Abstract
Background
Infliximab, a monoclonal antibody directed against tumour necrosis factor, is widely used in the treatment of chronic inflammatory conditions including Crohn’s disease and rheumatoid arthritis. Its use is limited by development of anti-infliximab antibodies, which can lead to loss of therapeutic efficacy. Serum infliximab and anti-infliximab antibody measurements have recently become routinely available in the UK. The study aimed to assess the clinical utility of antibodies as an adjunct to trough infliximab.
Methods
Serum trough infliximab was measured in 201 samples from 108 gastroenterology and rheumatology patients on maintenance infliximab therapy. Results were correlated with C-reactive protein concentrations. Total anti-infliximab antibodies were measured in 164 samples.
Results
The median (25th–75th percentile) trough infliximab was 3.7 µg/mL (1.2–5.2 µg/mL) and 23% of samples had a concentration ≤1 µg/mL. A notable proportion had positive anti-infliximab antibodies: 84/164 (51%), which subdivided to 85% and 28% with infliximab ≤1 and >1 µg/mL, respectively.
Serum C-reactive protein was found to be significantly higher where infliximab was ≤1 compared to >1 µg/mL (10 mg/L [<5–24 mg/L] vs. <5 mg/L [<5–8 mg/L], P < 0.01), although a strict correlation was not observed. The relationship between trough infliximab and C-reactive protein differed depending on antibody status and there was no association between C-reactive protein and the presence or absence of antibodies.
Conclusion
Our findings support measurement of anti-infliximab antibodies only in the context of low infliximab concentrations <1 µg/mL. A higher therapeutic cut-off may be relevant in patients with negative antibodies. Further work is indicated to investigate the clinical significance of positive antibodies with therapeutic infliximab concentrations.
Introduction
Infliximab (Remicade®) is a chimeric human-mouse monoclonal antibody directed against tumour necrosis factor-alpha (TNF-α) used in the treatment of various chronic inflammatory diseases, notably Crohn’s disease and rheumatoid arthritis. 1
Several studies have shown that infliximab therapy is effective for the induction of remission and prevention of relapse in patients with inflammatory intestinal2,3 and perianal fistulizing Crohn’s disease.4,5 Consequently, infliximab has been approved by NICE for the treatment of severe active disease in those who have an inadequate response to conventional therapy or have fistulizing disease. 6 Similarly, the efficacy of infliximab therapy in combination with methotrexate was demonstrated for rheumatoid arthritis, 7 and NICE recommend infliximab as a possible treatment for patients with active disease who have failed to respond to first-line drugs.8,9 More recently, the indications for infliximab have been extended to include psoriatic arthritis, psoriasis and ulcerative colitis.10–13
Infliximab is administered as an infusion over at least 2 h. A dose of 3 mg/kg is standard in the treatment of rheumatoid arthritis and 5 mg/kg in Crohn’s disease, ulcerative colitis and psoriasis, although these may be increased in cases of loss of response.6,12 Following a three-dose-induction regimen where infusions are administered at 0, 2 and 6 weeks, remission is maintained with further infliximab infusions at regular intervals, typically of 8 weeks. 14 This interval may be decreased down to four weeks to intensify treatment. 9 In episodic maintenance therapy, as formerly recommended by NICE in Crohn’s disease, 15 further infliximab is only given on relapse of the disease. However, it has been proposed that infliximab therapy is less beneficial when given episodically. 16
In spite of the success of infliximab treatment in many patients, there remains a significant failure rate. In some cases, this is attributed to the development of anti-infliximab antibodies that bind to infliximab with a neutralizing effect and lead to loss of therapeutic efficacy. Various reports have shown an association between the development of antibodies to infliximab and reduced or shortened drug efficacy in Crohn’s disease and rheumatoid arthritis, as well as an increased incidence of infusion reactions.17–20
While the complex pharmacokinetics of infliximab and the development of inactivating antidrug antibodies make it difficult to predict response to the drug based on dosing alone, a number of studies have demonstrated a correlation between serum trough drug levels and clinical response or duration of effect in rheumatoid arthritis18,21 and Crohn’s disease, 17 respectively. Furthermore, at nearly £1700 per standard infusion for a 73 kg patient, 12 infliximab therapeutic monitoring has been shown to be cost-effective compared to empiric dose escalation in inflammatory bowel disease with secondary treatment failure.22,23
With strong supporting evidence, therapeutic drug monitoring of infliximab is emerging in routine patient care in the UK and we, as a national referral centre, offer services for serum trough infliximab concentrations and anti-infliximab antibodies. However, optimal use and interpretation of these results are still being established and in particular, utility of anti-infliximab antibodies, now that they can be measured in the presence of detectable infliximab drug levels, needs further elucidation.
In this work, trough serum infliximab concentrations and anti-infliximab antibodies were measured in gastroenterology and rheumatology patients on infliximab. The cohort reflected a heterogeneous group of patients with different disease states and length of time on infliximab therapy. Data were correlated with C-reactive protein (CRP) concentrations. The aims were to review infliximab concentrations in this group, to evaluate the utility of measuring anti-infliximab antibodies and to relate these findings to current infliximab therapeutic drug monitoring algorithms.
Methods
Infliximab therapeutic drug monitoring was requested by Sandwell and West Birmingham Hospitals NHS Trust gastroenterology and rheumatology consultants on patients on infliximab seen over an 18 month period (2011–2013) as part of their routine clinical care. Samples were collected into plain gel tubes immediately prior to the infliximab infusion to determine trough drug levels. Relevant dosing information including, date and dose of previous infliximab infusion and infusion number were provided with each request.
Samples were centrifuged within 3 h of collection and serum stored at −20℃ until analysis. A proportion of samples was also assayed for anti-infliximab antibodies, including all those with infliximab concentrations ≤1 µg/mL and a random selection of those with infliximab concentrations >1 µg/mL. Preinfusion serum CRP results, where available, were recorded from the laboratory computer system.
Serum infliximab was measured by an in-house enzyme-linked immunosorbent assay (ELISA) method, as described by Ternant et al. 1 In brief, serum was added to a TNF-α coated microtitre plate, which had been previously saturated with bovine serum albumin solution. A sandwich complex was then formed with the bound infliximab by addition of a horse-radish peroxidase conjugated goat antihuman immunoglobulin G specific for the Fc fragment of infliximab. The reaction was detected following addition of substrate, o-phenylenediamine dihydrochloride, at 405 nm. Infliximab-spiked serum was used to produce a seven-point calibration curve from 0 to 12.5 µg/mL and further infliximab-spiked serum samples (0.4, 1.5, 4.0 and 10.0 µg/mL) served as internal quality control. All samples were assayed in duplicate. The between-batch coefficients of variation (CVs) for the assay (n = 8) were 8.6% at 0.4 µg/mL, 7.9% at 1.5 µg/mL, 8.5% at 3.9 µg/mL and 13.1% at 8.9 µg/mL.
The Immunodiagnostik total antibodies against TNF-α blocker (ADA) ELISA kit was used for the detection of serum anti-infliximab antibodies (Immunodiagnostik AG, Bensheim). The assay measured total antibodies and included a first step to dissociate the antibodies from infliximab. Serum was then incubated with a mixture of tracer and peroxidase conjugate antibody before being added to the plate, where the antibodies became bound to the precoating of TNF-α and infliximab. A washing step removed all unbound substances before tetramethylbenzidine substrate was added and the reaction detected at 450 nm. The kit contained three control samples, negative, cut-off and positive, which were run in duplicate. Patient samples were run in singlicate. Qualitative positive or negative antibody results were determined by relating the measured absorbance to that of the cut-off control, which had an assigned value of 10 AU/mL. The between-batch CV was 10.2% at 184 AU/mL (n = 17).
Throughout this work, results are expressed as median (25th–75th percentile) and Mann Whitney U analysis was used to compare datasets. A P value of <0.05 was deemed to be statistically significant. All statistical analyses were performed using Analyse-it for Microsoft Excel software.
Results
There were 201 serum samples from 108 patients (65 female) on maintenance infliximab therapy. Each patient had between one and five samples taken over the 18-month period. The patients had the following diagnoses: inflammatory bowel disease (61); rheumatoid arthritis (33); psoriatic arthritis (8); Behcets (2); sarcoidosis (2); ankylosing sponditis (1); scleroderma (1); uveitis (1) and no diagnosis (1). The maintenance dosing interval ranged from 5 to 11 weeks (75% with an eight-week interval), while the infliximab dose ranged from 3 to 10 mg/kg (27% and 54% on 3 and 5 mg/kg, respectively). Considering the first sample received from each patient, the number of previous infliximab infusions ranged from 3 to 65 (median [25th–75th percentile]: 16 [8–27]).
Trough infliximab concentrations
Median trough infliximab concentration for all samples was 3.0 µg/mL (1.1–6.1 µg/mL). Forty-six (23%) samples had infliximab concentrations ≤1.0 µg/mL, i.e. below the therapeutic cut-off currently used by our laboratory, while 24 (12%) samples had infliximab concentrations >10 µg/mL. There was no significant difference in infliximab concentrations between patients with a diagnosis of inflammatory bowel disease (2.9 µg/mL [1.1–6.6 µg/mL] n = 118) and those with rheumatoid arthritis (2.6 µg/mL [0.9–4.2 µg/mL] n = 67). Furthermore, although the trough infliximab concentrations were slightly higher in patients on a 5 mg/kg dose compared to 3 mg/kg, this did not reach statistical significance (3.4 µg/mL [1.1–6.9 µg/mL], n = 132 vs. 2.7 µg/mL [1.0–4.3 µg/mL], n = 62). Infliximab concentrations in patients with a <8-week dosing interval (5.0 µg/mL [1.5–5.0 µg/mL], n = 32) were found to be significantly higher (P = 0.04) than those on the standard dosing interval of eight weeks (2.8 µg/mL [1.1–5.7 µg/mL], n = 164). Meanwhile, lower infliximab concentrations were measured in those with <10 infusions (1.9 µg/mL [0.5–6.9 µg/mL], n = 50) compared to those who had been given ≥10 infusions (3.5 µg/mL [1.6–5.9 µg/mL], n = 133), but this did not reach statistical significance (P = 0.06). It was noted, however, that the spread of concentrations was greater in the lower number of infusions group.
Anti-infliximab antibodies
Anti-infliximab antibodies were assayed in 164 samples from 91 patients. The median trough infliximab concentration in this sample subset was 2.2 µg/mL (0.9–4.1 µg/mL). While 51% of samples had positive antibodies, when just the first sample from each patient was considered, 45% of patients had positive antibodies. Infliximab concentrations were significantly lower (P < 0.01) in the positive antibody group (1.1 µg/mL [0.5–2.4 µg/mL]) with respect to the negative antibody group (3.6 µg/mL [2.0–5.3 µg/mL]). Of the samples with infliximab concentrations ≤1.0 µg/mL, 85% had positive anti-infliximab antibodies, which compared to 37% in samples with infliximab >1.0 µg/mL.
Positive anti-infliximab antibodies in IBD and RA.
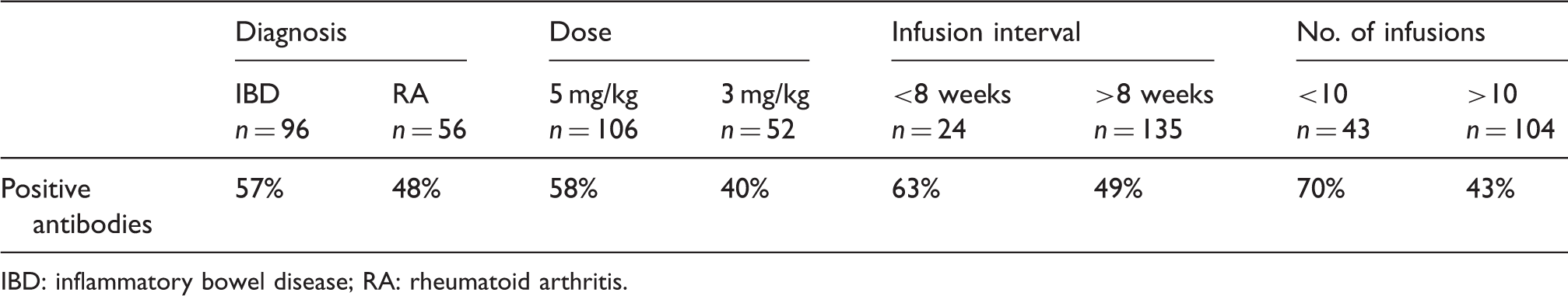
IBD: inflammatory bowel disease; RA: rheumatoid arthritis.
Comparing trough infliximab cut-offs
Comparison of different potential therapeutic infliximab cut-off values (µg/mL).
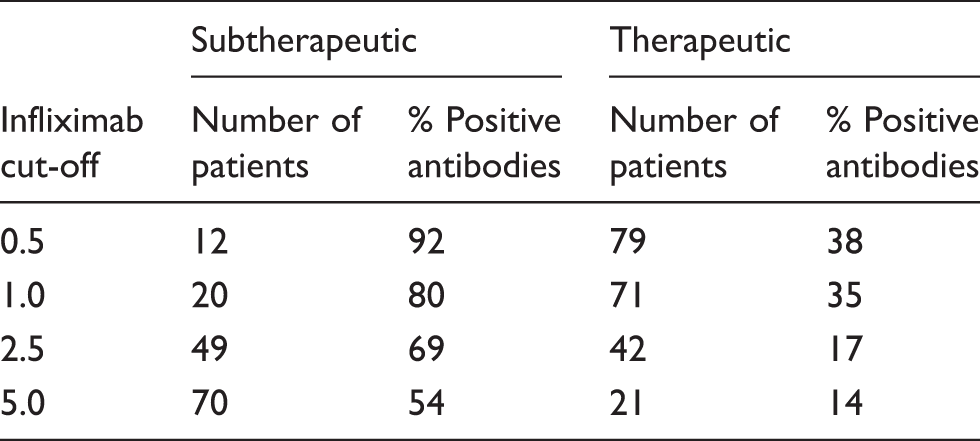
When the infliximab cut-off was lowered below 1.0 µg/mL, the percentage of positive antibodies was noted to increase to a greater extent in the subtherapeutic compared to the therapeutic group (12 vs. 3%) while upon increasing the cut-off to 2.5 µg/mL, the fall in positive antibody percentage was slightly more pronounced in the therapeutic group (18% compared to 11%). However, when the cut-off was increased to 5.0 µg/mL, there was only a very modest further reduction (3%) in the percentage of positive antibodies in the therapeutic group.
Correlation with serum CRP concentrations
There was a CRP measurement available in 172 samples from both inflammatory bowel disease and rheumatoid arthritis patients. Overall CRP concentrations were <5 mg/L (<5–12 mg/L). Figure 1 shows the relationship between trough serum infliximab and serum CRP concentrations, including the anti-infliximab antibody status where known.
Relationship between infliximab and CRP concentrations including antibody status where known; samples with negative antibodies are represented by grey circles, positive antibodies by white squares and unknown antibody status by black triangles. Undetectable infliximab concentrations are plotted as 0.2 µg/mL, over-range concentrations as 12.5 µg/mL and undetectable CRP concentrations as 5 mg/L.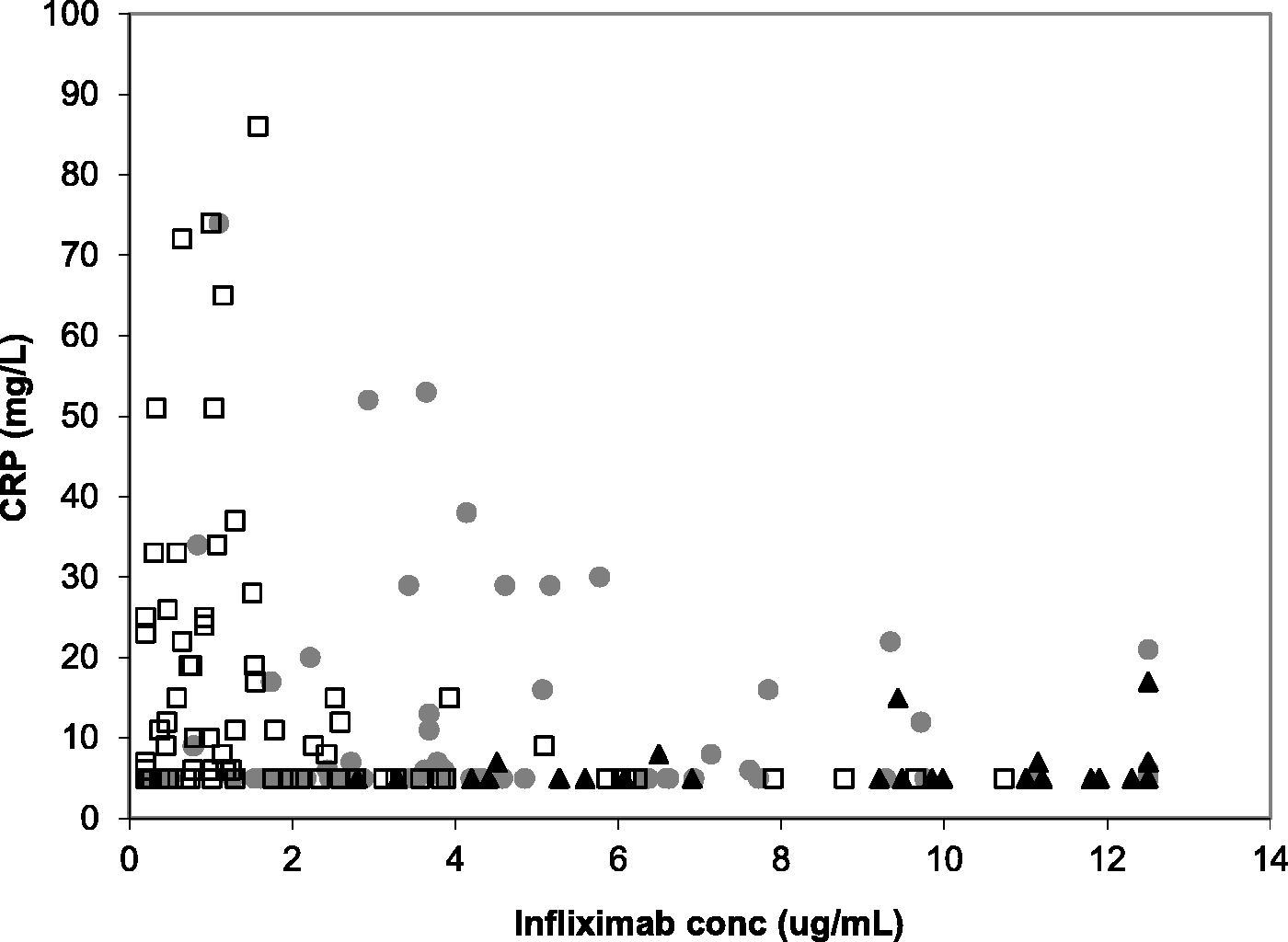
A spread of CRP results to much higher concentrations was observed at lower infliximab concentrations and this was reflected in the significantly higher (P < 0.01) CRP concentrations associated with subtherapeutic infliximab concentrations using the cut-off of 1 µg/mL: 10 mg/L (<5–24 mg/L), n = 38 vs. <5 mg/L (<5–9 mg/L), n = 134. However, a large number of samples with low infliximab concentrations and undetectable CRP were noted and there was no direct correlation observed between infliximab and CRP. Infliximab concentrations were significantly higher (P < 0.01) in samples with CRP <5 mg/L (4.2 µg/mL [2.0–7.8 µg/mL], n = 93) compared to those with CRP ≥5 mg/L (1.6 µg/mL [0.8–3.9 µg/mL], n = 93) while the percentage of positive antibodies was only modestly lower in the undetectable CRP group (44% [n = 68] vs. 59% [n = 74]).
There was no significant difference (P = 0.06) in CRP concentrations between the positive and negative antibody groups (7 mg/L [5–19 mg/L], n = 74 and <5 mg/L [<5–11 mg/L], n = 80, respectively). The relationship between infliximab concentration and CRP in the positive and negative antibody groups was of note. In the former, an infliximab concentration of 3 µg/mL clearly differentiated a subset of patients with higher CRP concentrations from another in which the vast majority had undetectable CRP concentrations. However, in the latter, the distribution of CRP concentrations was less clear cut; above an infliximab concentration of approximately 6 µg/mL, CRP was generally lower but there remained a large percentage of patients with detectable CRP concentrations.
Discussion
The study cohort reflected a heterogeneous patient group on infliximab in a UK secondary care setting, presenting with different diseases, infliximab doses, infusion intervals and number of previous drug infusions. Little data have been published so far on UK populations with typical dosing regimens. Furthermore, only a few studies to date include measurement of total anti-infliximab antibodies in the presence of therapeutic infliximab concentrations.
There was not a great spread in the trough infliximab concentrations measured in the group overall. Almost a quarter of samples had infliximab concentrations below 1.0 µg/mL even though the patients had not been selected on the basis of poor clinical response. This figure, however, compared well to 20% of patients in the rheumatoid arthritis ATTRACT study, 18 and 34% in a cohort of well-controlled rheumatoid arthritis patients described by Vande Mass et al. 24 Others have reported an even higher percentage (61%) of trough infliximab concentrations <1.4 µg/mL in ulcerative colitis patients, although this cohort had moderately severe to severe disease. 25 It was also noted in our study that high infliximab concentrations (>10 µg/mL) were measured in a considerable number of samples. While such concentrations may potentially result from samples that were not genuinely trough, this was deemed unlikely here since blood was collected preinfusion by the Specialist Inflammatory Bowel Disease and Rheumatology Nurses who also administered the infliximab. Hence, these results may indicate unnecessary exposure to an increased risk of side effects and wasted resources. 24
Trough infliximab concentrations did not vary significantly with a diagnosis of inflammatory bowel disease or rheumatoid arthritis or with different infliximab doses. The increased median infliximab concentration associated with shorter infusion intervals might be expected on the basis of there being less time for the drug to be metabolized. Interestingly, the shorter infusion interval group had a higher incidence of positive antibodies, perhaps explained by these patients having had their infusion interval reduced due to lack of response. Lower infliximab concentrations and a higher percentage of positive antibodies were found in patients who had had a lower number of previous infusions. Since steady state concentrations of serum infliximab are expected to be attained by dose four (first maintenance dose following induction therapy) 26 and all patients in the cohort were on maintenance therapy, the lower serum infliximab concentrations in this group are likely to reflect the increased incidence of positive antibodies. This supports the idea that patients at risk of an immunogenic reaction to infliximab do so within the first few weeks of treatment as has been described by Ungar et al. 27 ; herein, patients with inflammatory bowel disease who developed antibodies associated with loss of response did so within a median of 1.5 months of starting infliximab.
Nearly half of patients on maintenance infliximab therapy had positive anti-infliximab antibodies indicating that antibody development is a widespread problem. It was difficult to compare this finding with many literature reports, since until recently it has not been possible to reliably measure antibodies in serum with detectable infliximab concentrations.28,29 From studies of inflammatory bowel disease where antibodies were only measured in the absence of detectable drug levels, the incidence of positive antibodies ranged from 6 to 25% in scheduled therapy3,14 and 37 to 61% in episodic treatment.17,25,30,31 More recent studies utilizing assays capable of measuring antibodies in the presence of infliximab have shown earlier reports to underestimate the antibody prevalence in scheduled therapy, quoting values of 41–59%,24,27,32–34 to which our results compare well.
With an infliximab cut-off of 1.0 µg/mL, the majority of subtherapeutic infliximab samples did have positive antibodies, supporting a mechanism whereby antibody development leads to low infliximab concentrations. However, it was observed that a significant number of samples with low or undetectable infliximab had negative antibodies. Since compliance is not an issue here, other mechanisms must lead to the lower than expected trough infliximab concentrations. For example, very high levels of TNF-α may result in an increased proportion of bivalently bound infliximab, not detected by our assay. This is likely to reflect a clinically relevant decreased bioavailability of the drug. 34
A marked number of samples with therapeutic infliximab concentrations had positive antibodies. The percentage of antibody-positive patients, as opposed to samples, was also found to be similar, excluding the possibility that a few atypical patients were skewing the data. This may at least in part be due to the infliximab cut-off of 1 µg/mL not being suitable for this assay and/or population. However, increasing the cut-off to 5 µg/mL still resulted in a significant percentage of positive antibody patients in the therapeutic group. As serum factors are known to interfere in ELISA methods, 29 the possibility of false positive assay interference from non-specific binding of low-affinity antibodies, e.g. heterophilic antibodies or rheumatoid factor, cannot be excluded. However, similar findings have been described by other groups using different methodology.32,34
The clinical significance of positive anti-infliximab antibodies in the presence of therapeutic infliximab concentrations is not yet established. Recent references to transient antibodies,24,27,32,34 which disappear within 2–5 infusions and can account for 28–66% of antibodies measured, are likely to be relevant. Transient antibodies have been reported to appear later in the course of infliximab treatment with respect to persistent antibodies (median 13.5 vs. 1.5 months) 27 and are often not associated with a poorer clinical outcome. 32 Despite this, patients with sustained anti-infliximab antibodies and therapeutic infliximab concentrations may still be more likely to develop subtherapeutic drug levels in the future and further work is required to investigate this.
Serum CRP has been shown to reflect the clinical response to infliximab therapy;35–37 thus, CRP concentrations in this population were used to assess the suitability of 1 µg/mL as a therapeutic infliximab cut-off and to compare the utility of infliximab concentrations and anti-infliximab antibodies. The significant association between higher infliximab and lower CRP concentrations supports the idea that where infliximab is maintained at therapeutic levels, inflammation is reduced and there is improved clinical response. 36 Conversely, there was no statistical difference in CRP concentrations between positive and negative antibody groups. These findings suggest trough infliximab concentrations are more useful for therapeutic monitoring than antibody status. This view is held by Rutgeerts et al. 38 who propose undetectable infliximab trough levels are associated with unfavourable outcomes irrespective of antibody status and similarly, others have advocated low serum infliximab as being more directly associated with poor clinical response.28,32,39
The different relationship between infliximab concentrations and CRP in the positive antibody group compared to the negative antibody group was unexpected. In the former, low infliximab concentrations were clearly associated with higher CRP concentrations, in keeping with poor response, and higher infliximab with undetectable CRP, suggesting that the drug is working effectively despite the presence of antibodies. In the negative antibody group, however, the variation in CRP with high and low infliximab concentrations was less distinct and there was a surprising incidence of high CRP and intermediate or high infliximab concentrations. This suggests that infliximab is ineffective despite achieving therapeutic infliximab concentrations, which probably reflects non-TNF-α-mediated inflammation. 34 Based on these data, it may be that different infliximab cut-offs are applicable to patients with negative and positive anti-infliximab antibodies due to different underlying mechanisms of non-response. While the <1 µg/mL cut-off appears applicable for those with positive antibodies, this cut-off may be less relevant to the negative antibody group and further work is required to determine whether a higher infliximab cut-off (e.g. 3–5 µg/mL) would be more appropriate to indicate clinical response.
In those with low infliximab and low CRP concentrations, it is unknown whether infliximab is remaining effective at very low concentrations (i.e. trough concentrations are not representative of the entire infusion period) or disease remission is being maintained independently of infliximab, rendering further infusions inefficacious. Alternatively, not all patients will have a raised CRP during active disease and in such cases serum CRP is a poor indicator of response to infliximab. 37
A range of therapeutic trough infliximab cut-offs have been proposed in the literature and no consensus has been reached. Cut-off values, however, would need to be assay, disease and outcome specific. That of 1.0 µg/mL currently used by our laboratory was based primarily on data published from the rheumatoid arthritis ATTRACT study, 18 where infliximab concentrations >1 µg/mL were associated with a response of 50% or more. Klotz 8 also advocates a minimum target concentration of 1.0 µg/mL, while several other studies have verified an improved clinical response in inflammatory bowel disease with serum trough infliximab above 1 or 1.4 µg/mL.14,25,37 Other studies in inflammatory bowel disease have proposed cut-off values of 2–5 µg/mL.32,36,39,40
This work reinforces our current algorithm in which measurement of serum trough infliximab is the mainstay of therapeutic drug monitoring with assay of anti-infliximab antibodies only when trough infliximab is low. Our findings support an infliximab cut-off of 1 µg/mL as a threshold below which anti-infliximab antibodies are usefully measured; patients with positive antibodies are likely to benefit from a change in anti-TNF-α agent, whereas those with negative antibodies may show clinical improvement with dose intensification.17,18 With emerging reports of transient antibodies, it would be prudent to first ascertain antibody persistence rather than make treatment decisions on a single measurement. 41 The clinical relevance of positive antibodies in those with therapeutic infliximab concentrations is unclear and since patients with adequate infliximab concentrations and poor response are likely to be managed similarly (i.e. changed to a drug with a different mechanism of action) regardless of the presence of antibodies, 34 at present we would not advocate measuring antibodies when infliximab concentrations are therapeutic.
Conclusions
Infliximab results from a UK gastroenterology and rheumatology patient cohort were reviewed. The findings highlight the complexity of the relationship between trough infliximab and anti-infliximab antibodies and support measurement of antibodies only in the context of low or undetectable infliximab concentrations ≤1 µg/mL. Further work is warranted, first, to ascertain whether persistent positive antibodies in patients with therapeutic infliximab concentrations predicts the future development of low serum drug levels and loss of response, and second, to evaluate whether a higher cut-off is more indicative of clinical response in those with negative antibodies.
Footnotes
Acknowledgements
We acknowledge the Gastroenterology and Rheumatology Specialist Nurses at SWBH who facilitated collection of the samples with all of the correct information provided.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was not required for this study since the samples were collected for infliximab therapeutic drug monitoring as part of routine clinical care.
Guarantor
JB.
Contributorship
NB researched the literature, carried out the practical work, analysed the data and wrote the first draft of the manuscript. NB and PM designed the study. PM developed the sample collection protocol. JB provided the resources for the work. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
