification of thyroid hormone signatures that can be used to identify these syndromes.
All-cause mortality
Serum rT3 has been shown to positively correlate with all-cause mortality (hazard ratio of = 1.23; 95% CI, 1.12–1.34; P < 0.001), 46 which is likely to be solely due to the adverse effects of NTIS, of which rT3 is a sensitive marker. This is likely to be of epidemiological rather than clinical significance.
NTIS
The use of serum rT3 analysis in the management of NTIS remains controversial. 5 The potential benefits of rT3 analysis is to detect a cohort of patients that may benefit thyroid hormone replacement either because the diagnosis of hypothyroidism may be confounded by NTIS or that NTIS may directly benefit from thyroid hormone administration as an adjunctive therapy. In either situation, TSH may be a better marker for disease status, as increased TSH may better reflect coexisting hypothyroidism and suppressed TSH and fT4 the component of central hypothyroidism associated with NTI. A notable but rare exception is the diagnosis of coexisting NTI in patients with suspected secondary hypothyroidism. Serum rT3 is demonstrably low in patients with congenital central hypothyroidism, 47 so raised rT3 in the context would identify NTI as a confounding factor. In both children 48 and adults, 49 NTI associated with low TSH and fT4 is associated with a poorer prognosis than patients with altered T3:rT3 ratio, suggesting that the earlier change in deiodinase status may represent a beneficial metabolic and inflammatory response to NTI, with the latter central hypothyroidism a more sinister cytokine-mediated pathological process. 25 Thyroid hormone treatment is unlikely to be beneficial or may even be detrimental in the early phase of NTI in the absence of elevated TSH. The potential benefit of thyroid hormone replacement in the later central hypothyroid phase remains to be established. 50
Insulin resistance
The ratio of T3 to rT3 has been shown to positively associate with markers of insulin resistance and with gender, males having a lower T3:rT3 ratio. 51 These findings cannot be explained by changes in the pituitary thyroid axis, as demonstrated by a lack of response of serum TSH concentration. Insulin is known to increase DIO2 expression in human skeletal muscle but as to whether this relation is a cause, or an effect of insulin resistance awaits further study. A common polymorphism in DIO2 (Thr92Ala) has also been associated with insulin resistance, 52 which may be a tissue-specific effect, as evidence for any effect on circulating thyroid hormones are conflicting. 3
Drugs affecting deiodinase function
As previously discussed, several drugs can affect deiodinase function and cause alterations in the concentration of circulating thyroid hormone. rT3 analysis has the potential to identify situations when the diagnosis of hypothyroidism may be obscured by concurrent drug therapy, or potentially act as a marker for the drug action. Amiodarone has the most profound effect and is best studied. Unfortunately, serum rT3 concentration does not correlate with the antiarrhythmic benefits of amiodarone 53 so cannot be used in this context. Autoimmune hypothyroidism is common following amiodarone treatment occurring in over 5% of patients treated. As modest rise in serum TSH is also very commonly seen following the initiation of the iodine-rich amiodarone due to the Wolff–Chaikoff effect (thyroid hormone production is inhibited due to acute increase in the amount of available iodine), there is potential to over diagnose hypothyroidism in this context. rT3 has been suggested as a possible diagnostic aid, as the typical amiodarone-related increase in rT3 will not be apparent in hypothyroid patients. 54 However, the rise in TSH following amiodarone therapy rarely exceeds 20 mU/L 55 and typically normalizes within three months so, provided the modest increases in TSH is not over-interpreted serum TSH is likely to be adequate for the diagnosis of either drug-induced or concurrent hypothyroidism in this situation.
Ectopic expression of DIO3 and tumorigenesis
Overexpression of DIO3 in haemangioma causes a dramatic increase in rT3 and suppression of T3 and T4 with a concomitant increase in TSH.28,29 Serum rT3 can be useful in confirming the diagnosis and excluding other causes of hypothyroidism. Recent studies have shown that rT3 can increase tumour proliferation by binding at the integrin αvβ3 receptor. 33 As NTI often accompanies cancer concomitant rise in rT3 may act to support tumour growth. Whether NTI is a consequence of more aggressive cancers, or is independently associated with tumorigenesis awaits further study; however, NTIS has been shown to be a poor prognostic marker in lung cancer, 56 establishing a precedent for potential thyroid hormone-targeted therapeutic intervention. 34
Genetic variation and rare inherited syndromes
Genetic variation in the deiodinases
Common polymorphisms with DIO have been detected that cause an increase (Ala1814Gly) or a reduction (Cys785Thr) in the T3:rT3 ratio, consistent with higher and lower DIO1 activity, respectively. 3 While unlikely to be of clinical significance, this establishes DIO1 as a regulator of serum rT3.
SBP deficiency
All the deiodinase enzymes require post-translational modification to incorporate selenocysteine into the enzyme active site. This is achieved via a complex mechanism in which a stop codon in the deiodinase mRNA is over-ridden, and selenocysteine inserted via the actions of selenocysteine insertion sequence binding protein 2 (SECIS-BP2 or SBP2) and a transfer RNA specific for selenocysteine. This process is coordinated by a cis-acting element SECIS in the 3′ region of the selenoprotein mRNA sequences. While a total deficiency of selenoprotein is likely to be lethal, 57 mutations in SBP2 can cause partial deficiency syndromes with a variable phenotype depending on which selenoproteins are affected. These include growth retardation, myopathy, sensorineural deafness and primary infertility. The lack of thyroid deiodinase function presents with a characteristic thyroid function test of high serum rT3 and T4, and borderline low T3, which may the first indication that SBP2 action is impaired. 58
Allan Herndon Dudley syndrome
Mutations in the thyroid hormone transporter MCT8 are responsible for the X-linked mental retardation syndrome Allan Herndon Dudley syndrome (AHDS). 59 MCT8 is a specific thyroid hormone transporter that is widely expressed, but notably in the CNS, where it is the major thyroid transmembrane transporter. Cell culture studies show that the uptake of or both T4 and T3 are reduced in neuronal tissue, resulting in the activation of DIO2 and suppression of DIO3. As other transporters as well as MCT8 are expressed in the liver, the consequent increase in serum fT3 activates DIO1. This gives the pathognomonic pattern of thyroid function tests in AHDS with low serum rT3 concentration, borderline low T4 with high T3 and borderline elevations of TSH. 60
Thyroid hormone-binding proteins
As rT3 is extensively protein bound, mutations in the serum thyroid-binding proteins will affect serum rT3 concentration. As TBG is the most avid rT3-binding protein, 61 congenital or acquired TBG deficiency or excess will affect rT3 concentration. This effect is not well characterized, however, as the dramatic effect on serum T4 concentration is usually indicative of this condition.
As TTE and albumin are minor thyroid hormone-binding proteins, loss of function does not materially affect serum thyroid hormone concentration. Gain of function mutations, however, do impact on serum rT3 concentration, as both albumin and TTE have relatively high capacity, if not affinity, for rT3. Best described are the gain of function mutations in albumin that cause familial dysalbuminaemic hyperthyroxinaemia where mutation-dependant increases in serum rT3 concentrations have been demonstrated. These can be quite striking for the R221I mutation, 61 which can increase serum rT3 concentration 86-fold. Similar effects have been reported for gain of function mutations in TTE, which also present with increased serum rT3 as well as T4. 61
Thyroid hormone resistance
Two distinct clinical presentations of thyroid hormone resistance have been identified; these correlate with the tissue-specific expression of the two T3 nuclear hormone receptors THRα and THRβ. 62 Mutations in THRβ were described first in 1989 63 ; they cause an autosomal-dominant condition that is clinically silent or presents with features of short stature, attention deficit disorder and resting tachycardia. 64 As THRβ is expressed in the pituitary, the thyroid hormone feedback is reset at higher thyroid hormone concentration, giving a typical biochemical profile of thyroid hormones with non-suppressed TSH. While rT3 is not required to establish this diagnosis, its concentration in serum is elevated, along with that of T4 and T3. Conversely, rT3 analysis does aid the diagnosis of thyroid hormone resistance due to mutations in the THRα receptor which have only recently been identified. 64 THRα is expressed in the central nervous system, bone, heart, skeletal muscle and gastrointestinal tract. The clinical phenotype is more severe and presents with anaemia, constipation and growth and developmental delay. As THRα is not expressed in the pituitary, effects on thyroid function tests are usually subtle with borderline low T4 borderline raised T3 and TSH. Measuring rT3 (and the T3:rT3) ratio is of value in identifying thyroid hormone resistance α, as this is serum rT3 typically low and can add discriminatory power to an otherwise subtle changes in thyroid function tests. Changes in expression DIO1 or DIO3 expression are likely candidates for this effect; indeed, studies in THRα knock-out mice have demonstrated reduced expression of DIO3. 65
Conclusion
The routine measurement of serum rT3 is yet to find an application in the clinical laboratory. Analysis is of value in the investigation of rare genetic syndromes such as thyroid hormone resistance α and suspected tumour syndromes such as consumptive hypothyroidism due to haemangioma; this service can be offered via referral laboratories, rather than general clinical chemistry laboratories. rT3 analysis is likely to be of value in further defining the non-thyroidal illness syndrome, and as to whether some patients may benefit from intervention. While rT3 does not bind to the nuclear thyroid hormone receptors, the discovery that rT3 may bind to extranuclear receptors may allude to a more active physiological role for rT3. rT3 may have a direct effect on tumorigenesis, and the role of NTIS on this process is intriguing. Based on current literature, there is no evidence to support the routine measurement of rT3 as an aid to guide thyroxine replacement therapy.
Footnotes
Acknowledgements
This article was prepared at the invitation of the Clinical Sciences Reviews Committee of the Association for Clinical Biochemistry and Laboratory Medicine.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
DJH.
Contributor-ship
DJH wrote the article and SO reviewed, edited and proofed the article.
