Abstract
Background
Familial dysalbuminaemic hyperthyroxinaemia is an important cause of discordant thyroid function test results (due to an inherited albumin variant); however, the diagnosis can be challenging. A 51-year-old man had persistently elevated free thyroxine (T4), with discordant normal thyroid-stimulating hormone and normal free triiodothyronine. He was clinically euthyroid and had a daughter with similar thyroid function test results. We aimed to apply a whole protein mass spectrometry method to investigate this case of suspected familial dysalbuminaemic hyperthyroxinaemia.
Methods
Intact serum albumin was assessed directly using electrospray time-of-flight mass spectrometry. Results were confirmed using tryptic peptide m/z mapping and targeted DNA sequencing (exons 3 and 7 of the albumin gene). We also used this sequencing to screen 14 archived DNA samples that were negative for thyroid hormone receptor mutations (in suspected thyroid hormone resistance).
Results
Mass spectrometry analysis demonstrated heterozygosity for an albumin variant with a 19 Da decrease in mass, indicative of an Arg→His substitution. The familial dysalbuminaemic hyperthyroxinaemia variant was confirmed with peptide mapping (showing the precise location of the substitution, 218Arg→His) and DNA sequencing (showing guanine to adenine transition at codon 218 of exon 7). The same familial dysalbuminaemic hyperthyroxinaemia variant was identified in one additional screened sample.
Conclusions
Time-of-flight mass spectrometry is a novel procedure for diagnosing familial dysalbuminaemic hyperthyroxinaemia. The test is rapid (<10 min), can be performed on <2 μL of serum and requires minimal sample preparation.
Keywords
Introduction
Familial dysalbuminaemic hyperthyroxinaemia (FDH) is an important cause of discordant thyroid function test (TFT) results. It is due to an inherited (autosomal dominant) albumin variant exhibiting increased thyroxine (T4) binding1–3 and affects an estimated 1 in 10,000 (but may be more common in certain ethnic groups such as Hispanics).2,3 Despite the abnormal binding protein, FDH patients are euthyroid, and therefore treatment is unwarranted. 2
The diagnosis of FDH can be challenging; however, as free T4 levels are falsely elevated on some routine assays, 4 and current diagnostic tests are laborious (e.g. protein-binding studies using radiolabelled T4) or not widely available (e.g. equilibrium dialysis free T4 methods or genetic testing for FDH).2,3
We previously described a whole protein mass spectrometry (MS) method used to detect serum protein variants (in albumin and fibrinogen).5,6 We aimed to apply this method to investigate a case of suspected FDH.
Case report
A 51-year-old Caucasian man, had persistently (over at least five years) elevated free T4 levels, 27–29 pmol/L (reference interval [RI] 7–16), with discordant normal thyroid-stimulating hormone (TSH) 0.78–2.44 mU/L (RI 0.30–5.00) and free T3 4.8 pmol/L (RI 3.6–6.5) using Beckman assays (DxI; Beckman Coulter, Brea, CA, USA). He was clinically euthyroid with no history of thyroid disease (or medications known to interfere with TFTs) and interestingly, had one daughter, out of four children, with a similar TFT pattern (but he was not aware of any of his siblings or parents having similar results).
The discordant TFT results were confirmed by repeat analysis on a second immunoassay platform (Modular, Roche Diagnostics), which showed free T4 28 pmol/L (RI 10–20), free T3 5.2 pmol/L (RI 3.8–6.2) and TSH 1.99 mU/L (RI 0.30–4.00), with similar results following use of Heterophile Blocking Tubes (Scantibodies Laboratory). His total T4 concentration was 188 nmol/L (RI 55–140) and free T4 index 165 (RI 55–160) using in-house radioimmunoassays. Free alpha subunit concentration was normal and no mutations were identified on genetic testing for thyroid hormone resistance (sequencing exons 7, 8, 9, 10 of the thyroid hormone receptor-beta gene).
Methods
Reagents and materials
Serum (2 μL) was diluted in 20 μL water, 0.25 μL formic acid added (then microfuged) and 2 μL injected into an electrospray time-of-flight (TOF) MS system, consisting of an Agilent 1260 binary pump connected to an Agilent 6230 Accurate-Mass TOF liquid chromatography MS instrument (Agilent Technologies, Santa Clara, CA, USA) operating under MassHunter software. Chromatography was performed on a Luna C-8 5 μm (20 × 2 mm) column (Phenomenex, Torrance, CA, USA) with an acetonitrile/water solvent system (solvent A: 5% acetonitrile, 0.1% formic acid; solvent B: 85% acetonitrile, 0.1% formic acid; solvent gradient was run (300 μL/min) from 45 to 50% solvent B over 3 min, then to 100% solvent B over 1 min).5,6
Instrumental conditions: source gas temperature 300℃, capillary 3500 V, fragmentor 250 V, skimmer 50 V. Profile data were collected and multicharged spectral envelopes deconvoluted from 1050 to 1600 m/z using maximum entropy processing and BioConfirm (Agilent Technologies) software with an isotope width of 16.1 Da. 6
Peptide m/z mapping
Serum (20 μL) was precipitated with 14 μL of saturated ammonium sulphate and the supernatant dialysed against water before precipitation with an equal volume of acetone. The protein (800 μg) was digested with trypsin and taken up in 50% acetonitrile, 0.5% formic acid before direct injection (1 μg) into the TOF ion source at 50 μL/min. 6
DNA sequencing
DNA was extracted from whole blood (NucleoSpin Blood kit), primers for exons 3 and 7 of the albumin gene were used and the target DNA amplified on a Bio-Rad c1000 Thermal Cycler. DNA sequencing was performed on an Applied Biosystems 3130 Genetic Analyzer instrument (Thermo Fisher Scientific) and the sequence obtained was compared to the reference sequence NM_00077.5 using SeqScape software. Although the known FDH variants all occur in exon 7, sequencing of exon 3 was also performed as another variant affecting thyroid hormone binding (giving an increased total T3) has been described in this exon.2,3
We also used this sequencing to screen a further 14 archived DNA samples referred to our laboratory over nine years that were negative for thyroid hormone receptor mutations (in suspected thyroid hormone resistance).
Results
MS analysis of serum albumin from a normal control showed the expected major isoform at 66,439 Da (and its cysteinylated derivative at +119 Da, in excellent agreement with the theoretical values of 66,438 and 66,557 Da, respectively). The proband showed these same two isoforms but with additional companion peaks at −19 Da, suggesting he was heterozygous for an arginine to histidine (Arg→His) amino acid substitution (Figure 1). The precise location of this substitution was confirmed by peptide mapping, with the yield of the affected peptide (AWAVAR218, predicted m/z 673.4), decreased by approximately 50% and the presence of an ion from the new peptide (AWAVHLSQR, precisely at its predicted location of 569.8 m/z) indicating a 218Arg→His substitution. DNA sequencing of exon 7 of the albumin gene also confirmed a heterozygous guanine to adenine transition (c.725 G > A) at codon 218 (or p.Arg242His including the signal and propeptide).
TOF MS spectra (deconvoluted) of serum albumin. Serum results from the proband (a) and a second patient identified with FDH (c) are shown, with normal control plasma (b) for comparison. Axes show relative signal intensity versus mass. The major isoform (and its cysteinylated derivative) can be seen at 66,439 Da and 66,558 Da, respectively. These peaks are duplicated at −19 Da in the FDH patients suggesting a substitution of Arg→His.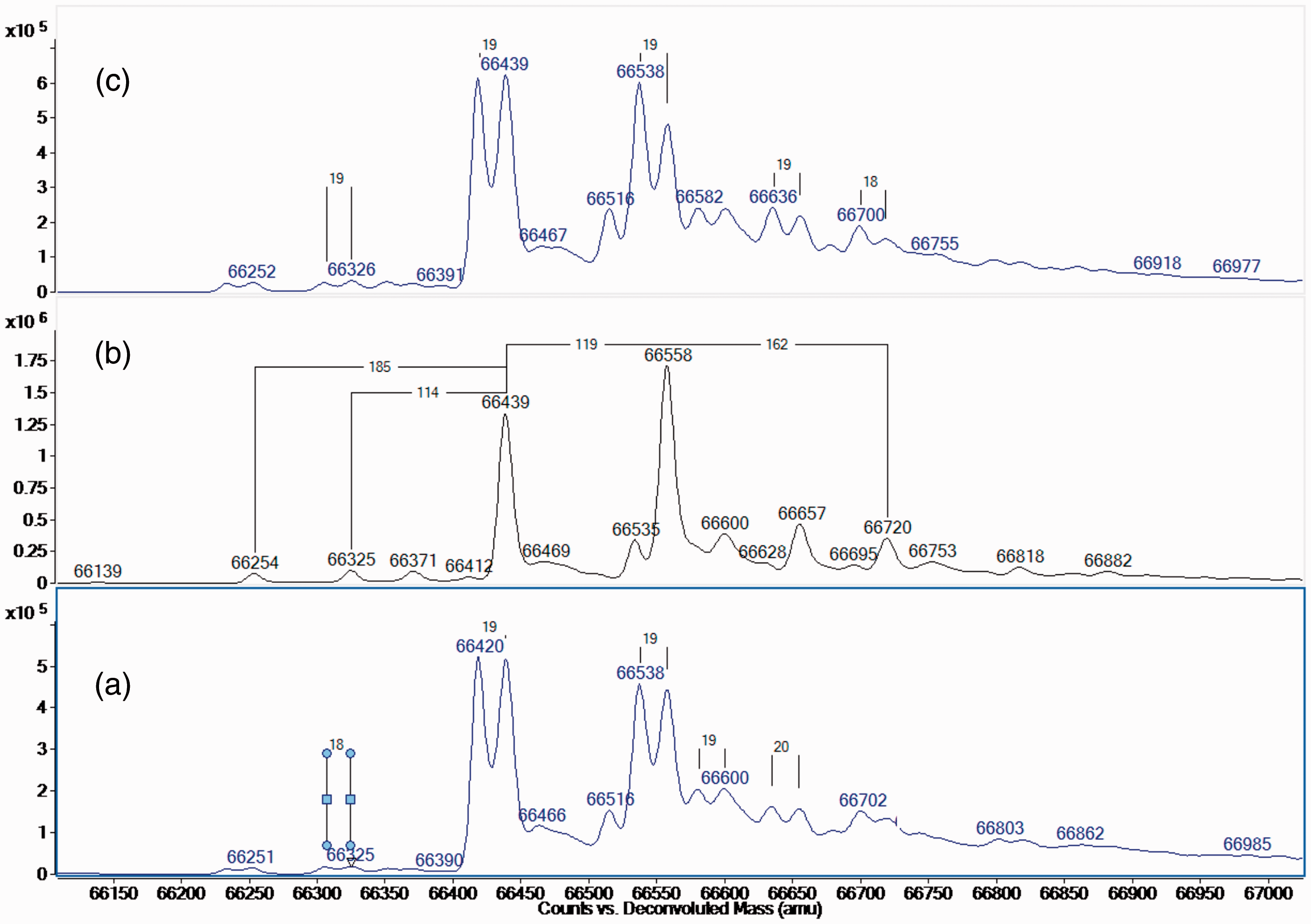
Results of thyroid function tests from the proband (A) and a second patient (B) identified with FDH (R218H variant), performed on four different assay platforms.
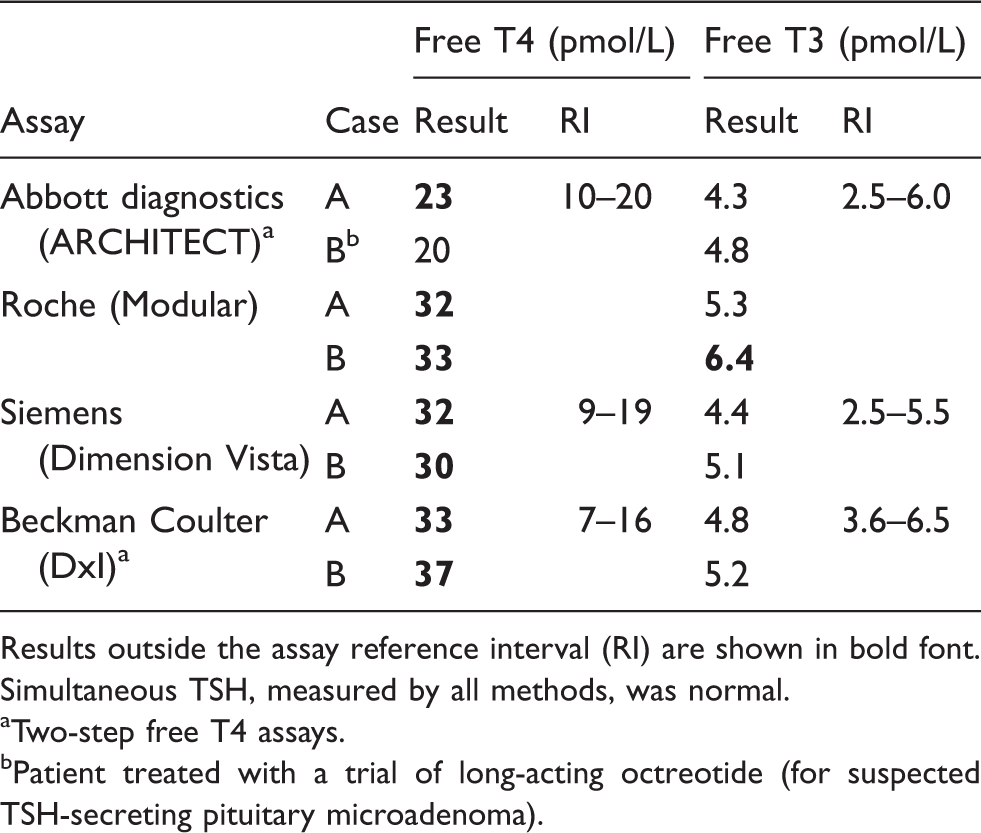
Results outside the assay reference interval (RI) are shown in bold font. Simultaneous TSH, measured by all methods, was normal.
Two-step free T4 assays.
Patient treated with a trial of long-acting octreotide (for suspected TSH-secreting pituitary microadenoma).
Discussion
Our study describes a rapid and novel whole protein MS approach used to diagnose FDH in two patients. This was supported by our MS data showing an albumin variant with the expected 19 Da decrease in mass (from an arginine to histidine substitution) in both patients. Tryptic digestion peptide mapping confirmed the location of the substitution (218Arg→His) and DNA sequencing also confirmed this variant (c.725 G > A).
The high-resolution TOF MS method used here has previously been used to identify serum protein variants (in albumin and fibrinogen)5,6 but has not previously been applied to FDH in the published medical literature.
The albumin variant detected, has increased T4 binding, due to reduced steric occlusion of the domain 2 ligand-binding site (because of the smaller side chain on the substituted amino acid). 3 The magnitude of the total T4 elevation found in the proband is consistent with a previous report of this variant (with increased total T4 levels to less than twice the upper reference limit, 1 although higher concentrations have been noted with some other FDH variants). 2
The three other causative FDH albumin variants reported include R218P (−59 Da mass change), R218S (−69 Da) and R222I (−43 Da) (and a further variant in exon 3, L66P (−16 Da), that gives a phenotype of familial dysalbuminaemic hypertriiodothyroninaemia).2,3 All of these variants have an amino acid substitution with a mass change of ≥16 Da and therefore would be readily detected, using the MS method described.
FDH is an important diagnosis to consider in clinically euthyroid patients with persistent discordant TFTs, after having excluded other sources of analytical interference and conditions such as TSH-secreting pituitary tumour and thyroid hormone resistance. As a separate issue, although FDH patients are asymptomatic, the albumin variant in question has been suggested to confer altered pharmacokinetics of certain drugs (e.g. warfarin) that bind to the modified region of albumin. 7
Free T4 levels in FDH may be falsely elevated on some assays.3,4 We found the Abbott assay (a ‘two step’ analog assay with an additional ‘wash step’ that removes binding proteins prior to adding T4 analog) gave borderline or only minimally elevated free T4 levels and this is consistent with a previous report. 4 More marked interference, however, can occur with certain two-step and one-step free T4 assays (Table 1).3,4,8 Reagent composition has been suggested as an important factor in determining FDH interference in free T4 assays particularly the concentration of ‘inhibitors of T4 binding to albumin’ such as chloride.4,8
The method described here is suitable for use in specialist clinical laboratories, as although assay time is <10 min and minimal sample preparation is required, significant expertise is needed to set up and maintain methods using MS. The sample requirements for this method are particularly favourable for investigating patients with discordant TFT results, as unlike genetic testing, this test may be applied to a small volume of the serum or plasma on which the TFT results were obtained. Furthermore, the method could also be used on archived frozen serum samples (with in-house data showing stability of >2 years in frozen serum).
Transthyretin, another thyroid hormone-binding protein, has also previously had two rare variants, with increased T4 binding described. 9 A similar approach to the one we describe for assessing albumin, may also be applied to assess serum transthyretin for the ≥28 Da mass change seen in these variants.
In conclusion, TOF MS provides a novel procedure for investigating patients with suspected FDH and has favourable test characteristics as it is rapid, can be performed on very small volumes (<2 μL) of serum and requires minimal sample preparation.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Written informed consent to publication could not be obtained from the presenting patient in the case report, as subsequent to our investigations, the patient died from an unrelated illness.
Guarantor
PMG.
Contributorship
JBR and SOB collected the data and researched the literature. JBR wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
