Abstract
The International Standard, ISO 15189 which specifies requirements for quality and competence in medical laboratories was written by medical laboratory professionals. Since its first publication in 2003, it has raised awareness of the importance of medical laboratories with governments throughout the world and provided a tool with which laboratories may develop their quality management systems and assess their own competence. This paper examines the use of the Standard in the context of self-assessment, accreditation and regulation of medical laboratories.
Keywords
The question implied in the title of this paper is that ISO Standards are an unnecessary imposition on a laboratory and it is hoped that a better understanding of the context in which standards can be used will result in a resounding answer of ‘absolutely not’. An equally appropriate question, to which the answer might be the same, is ‘Do you want to risk patient safety by running a laboratory that does not operate in compliance with the minimal requirements for quality and competence set out in ISO 15189’? 1 If there is a problem it is largely attitudinal, with standards and accreditation being seen as something imposed from outside rather than something to be embraced as a valuable tool for the management of the laboratory.
The author has previously described an ‘international model’ for accreditation and regulation of medical laboratories that traces the relationship of ISO 15189 to a medical laboratory, from its use by the laboratory as a ‘self assessment’ tool to its use by governments in the regulation and licensing of medical laboratories.2,3 The model (Figure 1) has three elements, and five stages A–E.
ISO 15189, accreditation and regulation of medical laboratories.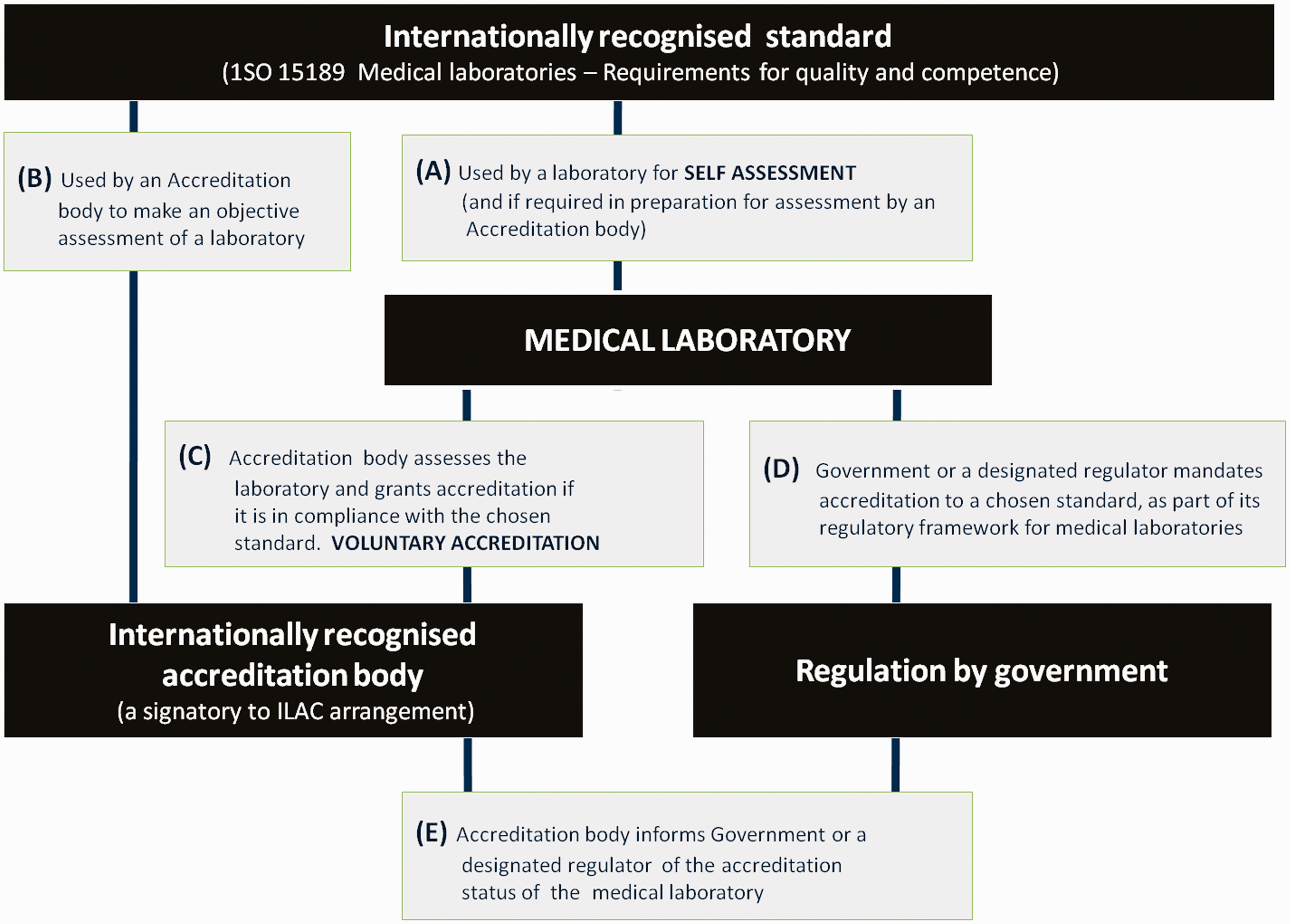
The first element is an ‘internationally recognized standard’, and ISO 15189 is recognized by the International Federation of Clinical Chemistry and Laboratory Medicine and by the International Laboratory Accreditation Co-operation (ILAC) for use by accreditation bodies in confirming or recognizing the competence of medical laboratories. ISO standards are developed by Technical Committees composed of experts that participate as national delegates, chosen by the ISO national member body for the country concerned. ISO itself is a network of the National Standards Institutes of 161 countries, one member per country, with the Central Secretariat in Geneva.
The second element is that accreditation be undertaken by an ‘internationally recognized accreditation body’ and the introduction to ISO 15189 states that ‘if the laboratory seeks accreditation it should select an accrediting body which operates to appropriate international standards and which takes into account the particular requirements of medical laboratories’. Those criteria can be met by an accreditation body that is a signatory to the so-called ‘ILAC mutual recognition arrangement’. Although within Europe a laboratory is required to use its own National Accreditation Body (NAB), it is important that the laboratory views itself as a ‘customer’ and the accreditation body as a ‘service provider’. NABs are required to operate an appeal mechanism should the laboratory be dissatisfied with the service provided. In the UK, the United Kingdom Accreditation Service (UKAS) is a signatory to the ILAC arrangement and the NAB.
The third element is regulation by Government. This is where Government or a designated regulator mandates accreditation to a chosen standard. In the UK, health is a devolved function of government, with England, Northern Ireland, Scotland and Wales each having independent regulators of health and social care services. For example, the Care Quality Commission (CQC) in England uses accreditation to ISO 15189 as part of its regulatory framework for laboratories. In other countries, such as France according to the order of 13 January 2010 and Law no.2013-442 of 30 May 2013 on a general reform of medical biology, all French medical laboratories have to be accredited in accordance with ISO 15189 standard in order to be able to carry out their activities. 4
The five stages in the relationship between the use of ISO 15189 and the laboratory are:
stage A, ISO 15189 is used by the medical laboratory for ‘self-assessment’ and, if required, in preparation for assessment by an accreditation body. stage B, ISO 15189 is used by an accreditation body to make an objective assessment of the laboratory. stage C, where the laboratory is assessed as working in compliance with ISO 15189 it is granted accreditation, a process that can be termed ‘voluntary accreditation’. stage D, where Government mandates accreditation to ISO 15189 as part of its regulatory framework. stage E, the accreditation body informs Government or the designated regulator of the accreditation status of the laboratory to provide evidence of fulfilment of a regulatory requirement.
Reaching stage E is often described as ‘mandatory accreditation’, but it is important to recognize that Government ‘mandates’ accreditation, while accreditation bodies provide assessment and grant accreditation under that mandate. Accreditation bodies do not have the authority to mandate accreditation. Accreditation is granted only to the requirements of the International Standard and the checking of compliance to other indicators should only be done within that context, i.e. that it is a requirement of the standard. When using performance or quality indicators, such as ‘critical value reporting’, to evaluate the significance of changes with time or across facilities it is important to understand the importance of sample size on the confidence limits that can be attached to estimates of performance. A suitable calculator is available from the Association for Clinical Biochemistry and Laboratory Medicine. 5
Whilst the individual elements of the model are inter-dependent, it is at its best when the elements function independently. In other words those who write standards, those who accredit and those who regulate have defined and independent roles. When this is so it provides the opportunity for Government to set regulatory requirements, which might include a requirement for a laboratory to operate in conformity with the International Standard ISO 15189 but, at the same time, remain at arms’ length from the assessment and accreditation process. In countries where there is no government regulation, it may still be explicit or implicit that medical laboratories be accredited in order, for example, to obtain contracts for the provision of services.
Finally, and most importantly, what should be the attitude of the medical laboratory and its leadership to participation in these processes? The author summarizes what he considers an appropriate approach as that taken in the virtual laboratory at St Elsewhere’s. ‘In common with many laboratories seeking accreditation, the laboratory at St Elsewhere’s used to ask, “Does this activity meet the requirements of ISO 15189?”, but now every time the laboratory undertakes a particular activity it asks “How does this activity ensure the quality of the service provided to the user?” This change in approach causes each member of staff to think carefully about the rationale behind each ISO 15189 requirement before implementing it'. 3
Footnotes
Declaration of conflicting interests
None.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Not required.
Guarantor
DB.
Contributorship
DB, the sole author.
