Abstract
The presence of latent fibrin clots is a recognised pre-analytical factor that causes inaccurate immunoassay results. This report details a case of a patient with Graves’ disease and congenital dysfibrinogenemia (CD) that had serum thyroid function test results (TFTs) that were not in keeping with clinical signs or symptoms. Analysis of plasma samples taken from the patient was shown to provide more accurate results than those obtained using serum samples. Further cases of patients with CD, all sharing the same genetic mutation of fibrinogen, and discordant TFTs are described, where TFTs measurement in serum samples proved to be unreliable. Despite evidence of fibrin effecting immunoassays, this is the first report of its kind linking CD to erroneous immunoassay results. The mechanism is postulated to be related to atypical forms of fibrinogen resulting in latent fibrin in serum samples blocking the antigen binding site and leading to incorrect results. Congenital dysfibrinogenemia is asymptomatic in most patients and therefore abnormal, albeit inaccurate, TFTs may be the first finding. Recognition of CD as a cause of discordant results is important when interpreting TFTs to avoid unnecessary investigations and inappropriate clinical interventions to those with the disorder and potentially identify undiagnosed cases.
Introduction
Thyroid function tests (TFTs) among the most commonly requested tests in Clinical Biochemistry due to the wide spectrum of presentations of thyroid disease. In the majority of patients the patterns of TFTs are straightforward to interpret and are consistent with the clinical impression of thyroid status. However, in a small number of patients, the interpretation of TFTs can be difficult with results differing with the clinical picture or incongruent with each other. The understanding of potential pitfalls in the measurement and interpretation of TFTs is crucial to avoid unnecessary investigations and treatments of such patients.1,2
Preanalytical factors have been recognised as a common cause of misleading results in biochemical testing. Interferences with immunoassays by several factors have been understood for some time.3,4 There have been several reports of erroneous results from immunoassays due the presence of fibrin in serum samples tubes after incomplete clot formation. Fibrin interference has been identified as the cause of inaccurate results in troponin assays in several instances5–7 and has also been observed in human chorionic gonadotrophin assays. 8 More recently there has been a report of inaccurate TFTs in patients receiving oral anticoagulant therapy. 9
This report describes unusual pattern of TFT results in a patient with an inherited disorder of the coagulation called congenital dysfibrinogenemia (CD) and comorbid Graves’ disease. Congenital dysfibrinogenemia is a condition in which subjects have abnormal fibrinogen leading to defective clot formation. When TFT analysis was performed on serum samples from the patient, the pattern of TFTs results was not in accordance with the patient’s thyroid status and TFT results altered upon re-analysis. Recognising the correlation between CD and the discordant TFTs measured in serum in this patient prevented further investigation and mismanagement of their existing thyroid disorder. Further to the index case, similarly discordant TFTs were obtained when serum samples were analysed in several patients with confirmed CD. The results had the potential to lead to further unwarranted investigations, misdiagnoses and inappropriate management if not recognised at time of analysis.
Despite the evidence of the impact of fibrin on immunoassays we have found no other reports of erroneous immunoassay results in patients with CD, and to our knowledge, this is the first report of this kind.
Case description
An 89-year-old female with a history of Graves’ disease and thyroid eye disease was having her thyroid function tests measured to evaluate response to carbimazole therapy. The patient had gradually been losing weight over the past 6 months and re-called lethargy and somnolence in this time period. Their thyroid eye disease was inactive but the patient had double vision in the left eye which required prism correction. The patient had a slight hand tremor, and heart rate of 68 beats per minute with a missed beat. Other medical history included diverticular disease, hypertension, gout, type 2 diabetes and congenital dysfibrinogenemia. Along with carbimazole, the patient was prescribed allopurinol, omeprazole and amlodipine.
Graves’ disease had been diagnosed 12 years earlier at age 77, when symptoms of thyroid eye disease developed. Thyroid receptor antibody (TRAB) were positive at the time of diagnosis and the patient had been managed by Endocrinology since. The daily dose of carbimazole had been reduced from 20 mg to 15 mg 7 months earlier due to the case coming to the attention of the laboratory when the patient’s TFT results were: thyroid stimulating hormone (TSH) 8.18 mU/L (0.35–5.00) and free thyroxine (FT4) 12.1 pmol/L (9.0–21.0). After the dose titration, the patient had TFTs measured on five separate occasions. In all of these instances the TSH results were significantly elevated, ranging between 11.2 and 23.6 mU/L and the FT4 measuring above or close to the top of the reference range, with results between 17.0 and 26.5 pmol/L. The team managing the patient believed the results to be unusual with the patient displaying hypothyroidal symptoms rather than that of central hyperthyroidism as predicted by the TFT results.
Patients 1’s TFT results in serum and plasma samples *Free T3 measured in pmol/L. Reference ranges for Roche Cobas methods: TSH = 0.27–4.2 mU/L, FT4 = 12–22 pmol/L, FT3 = 3.1–2.8 pmol/L.
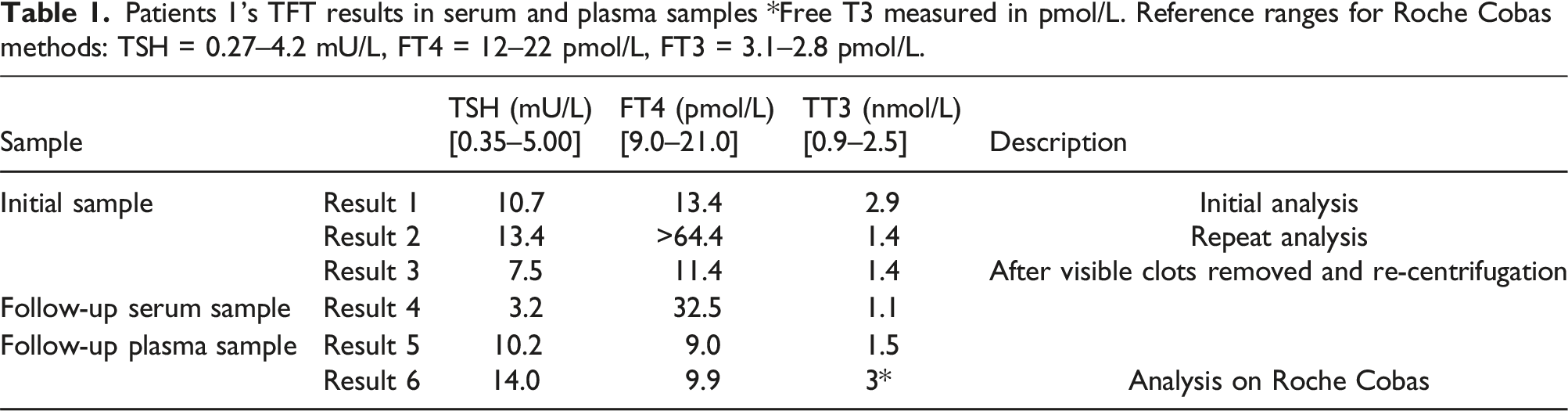
At this point it was considered that the TFT results observed may be related to the patient’s diagnosis of CD. The patient had a history of asymptomatic CD having been diagnosed following a routine coagulation screen prior to a surgical procedure 8 years previously and was found to be heterozygous for the pathogenic missense variant FGA c.104G>A (p.Arg35His). This co-diagnosis led to the hypothesis that the inconstancy between results was related to incomplete clot formation in vitro in the patient’s serum sample tube, potentially leaving residual fibrinogen in the patient’s serum after separation.
To investigate this theory a further fresh paired serum and lithium heparin plasma samples were obtained. The repeat samples taken 2 weeks after the initial discordant results, showed a marked difference between serum and plasma. The TSH and TT3 results in serum were within the reference range and FT4 significantly elevated (result 4, Table 1). The results obtained in plasma showed an elevated TSH, a TT3 within reference range and FT4 towards the low end of the reference range (result 5, Table 1). The plasma results were confirmed using an alternative TFT immunoassay, (result 6, Roche Cobas) as opposed to repeat measurement to the Abbott Architect assay used for the original analyses. Unfortunately the pattern of results obtained in serum could not be confirmed by an alternative TFT method due to insufficient sample volume. It was noted that there were differences between plasma results obtained by the different immunoassay methods used here: Roche Cobas and Abbott Architect results where the TSH result was 3.8 mU/L higher on the Roche Cobas TSH immunoassay. This difference is largely in keeping with method comparison data between the two laboratories with a where linear regression shows a relationship of Roche Cobas (Wishaw General) TSH = 0.79 Abbott Architect (QEUH) TSH +0.1133, R2 = 0.9948. Importantly the clinical interpretation of the TFT results are the same regardless of platform used with the TSH significantly above the upper limit of both reference ranges.
Further cases
TFTs in serum and plasma in patients 2–6 with genetically confirmed CD. Lithium heparin was used as an anticoagulant in all plasma samples unless notes to be EDTA.
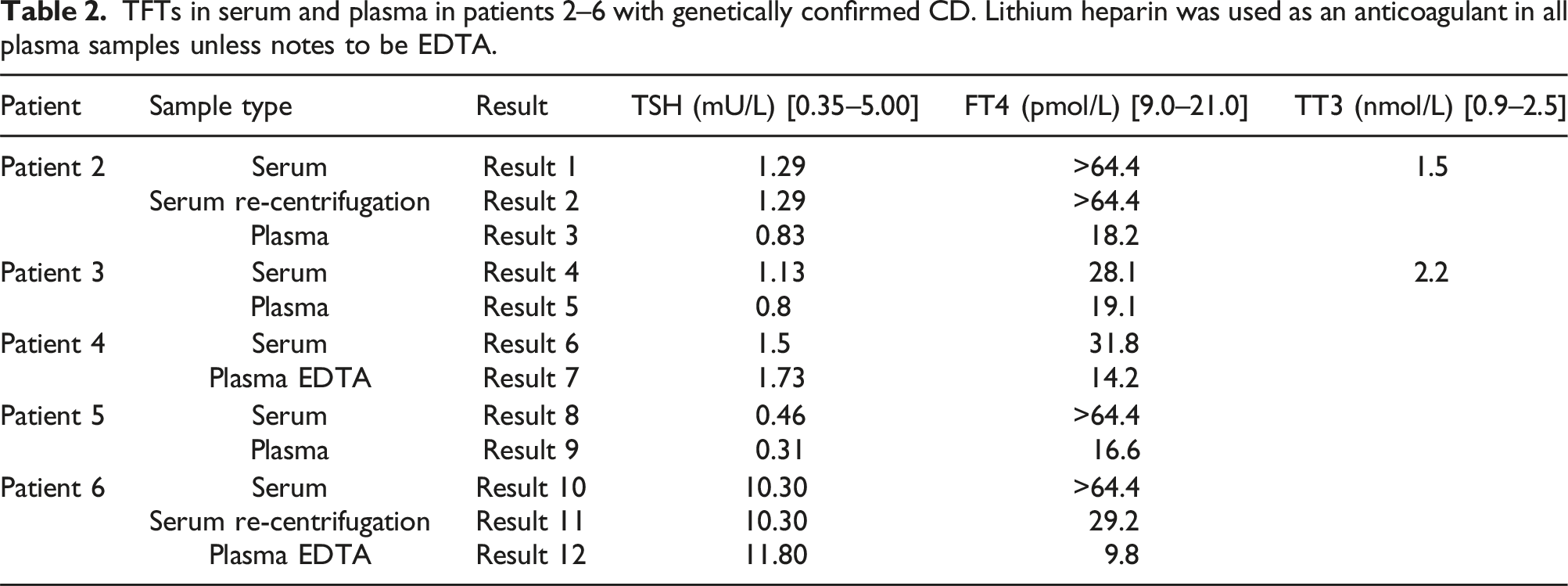
The appearance of the serum samples was variable in these patients. Some upon examination appeared to have visible clots which could be removed, but some did not. When visible clots were not observable it was noted that the samples appeared more viscous than a typical serum.
Re-centrifugation of serum samples under standard local laboratory practices (3000 RCF for 7 min at room temperature) followed by re-analysis of the serum samples gave somewhat unpredictable results. In the initial patient, patient 1, re-centrifugation after a visible clot was removed gave results that were comparable to the plasma samples (Table 1 result 3 and 5). This was opposed to patients 2 and 6 where no visible clots were observed. The samples serum samples on patient 2 and 6 were re-centrifugated but this process did not align serum and plasma results with (Table 2 result 1–3 and results 10–12) where the serum FT4 remained at >64.4 pmol/L after re-centrifugation in patient 2 and dropped from >64.4 to 29.2 pmol/L which is still considerable above the reference range. Evidently, this lack of consistency infers that this approach to handling samples is likely to result in inaccurate TFT results in some patients.
To establish whether the difference between serum and plasma samples observed in CD patient was also observed in non-CD subjects, an in-house study was performed. Euthyroid, healthy volunteers (n = 10) that did not knowingly have a diagnosis of CD, donated paired serum and lithium heparin plasma samples. When paired results were compared using the Wilcoxon signed-rank test there was no significant difference between FT4 (P = .275) and TT3 results (P = .557). A difference was observed between serum and plasma TSH results with plasma results marginally higher with the linear regression showing an equation of serum TSH = 0.8571 TSH plasma +0.02053 and R2 0.98809. Importantly, this did not change the clinical interpretation of the TSH in this cohort with all results remaining within the reference range.
For most patients in this series, this was the first incidence that TFTs had been measured; however, patient 5 had TFTs monitored over the course of a 10-year period. On the 11 occasions that the patient’s TFTs were assessed in this time period, there were four instances in which there were similar patterns of high FT4 with measurements of FT4 >64.4 pmol/L and TSH between 0.33 and 0.36 mU/L. There were no noted changes in sample handling within this time period. The patient has never been prescribed sodium levothyroxine and had no history of thyroid disease.
Materials and methods
All TFTS were measured on the Abbott Architect analysers using the Architect TSH, FT4 and TT3 chemiluminescent microparticle immunoassay (CMIA) at the Queen Elizabeth University Hospital, Glasgow. Where stated, TFTs were analysed on a Roche Cobas e801s analysers at Wishaw General Hospital, Lanarkshire. The TSH method in the latter was the Elecsys TSH, Elecsys FT4 III and Elecsys FT3 III methods.
All serum samples were collected in Greiner VACUETTE® Serum Separator Clot Activator tubes and plasma samples in Greiner VACUETTE® with lithium heparin or K3EDTA anticoagulants.
Sample handling at the Queen Elizabeth University Hospital Glasgow involves serum samples being left to clot for more than 30 min at room temperature followed by centrifugation on a tracked centrifuge by VWR Catalyst centrifuge set to 3000 relative centrifugal force (RCF) (3574 revolutions per minute (rpm)) for 7 min at room temperature. Re-centrifugation of samples was performed using standard local procedure for small or urgent samples. Samples were aliquoted to a separate tube and in a Hettich centrifuge at 2987 RCF (4000 r/min) for 5 min at room temperature.
Case discussion
Serum samples are the recommended sample type for the majority of biochemistry and immunodiagnostic tests in most healthcare settings. The observed results in patient 1’s serum samples were not in keeping with the patient’s clinical presentation. The patient had signs of hypothyroidism secondary to thyroid suppressing therapy, carbimazole, opposed to central hyperthyroidism as the TFTs pointed towards.
Accurate measurement of TFTs in patients who are treated for thyroid disorders such as Grave’s disease is essential for patient management. Both the FT4 and TSH appear to differ in between serum and plasma sample in patient 1 with the results from the plasma sample more in keeping with the clinical presentation. This was brought to the attention of the laboratory by clinicians with experience in interpreting TFT results who already have an awareness of the potential analytical errors and interferences in measurement. This expertise, however, cannot be expected by the majority of those requesting TFT analyses.
The proceeding cases presented here of patients demonstrates that the of inaccurate TFT results measured in serum samples is more widespread in CD patients. Interestingly, all the patient described in this report hold the same FGA gene mutation and showed inconsistent TFT results when both serum samples and plasma samples were compared. To establish whether there were similar inconsistencies in TFT results were also observed in those without CD, a small in-house study compared serum and lithium heparin plasma was performed and this showed no significant difference between FT4 and TT3 results and a marginal increase in plasma TSH results.
Re-centrifugation using local protocols, which are likely to be broadly similar to those employed in many diagnostic laboratories, did not appear to be reliable with the process not consistently aligning serum TFT results with those measured in plasma. It appears that the unusual TFT pattern is likely to be sporadic in nature when standard handling processes are adhered to.
Congenital dysfibrinogenemia (OMIN # 616004, CD) is a rare inherited disorder with heterogeneous clinical presentations but is frequently clinically silent and can go undiagnosed. Patients with the disease may exhibit bleeding diathesis, thromboembolic complications or sometimes both. 10 In a study of 250 identified dysfibrinogemia cases, 55% of patients were asymptomatic, 25% exhibited bleeding and 20% displayed a tendency to thrombosis, 11 with asymptomatic patients typically detected through abnormal coagulation tests or through family screening after a family member has been diagnosed. Naturally, due to the asymptomatic nature of many CD cases, the true prevalence has not been established. 10
Clearly the gap difference between plasma and serum result implies that abnormal forms of fibrinogen found in CD patients are not directly causative interference on TFTs observed in the results but more likely a downstream process. The clotting of serum within the sample tube is a process that happens in vitro and can be accelerated by clot activators. As in many diagnostic laboratories, our laboratory uses sample tubes that have coatings of microscopic silica particles on the interior walls that activate the clotting process, alongside a serum separator polymeric gel which acts as a barrier between the serum and the cellular contents of blood. 12 The process of blood clotting results in fibrin fibres being built up by the assembly of fibrinogen molecules in a process catalysed by thrombin. 13 In CD this process is altered. The atypical forms of fibrinogen in CD are deficient at binding thrombin and the fibrin clots that form are resistant to plasmin degradation. 10
Despite previous reports of fibrin interferences with different immunoassays, the underlying mechanism is poorly understood.5–7 Fibrin interferences in TFTs have previously been observed in patients receiving oral anticoagulant therapy. 9 Similarly to the patients in this case report, patients receiving warfarin therapy appeared to have falsely high FT4 results when serum samples were analysed which was not reproduced when plasma samples were analysed. The Abbott Architect FT4 method is, along with many FT4 assays, a two-step competitive immunoassay and the TSH method a sandwich immunoassay. In this report it has been repeatedly shown that FT4 results measured in serum samples are higher than those measured in paired plasma samples from CD patients. It is hypothesised that the process underlying the effect on FT4 is the same as suggested by Tokumaru M et al. (2023). The mechanism is proposed to be related to fibrin blocking the antigen binding site on the capture antibody. This blockade leads to less binding of the competitive T3 acridinium-labelled conjugate employed in the FT4 assay. Subsequently the reduced binding of conjugate yields a lower signal from the proceeding chemiluminescent reaction measured and hence a higher FT4 result. It is conceivable that the same effect could happen with sandwich assays and hence result in a lower signal and yield lower TSH results but this was not consistently observed here. Whether the abnormal forms of latent forms in CD is influential in these results or whether the route is only related to presence of any fibrin, it is hard discern.
The generalisability of this phenomenon to other immunoassays is unknown. Although not studied in these cases, immunoassays for other analytes, particularly the competitive ones, may to be susceptible to fibrin interference in patients with CD.
The cases described here share the same mutation in the FGA exon 2 c.104G>A (p.Arg35His). It has not been possible to confirm if other CD-associated mutations would have the same impact on fibrin formation and henceforth a similar pattern of TFT discordant results. However, given that the location of the mutation is on FGA Arg35 which is part of a mutation hotspot, 14 it is possible that other mutations within this hotspot may act similarly and potentially have the same behaviour in vitro in serum samples. CD is an inherited form dysfibrinogenemia but non-inherited dysfibrinogenemias are more common. It is unclear whether the mechanism behind this phenomenon is likely to result in misleading TFTs in patients with acquired dysfibrinogenemia but if so, then the impact could be much more widespread.
Conclusion
An appreciation of the potential for inaccurate TFT results in patients with CD should be considered when patients are having their thyroid function assessed. Analysis using plasma samples has been shown to be the most reliable approach to producing accurate results in those affected and should be used routinely in these patients when measuring TFTs. Due to the asymptomatic nature of CD in many patients, inaccurate TFTs may be the first finding in this patient group. An understanding of this phenomenon is important when interpreting TFTs and should be considered as a source of discordant TFTs results. This should be particularly borne in mind, if TFTs are incongruent with a patient’s clinical signs and symptoms and there is a family history of CD before embarking on the route towards investigating rare causes of hyperthyroidism.
Footnotes
Acknowledgements
Thank you to the Department of Biochemistry at Wishaw General Hospital in NHS Lanarkshire for helping investigating these patients and undertaking method comparison between the laboratories. Additionally, we are grateful to the willingly volunteers in the Department of Biochemistry, Queen Elizabeth Hospital Glasgow for to take part an in-house study comparing serum and plasma TFT results in non-CD.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
AB.
Contributorship
All authors, AB, CK and JM, confirm that they have contributed to the intellectual content of this paper and met the following three requirement (a) substantial contribution to the concept or design of the work; or acquisition, analysis or interpretation of data, (b) drafted the article or revised it critically for important intellectual content and (c) approved the version to be published.
